Toxicology Drug Screening Market Size and Share
Toxicology Drug Screening Market Analysis by Mordor Intelligence
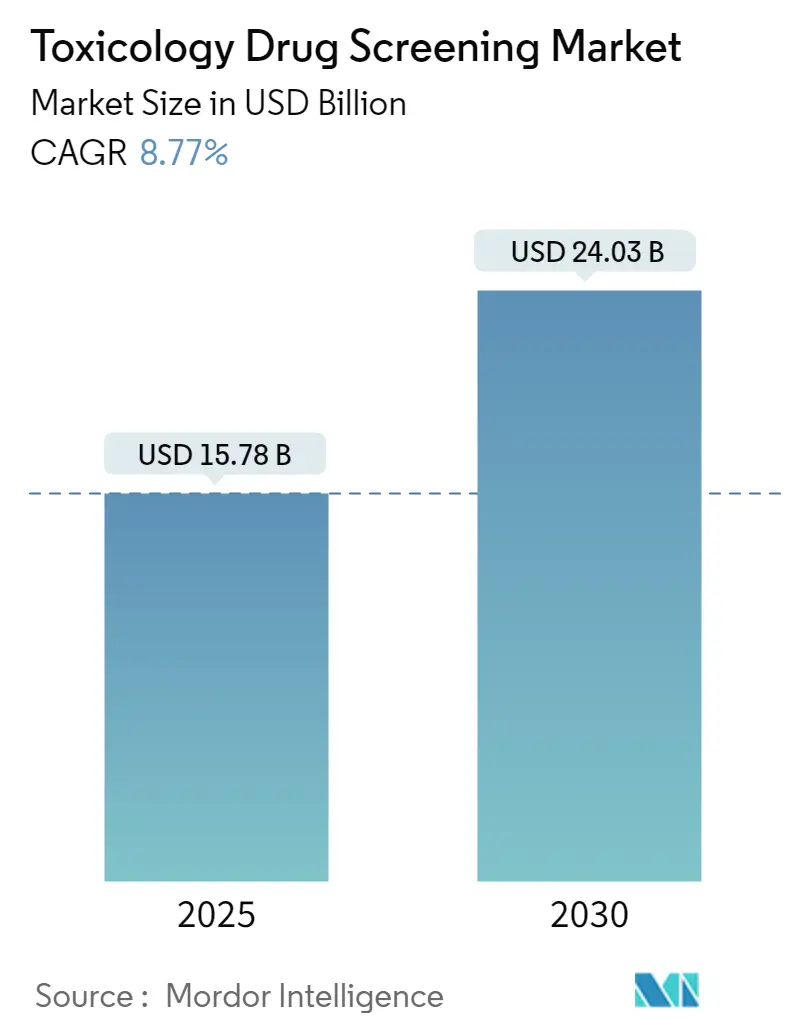
The Toxicology Drug Screening Market size is estimated at USD 15.78 billion in 2025, and is expected to reach USD 24.03 billion by 2030, at a CAGR of 8.77% during the forecast period (2025-2030).
The toxicology drug screening industry is experiencing significant transformation driven by the increasing prevalence of chronic diseases and the subsequent need for novel therapeutic solutions. According to Cancer Research UK's 2022 update, new cancer cases are projected to reach 27.5 million worldwide by 2040, highlighting the critical need for enhanced drug development and safety testing protocols. This disease burden has catalyzed substantial investments in preclinical testing infrastructure and advanced screening methodologies. The pharmaceutical industry's response to this growing healthcare challenge has been marked by increased emphasis on early-stage safety assessment and more comprehensive toxicology testing during drug development.
The landscape of drug safety assessment is evolving rapidly with the integration of advanced technologies and innovative screening approaches. A notable development in this direction was CN Bio's launch of the PhysioMimix 'in-a-box' reagent kit for non-alcoholic steatohepatitis (NASH) in April 2022, representing a significant advancement in organ-on-chip technology for drug screening. This technological progression has enabled more accurate prediction of drug toxicity and a better understanding of biological responses to chemical compounds in human tissues, leading to more efficient and reliable screening processes.
The biopharmaceutical sector's commitment to innovation is reflected in its substantial research investments, which, according to IFPMA's report, are 7.3 times higher than those of the aerospace and defense industries and 6.5 times more than the chemicals industry. The industry's R&D spending is expected to reach USD 213,000 million by 2024, demonstrating the sector's strong focus on developing new therapeutic solutions. This unprecedented level of investment has led to the emergence of sophisticated screening methodologies and more comprehensive safety assessment protocols, including advanced drug monitoring techniques.
The industry is witnessing a paradigm shift towards more efficient and accurate testing methodologies, with a growing emphasis on predictive toxicology and personalized medicine approaches. Contract research organizations and pharmaceutical companies are increasingly adopting integrated testing strategies that combine multiple screening methods to provide more comprehensive safety assessments. This evolution in testing strategies has been accompanied by the development of more sophisticated analytical tools and screening platforms, enabling better prediction of potential drug toxicity issues earlier in the development process. These advancements are pivotal in the realm of clinical toxicology, ensuring that drug testing services are both reliable and effective.
Global Toxicology Drug Screening Market Trends and Insights
Increasing Research and Development Activities
The pharmaceutical sector's commitment to research and development has become increasingly robust, with significant investments driving innovation in drug development and testing methodologies. The pharmaceutical sector's substantial investment of USD 83 billion in research and development in 2019 represents a tenfold increase compared to annual spending in the 1980s, highlighting the industry's growing focus on innovation and drug development. This increased R&D investment has created a greater need for comprehensive drug screening and toxicology testing services, as more drug candidates require thorough safety evaluation before proceeding to clinical trials.
The surge in R&D activities is further evidenced by the increasing number of drug approvals and regulatory submissions. According to the Regulatory Affairs Professionals Society (RAPS), the Center for Drug Evaluation and Research (CDER) approved 50 novel drugs in 2021, with 38 of these approvals occurring in the United States, demonstrating the robust pipeline of new therapeutic candidates requiring drug screening. This heightened level of drug development activity has created a substantial demand for advanced laboratory drug testing methods, as pharmaceutical companies seek to ensure the safety and efficacy of their drug candidates while optimizing their development timelines and resources.
Technological Advancements in Toxicology Drug Screening
The field of toxicology drug screening has witnessed significant technological evolution, particularly with the integration of advanced computational methods and innovative screening platforms. The emergence of in-silico methods has revolutionized drug target identification and analysis, enabling researchers to utilize sophisticated bioinformatics tools for analyzing target structures, generating candidate molecules, identifying binding sites, and optimizing molecular properties for improved binding characteristics. These technological advances have substantially enhanced the efficiency and accuracy of drug detection processes, allowing for more comprehensive safety assessments early in the drug development pipeline.
The integration of various cutting-edge technologies, including proteomics, genomics, and bioinformatics, has transformed the traditional drug discovery process. Modern toxicology screening now incorporates structure-based drug design, virtual screening, and de novo design methodologies, providing more sophisticated approaches to detecting drug toxicology. These technological advancements have enabled researchers to conduct more thorough safety assessments while significantly reducing the time and resources required for initial screening phases, ultimately accelerating the drug development process while maintaining rigorous safety standards.
Rise in Demand for Toxicology Drug Screening in the Pharmaceutical and Biotechnology Industries
The pharmaceutical and biotechnology industries have experienced a significant increase in demand for toxicology drug screening services, driven by the growing complexity of drug development and stricter safety requirements. The rising trend of outsourcing analytical testing by pharmaceutical and biopharmaceutical companies has created a robust market for specialized laboratory drug testing services, with companies seeking to leverage external expertise and advanced screening capabilities to enhance their drug development processes. This shift towards outsourced testing has been particularly pronounced in regions with well-established pharmaceutical industries and strong research infrastructure.
The increasing focus on early-stage toxicity detection has further amplified the demand for comprehensive screening services. Pharmaceutical companies are now placing greater emphasis on identifying potential safety concerns during the early phases of drug development, recognizing the significant cost savings and efficiency gains of early toxicity detection. This trend is supported by the strong presence of major service providers who continue to invest in advanced drug testing equipment and expanded testing capabilities, creating a more sophisticated and comprehensive toxicology screening ecosystem that better serves the evolving needs of the pharmaceutical and biotechnology industries.
Segment Analysis: By Testing Type
In Vitro Segment in Toxicology Drug Screening Market
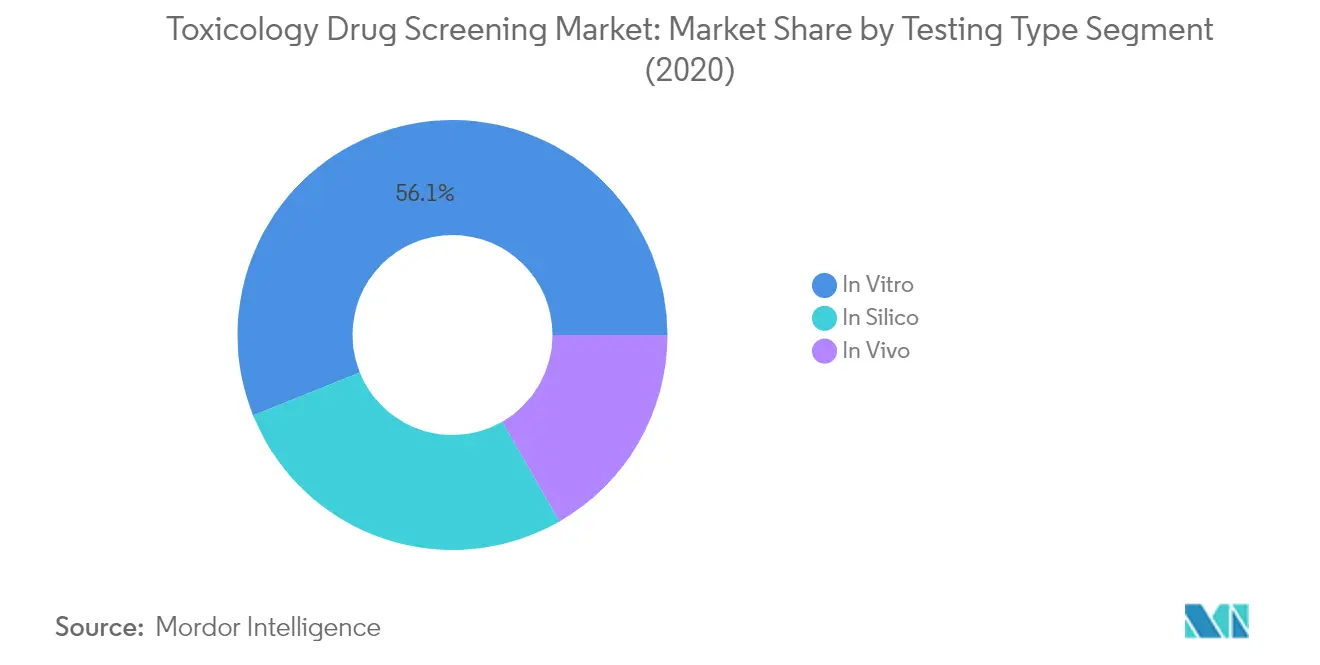
The in vitro testing segment dominates the toxicology drug screening market, commanding approximately 56% of the total market share in 2024. This significant market position is attributed to the growing opposition to animal testing in preclinical research, substantial advancements in in vitro toxicology assays, and increasing awareness regarding drug product safety. In vitro tests provide toxicity information in a cost-effective and time-saving manner, making them particularly attractive to pharmaceutical companies and research organizations. The segment's dominance is further strengthened by substantial government support for research, combined with specific reimbursement norms, which has led to the development of automated and technologically advanced toxicology instruments for laboratory analysis and in vitro examinations. Additionally, prominent players are actively expanding their market presence through strategic partnerships with smaller clinical research organizations and introducing innovative testing solutions.
Remaining Segments in Testing Type Market
The in silico and in vivo testing segments represent significant portions of the toxicology drug screening market, each offering unique advantages in drug development processes. The in silico segment leverages advanced computational methods and mathematical models to predict adverse health effects, while also helping identify drug targets using bioinformatics tools. This approach has gained traction due to its ability to reduce expenses, shorten time to market, and improve understanding of drug-receptor interactions. Meanwhile, the in vivo testing segment continues to play a crucial role in establishing toxicological profiles of novel drug candidates in the preclinical phase, particularly in pharmacokinetic and toxicokinetic research. These testing methods complement each other and provide comprehensive insights into drug safety and efficacy, making them essential components of the modern drug development process.
Segment Analysis: By Product
Instruments Segment in Toxicology Drug Screening Market
The instruments segment maintains its dominant position in the toxicology drug screening market, accounting for approximately 38% of the total market share in 2024. This significant market share is attributed to the increasing adoption of advanced analytical instruments in pharmaceutical and biotechnology companies for drug screening applications. The segment's growth is driven by continuous technological advancements in screening instruments, increasing automation in laboratory processes, and rising demand for high-throughput screening systems. The expansion of research and development activities in both developed and emerging markets has further strengthened the demand for sophisticated screening instruments, while the integration of artificial intelligence and machine learning capabilities in modern instruments has enhanced their analytical capabilities and efficiency.
Reagents and Consumables Segment in Toxicology Drug Screening Market
The reagents and consumables segment is projected to exhibit the highest growth rate of approximately 13% during the forecast period 2024-2029. This remarkable growth is primarily driven by the increasing volume of drug screening tests being conducted globally and the rising demand for specialized reagents in toxicology testing. The segment's expansion is further supported by the growing adoption of in vitro testing methods, which require a continuous supply of high-quality reagents and consumables. Additionally, the development of novel biomarkers and increasing focus on personalized medicine has created a sustained demand for specialized reagents, while the emergence of new drug development technologies and increasing research activities in emerging economies continue to fuel the segment's growth momentum.
Remaining Segments in Product Segmentation
The remaining segments in the toxicology drug screening market include software, animal models, and other products, each playing crucial roles in comprehensive toxicology testing. The software segment has gained significant importance due to the increasing digitalization of laboratory processes and the need for efficient data management and analysis tools. Animal models, while representing a smaller portion of the market, remain essential for specific types of toxicology studies and continue to be valuable in preclinical testing phases. The other products category encompasses various supporting technologies and tools that contribute to the overall toxicology screening workflow, including specialized testing kits, calibration standards, and quality control materials.
Segment Analysis: By Application
Immunotoxicity Segment in Toxicology Drug Screening Market
The immunotoxicity segment continues to dominate the global toxicology drug screening market, commanding approximately 32% of the total market share in 2024. This significant market position is attributed to the increasing focus on understanding immune system responses to various drug compounds and the growing importance of immunotoxicity testing in drug development processes. The segment's prominence is further strengthened by the rising demand for comprehensive immune system safety assessments in new drug candidates, particularly in areas such as biologics and immunotherapies. The expansion of this segment is also driven by technological advancements in immunotoxicity testing methods, including the development of more sophisticated in vitro testing platforms and the integration of artificial intelligence in toxicity screening processes.
Developmental and Reproductive Toxicity (DART) Segment in Toxicology Drug Screening Market
The DART segment is emerging as the fastest-growing segment in the toxicology drug screening market, with a projected growth rate of approximately 12% during the forecast period 2024-2029. This remarkable growth is primarily driven by increasing regulatory requirements for reproductive toxicity testing in drug development processes and growing awareness about the importance of evaluating developmental toxicity in pharmaceutical compounds. The segment's growth is further fueled by advancements in testing methodologies, including the development of more sophisticated in vitro models and the integration of advanced screening technologies. The rising focus on women's health and pediatric medications has also contributed to the increased demand for DART testing services, making it a crucial component of the drug development process.
Remaining Segments in Application Segmentation
The other significant segments in the toxicology drug screening market include systemic toxicity, endocrine disruption, and other applications. The systemic toxicity segment plays a crucial role in evaluating the overall impact of drug compounds on various organ systems, while the endocrine disruption segment focuses on assessing potential effects on hormonal systems. These segments are essential in providing a comprehensive toxicological profile of drug candidates. The remaining applications segment encompasses various specialized toxicity tests, including genetic toxicity, ocular toxicity, and neurotoxicity testing, which are vital for specific drug development programs and regulatory requirements. Each of these segments contributes uniquely to the overall market dynamics and plays a crucial role in ensuring drug safety and efficacy.
Toxicology Drug Screening Market Geography Segment Analysis
Toxicology Drug Screening Market in North America
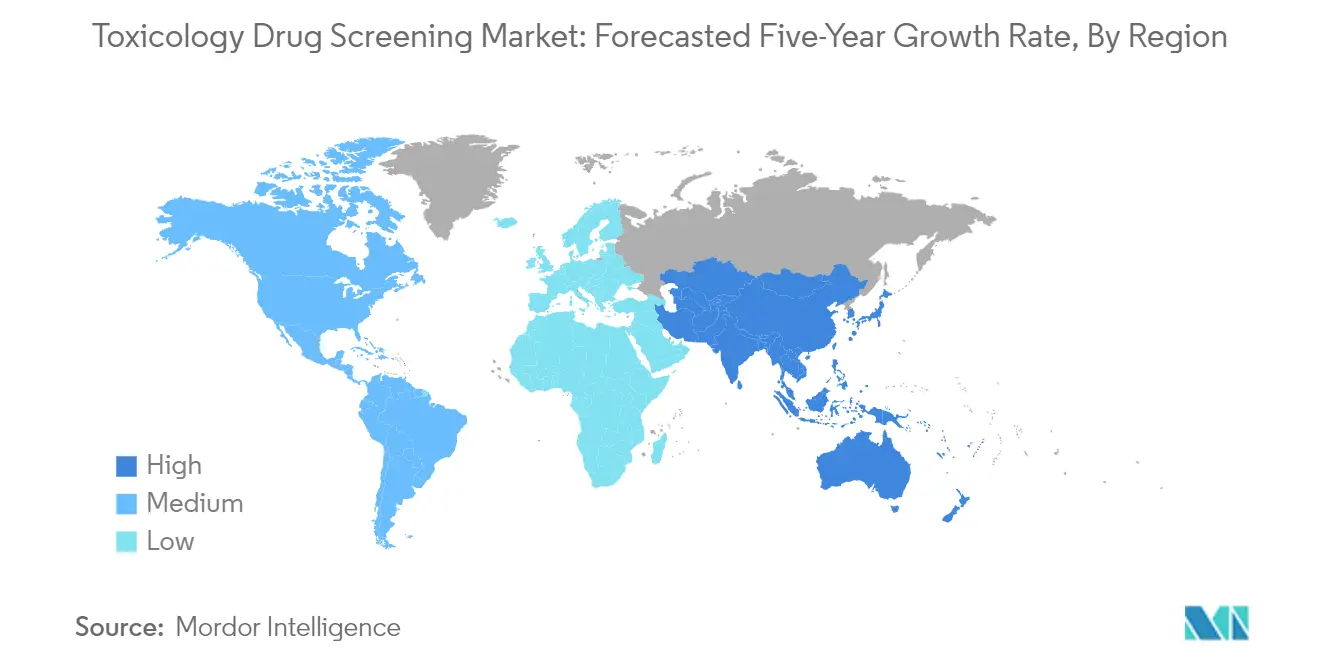
North America represents a dominant force in the global drug screening market, driven by advanced healthcare infrastructure, substantial research and development investments, and the presence of major pharmaceutical companies. The region benefits from well-established regulatory frameworks, particularly in the United States and Canada, which mandate thorough toxicology testing during drug development. Mexico is emerging as a growing market with an increasing focus on pharmaceutical research and manufacturing capabilities. The presence of leading contract research organizations and testing facilities further strengthens the region's position in drug testing services.
Toxicology Drug Screening Market in United States
The United States leads the North American market with approximately 79% market share in the region. The country's dominance is attributed to its robust pharmaceutical industry, extensive research infrastructure, and significant healthcare expenditure. The presence of major pharmaceutical companies, biotechnology firms, and research institutions drives continuous innovation in drug screening methods. The FDA's stringent regulatory requirements for drug safety assessment further boost market growth. The country's leadership in developing novel therapeutic solutions and implementing advanced screening technologies maintains its position as the largest market in the region.
Toxicology Drug Screening Market in Canada
Canada emerges as the fastest-growing market in North America with a projected growth rate of approximately 11% during 2024-2029. The country's growth is driven by increasing government support for pharmaceutical research, rising investments in healthcare infrastructure, and a growing focus on drug development initiatives. Canadian research institutions and biotechnology companies are increasingly adopting advanced toxicology testing technologies. The country's favorable regulatory environment and growing collaboration between academic institutions and pharmaceutical companies contribute to market expansion. The emphasis on developing innovative screening methods and increasing research activities in toxicology testing supports sustained market growth.
Toxicology Drug Screening Market in Europe
Europe represents a significant market for the toxicology testing market, characterized by strong research capabilities, advanced healthcare systems, and a robust pharmaceutical industry presence. The region benefits from well-established regulatory frameworks and significant investments in drug development research. Countries like Germany, the United Kingdom, France, Italy, and Spain lead the market with their advanced research infrastructure and growing focus on innovative screening technologies. The European market is further strengthened by increasing collaboration between academic institutions and pharmaceutical companies.
Toxicology Drug Screening Market in Germany
Germany maintains its position as the largest market in Europe, holding approximately 26% of the regional market share. The country's leadership is supported by its strong pharmaceutical industry, substantial research and development investments, and advanced technological capabilities. German research institutions and pharmaceutical companies are at the forefront of developing innovative drug screening methods. The presence of major contract research organizations and testing facilities, coupled with significant healthcare expenditure, reinforces Germany's dominant position in the European market.
Toxicology Drug Screening Market in United Kingdom
The United Kingdom emerges as the fastest-growing market in Europe with an expected growth rate of approximately 11% during 2024-2029. The country's growth is driven by increasing investments in pharmaceutical research, a strong focus on innovative drug development, and robust healthcare infrastructure. British research institutions and biotechnology companies are actively adopting advanced screening technologies and developing novel testing methods. The government's support for research and development activities, coupled with strategic initiatives to strengthen the pharmaceutical sector, contributes to the market's rapid growth.
Toxicology Drug Screening Market in Asia-Pacific
The Asia-Pacific region demonstrates significant growth potential in the drug screening market, driven by increasing research and development activities, a growing pharmaceutical industry, and rising healthcare investments. Countries like China, Japan, India, Australia, and South Korea are making substantial investments in drug development infrastructure. The region benefits from increasing government support, a growing presence of contract research organizations, and rising adoption of advanced screening technologies.
Toxicology Drug Screening Market in China
China leads the Asia-Pacific market as the largest country in terms of market size. The country's dominance is attributed to its expanding pharmaceutical industry, increasing research and development investments, and a growing focus on drug development. Chinese research institutions and pharmaceutical companies are rapidly adopting advanced toxicology testing technologies. The government's support for biotechnology research and development, coupled with the presence of major contract research organizations, strengthens China's position in the regional market.
Toxicology Drug Screening Market in India
India emerges as the fastest-growing market in the Asia-Pacific region. The country's growth is driven by increasing investments in pharmaceutical research, rising demand for innovative drug development, and a growing presence of contract research organizations. Indian pharmaceutical companies and research institutions are actively expanding their toxicology testing market capabilities. The government's support for pharmaceutical research and development, coupled with the country's growing role in global drug development, contributes to market expansion.
Toxicology Drug Screening Market in Middle East & Africa
The Middle East & Africa region shows growing potential in the drug screening market, with increasing investments in healthcare infrastructure and a rising focus on pharmaceutical research. The GCC countries and South Africa lead the regional market, with the GCC emerging as the largest market and South Africa showing the fastest growth. The region is witnessing increasing adoption of advanced screening technologies and a growing presence of research facilities. Government initiatives to strengthen healthcare infrastructure and rising investments in pharmaceutical research contribute to market growth.
Toxicology Drug Screening Market in South America
South America demonstrates growing potential in the drug screening market, with Brazil and Argentina leading the regional development. Brazil emerges as both the largest and fastest-growing market in the region, driven by its robust pharmaceutical industry and increasing research activities. The region benefits from growing investments in healthcare infrastructure, a rising presence of contract research organizations, and an increasing focus on drug development. Government initiatives to support pharmaceutical research and development, coupled with growing collaboration between academic institutions and industry players, contribute to market expansion.
Competitive Landscape
Top Companies in Toxicology Drug Screening Market
The competitive landscape is characterized by major drug screening companies demonstrating a strong commitment to innovation and technological advancement in drug screening solutions. Companies are actively expanding their research and development capabilities, with significant investments in developing new testing methodologies and automated platforms. Strategic partnerships with pharmaceutical companies, contract research organizations, and academic institutions have become increasingly common to enhance service offerings and market reach. Market leaders are focusing on geographical expansion through both organic growth and acquisitions, particularly in emerging markets. The industry has seen a notable trend toward developing integrated solutions that combine traditional screening methods with advanced analytics and artificial intelligence capabilities, while also emphasizing the development of specialized services for different therapeutic areas and regulatory requirements.
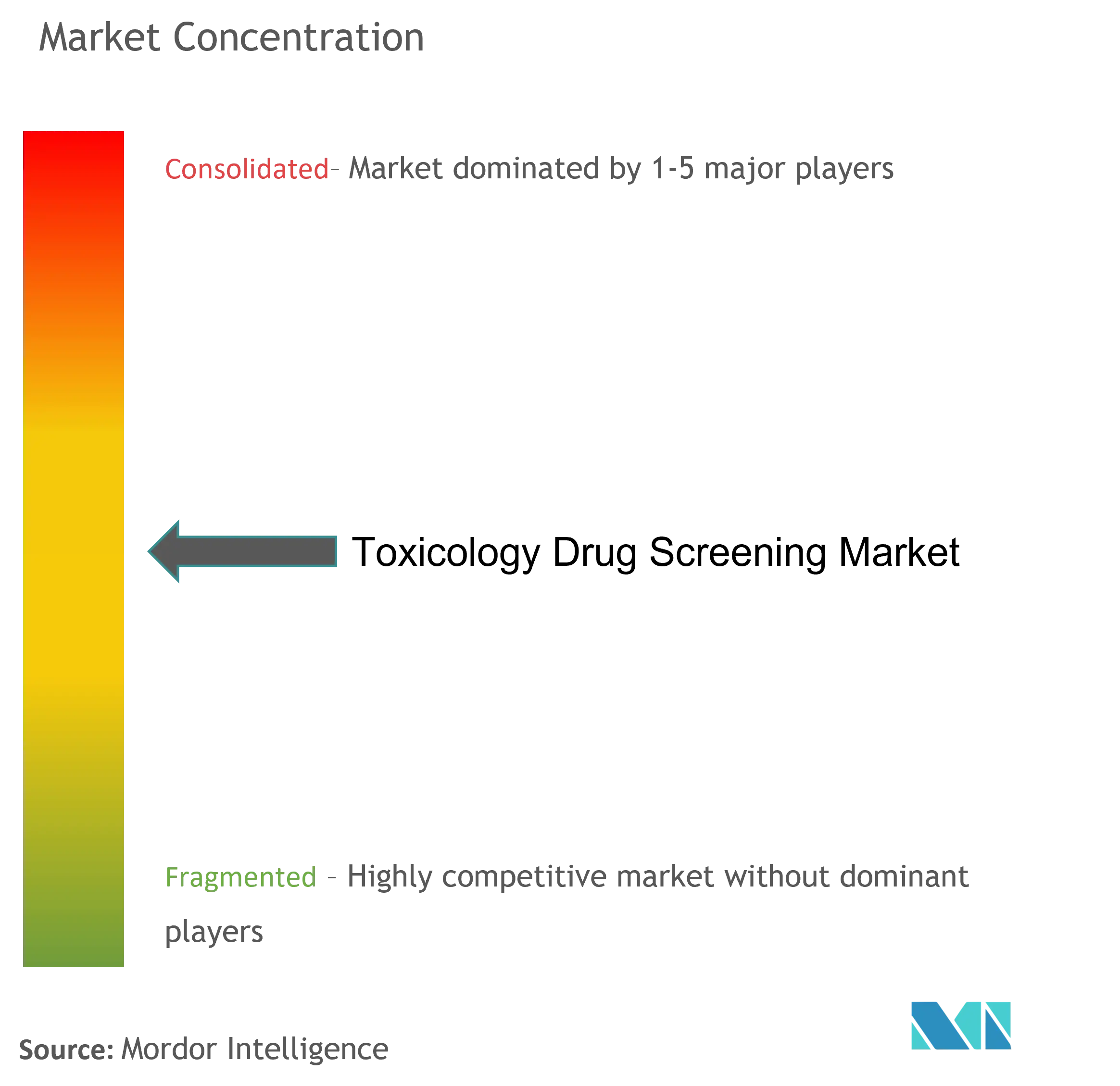
Fragmented Market with Strong Global Players
The toxicology drug screening market exhibits a highly fragmented structure with a mix of large multinational corporations and specialized regional players. Global conglomerates like Thermo Fisher Scientific, Danaher Corporation, and Merck KGaA maintain a significant market presence through their comprehensive product portfolios and extensive geographic footprint. These industry leaders leverage their established distribution networks and strong financial resources to maintain competitive advantages. The market also includes numerous specialized toxicology testing companies focusing on specific segments or regional markets, contributing to the competitive diversity.
The industry has witnessed considerable consolidation through mergers and acquisitions, particularly among larger players seeking to expand their technological capabilities and geographic presence. Companies are increasingly pursuing strategic acquisitions to gain access to new technologies, enhance their service offerings, and strengthen their market position in specific regions. This consolidation trend is particularly evident in emerging markets where established players are acquiring local companies to gain market entry and expand their customer base. The market structure encourages both vertical integration strategies by major players and specialization approaches by smaller firms focusing on niche segments.
Innovation and Integration Drive Market Success
Success in the toxicology drug screening market increasingly depends on companies' ability to offer comprehensive, integrated solutions while maintaining technological leadership. Incumbent players are focusing on developing end-to-end solutions that combine traditional screening methods with advanced analytics and artificial intelligence capabilities. Companies are also investing in building stronger relationships with pharmaceutical and biotechnology companies through customized service offerings and collaborative research initiatives. The ability to provide regulatory compliance support and adapt to evolving regulatory requirements has become a crucial differentiator in the market.
For new entrants and growing players, success lies in identifying and serving underserved market segments or developing specialized technological solutions. Companies are focusing on developing cost-effective drug testing services while maintaining high accuracy and reliability standards to address the growing demand from small and medium-sized pharmaceutical companies and research institutions. The market shows relatively low substitution risk due to the critical nature of toxicology testing in drug development, but companies must continuously innovate to maintain their competitive position. Regulatory compliance and quality standards remain crucial factors, with successful companies demonstrating strong capabilities in meeting evolving regulatory requirements across different regions.
Toxicology Drug Screening Industry Leaders
Agilent Technologies, Inc
Bio-Rad Laboratories, Inc
Eurofins scientific
Danaher
Laboratory Corporation of America Holdings
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- November 2022: Instem, a provider of information and technology (IT) solutions and services to the global life sciences market, updated its Computational Toxicology Software Suite solution. The 2022 software update allows its clients to access more than 600,000 toxicology studies for more than 200,000 chemicals, enabling fast, accurate, defendable, and regulatory-accepted predictions.
- June 2022: Thermo Fisher unveiled a new liquid chromatography-mass spectrometry (LC-MS) system for forensic toxicology to help forensic toxicologists, clinical research toxicologists, employee drug testing facilities, and wellness organizations keep pace with emerging and illicit drugs.
Global Toxicology Drug Screening Market Report Scope
As per the scope of the report, toxicology screening is a set of diagnostic tests conducted to evaluate the safety of potential drug candidates. The Toxicology Drug Screening Market is Segmented by Testing Type (In Vitro, In Vivo, and In Silico), Product (Instruments, Reagents and Consumables, Animal Models, Software, and Other Products), Application (Immunotoxicity, Systemic toxicity, Developmental and Reproductive Toxicity (DART), Endocrine Disruption, and Other Applications), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, and South America). The market report also covers the estimated market sizes and trends for 17 different countries across major regions globally. The report offers the value (USD million) for the above segments.
| In Vitro |
| In Vivo |
| In Silico |
| Instruments |
| Reagents and Consumables |
| Animal Models |
| Software |
| Other Products |
| Immunotoxicity |
| Systemictoxicity |
| Developmental and Reproductive Toxicity (DART) |
| Endocrine Disruption |
| Other Applications |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Testing Type | In Vitro | |
| In Vivo | ||
| In Silico | ||
| By Product | Instruments | |
| Reagents and Consumables | ||
| Animal Models | ||
| Software | ||
| Other Products | ||
| By Application | Immunotoxicity | |
| Systemictoxicity | ||
| Developmental and Reproductive Toxicity (DART) | ||
| Endocrine Disruption | ||
| Other Applications | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
How big is the Toxicology Drug Screening Market?
The Toxicology Drug Screening Market size is expected to reach USD 15.78 billion in 2025 and grow at a CAGR of 8.77% to reach USD 24.03 billion by 2030.
What is the current Toxicology Drug Screening Market size?
In 2025, the Toxicology Drug Screening Market size is expected to reach USD 15.78 billion.
Who are the key players in Toxicology Drug Screening Market?
Agilent Technologies, Inc, Bio-Rad Laboratories, Inc, Eurofins scientific, Danaher and Laboratory Corporation of America Holdings are the major companies operating in the Toxicology Drug Screening Market.
Which is the fastest growing region in Toxicology Drug Screening Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Toxicology Drug Screening Market?
In 2025, the North America accounts for the largest market share in Toxicology Drug Screening Market.
What years does this Toxicology Drug Screening Market cover, and what was the market size in 2024?
In 2024, the Toxicology Drug Screening Market size was estimated at USD 14.40 billion. The report covers the Toxicology Drug Screening Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Toxicology Drug Screening Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:








