Global Serological Testing Market Size and Share
Global Serological Testing Market Analysis by Mordor Intelligence
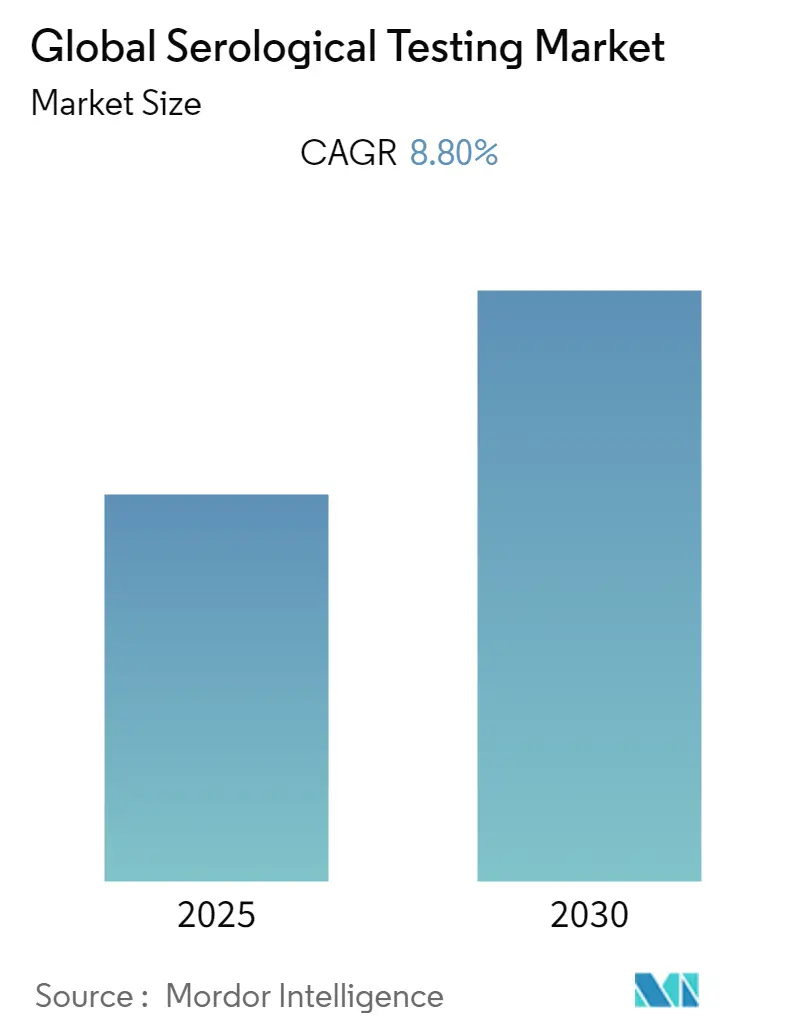
The Global Serological Testing Market is expected to register a CAGR of 8.8% during the forecast period.
The serology testing market is driving due to the rising incidences of COVID cases across the globe, and growing healthcare expenditure. However, a shortage of serology test kits for the examination is expected to hamper the growth of the global serology testing market. Moreover, the presence of several market players offering their products, increasing R&D investments for COVID analysis kits, and the presence of supportive government initiatives for rising research in this arena are anticipated to drive demand for the growth of the market for serology testing. For instance, the NCI Serological Sciences Network (SeroNet) is the United States' largest coordinated effort to study the immune response to COVID-19. The network aims to develop, validate, improve, and implement serological testing and associated technologies. Such initiatives will be expected to drive market growth during the forecast period.
Serological tests are important as they offer an additional piece of information to aid in characterizing possible prior exposure to SARS-CoV-2, especially since many infections are mild or asymptomatic in severity. The test analyzes blood, serum, or plasma samples for the presence of immunoglobulin M (IgM) and Immunoglobulin G (IgG) antibodies associated with the coronavirus (SARS-CoV-2). The detection of COVID-19 IgM antibodies tends to indicate recent exposure to COVID-19, and detection of COVID-19 IgG antibodies indicates a later stage of infection, so this combined antibody test could also provide information on the stage of the disease in patients. Thus, an exponential rise in the number of COVID-19 cases globally is boosting the demand for serological testing.
Moreover, the increasing product launches by key players for detecting the COVID pandemic crisis are further responsible for the growth of the market. For instance, in March 2020, Becton, Dickinson and Company, and BioMedomics Launched Rapid Serology Test to Detect Exposure to COVID-19. The new test, developed and manufactured by BioMedomics, is available through BD and distributed exclusively by Henry Schein, Inc. to health care providers throughout the United States.
Also, in August 2020, Transasia Bio-Medicals launched a test kit called ErbaLisa COVID-19 IgG ELISA in the Indian market. Similarly, in August 2020, Promega Corporation announced the launch of its new serological antibody test for COVID-19 to detect the presence of antibodies against the SARS-CoV-2 virus. A positive result may indicate whether an individual has been exposed to, and mounted an immune response to, the virus. Thus, the novel product development and approvals for serological tests for a variety of bacterial and viral infections will provide growth opportunities for the serological testing market during the forecast period.
Furthermore, the higher cases of infectious diseases such as hepatitis, tuberculosis, and HIV/AIDS globally is acting as a leading driver for the serological testing market. The high prevalence of hepatitis C infection is one of the key factors that has widened the growth prospects of the target market. For instance, globally, an estimated 58 million people were having chronic hepatitis C virus infection, with around 1.5 million new infections occurring per year. Additionally, World Health Organization estimated that in 2019, approximately 290000 people died from hepatitis C, mostly from cirrhosis and hepatocellular carcinoma (primary liver cancer). Thus, the increasing cases of hepatitis will likely increase the demand for these diagnostic tests over the forecast period, thereby driving the market.
Therefore, owing to the aforementioned factors the studied market is anticipated to witness growth over the analysis period. However, the Stringent Regulatory Scenario and Reimbursement Policies would impede the growth of the serological testing market during the forecast period. The lack of serology test kits for examination would pose a challenge to the growth of the market in the above-mentioned forecast period.
Global Serological Testing Market Trends and Insights
Consumables and Reagents are Expected to Witness Growth Over the Forecast Period
The reagents and consumables hold a significant share in the product segment of the Serological Testing Market in 2021 and are anticipated to register the highest CAGR in the market during the forecast period. The reagents and consumables are holding a significant share in the market segment due to the factors such as their higher adoption in the assays for achieving accurate and reproducible results. For instance, consumables such as the Fisherbrand accuWash and accuWash Versa microplate washers by Thermo Fisher Scientific, Inc. are used in many applications such as ELISA, cell-based assays, and microsphere-based assays. This washer is quiet, easy to clean, and leaves no residue, preventing contamination and reducing background. It removes reagents from microplate wells during assay wash steps. Thus, the advantages offered by the consumables are rising demand for the product which is driving the growth of the market.
Moreover, the increasing serological test is generating the demand for consumables and reagents which is further driving the growth of the market. For instance, as per world health organization (WHO) data, the serological test rate has increased by 16.1% for the year 2020 as compared to 2019. Thus, the increasing serological testing is generating requirements for consumables and reagents for the testing process which is boosting the growth of the market.
Market players are involved in the development and launch of novel products to increase their market presence. In 2020, Bio-Rad Laboratories, Inc has announced the launch of its in vitro diagnostics VIROTROL SARS-CoV-2 and VIROCLEAR SARS-CoV-2 positive and negative quality controls for use in antibody testing of SARS-CoV-2, the virus associated with COVID-19. Additionally, in 2021, a new serological test for Covid-19 antibodies was launched to the market, developed by researchers from the Spanish Council for Scientific Research (CSIC), in collaboration with the Madrid Hospitals of La Princesa and La Paz, and produced by the Spanish biotechnology company Immunostep. Such launches are expected to increase segment revenue over the forecast period.

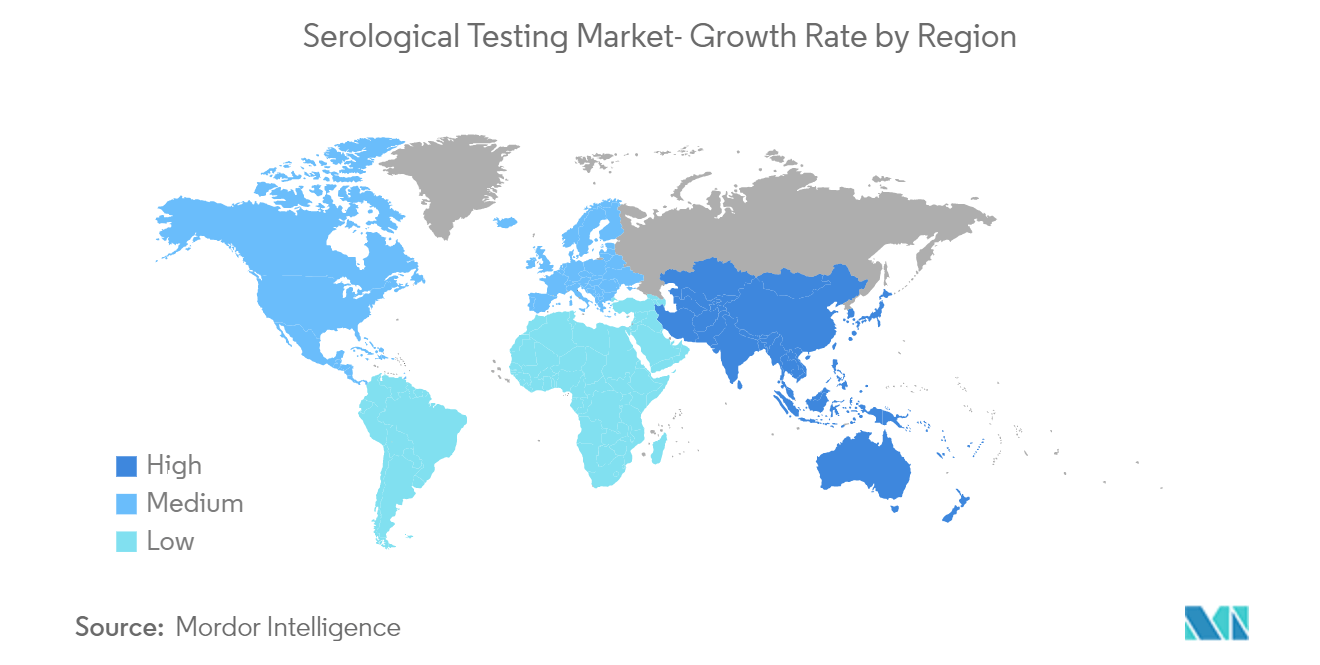
Europe is Expected to Hold Significant Share in the Serological Testing Market
Europe is expected to hold a significant share of the market due to the growing prevalence of infectious diseases, well-established health care infrastructure, and an increase in the number of geriatric populations in the region. Increasing cases of coeliac disease are driving the growth of the market in the region. For instance, in 2020, as per the Association of European Coeliac Societies (AOECS), around 1 in 100 people were estimated to suffer from coeliac disease in Europe. Applying that ratio, it was assumed that more than 7 million people were affected by coeliac disease across Europe, with only approximately 25% of those receiving a diagnosis. Serologic testing is very useful for screening patients with suspected coeliac disease. Early diagnosis is essential to prevent complications of coeliac disease. Thus, the increasing cases of coeliac disease are further driving the growth of the market in Europe.
As per the European chronic disease alliance article published in 2020, Chronic diseases, such as heart disease, stroke, cancer, chronic respiratory diseases, and diabetes, are by far the leading cause of mortality in Europe, representing 77% of the total disease burden and 86% of all deaths. Similarly, in 2020, about2.7 million people in the 27 European countries were diagnosed with cancer, and nearly 1.3 million died from it as per the report published by Joint Research Centre in 2020. Thus the increasing burden of chronic diseases in the region is boosting regional growth.
Additionally, the increasing product launches by the major players in this region are further driving the growth of the market. For instance, in 2020, F. Hoffmann-La Roche Ltd. launched its Elecsys Anti-SARS-CoV-2 serology test to detect antibodies in people who have been exposed to the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Similarly, in May 2020, IDvet launched a new serological test to detect antibodies against COVID-19 .This diagnostic kit is based on the ELISA technique and is designed for high-volume testing in laboratories.
Similarly, in April 2020, Eurofins Technologies announced the CE-IVD marking of the first range of testing kits for serology-based antibody detection by ELISA of patients who have been exposed to COVID-19. The development of the assays has been spearheaded by affiliate companies Gold Standard Diagnostics Inc., ENVIROTECH Diagnostics GmbH, NovaTec Immundiagnostica GmbH, and Ingenasa (Inmunologia Y Genetica Aplicada SA). Thus, the increasing novel launches for serological tests for a variety of bacterial and viral infections would provide growth opportunities for the serological testing market in the European region.
Therefore, owing to the aforesaid factors the growth of the studied market is anticipated in the European Region.
Competitive Landscape
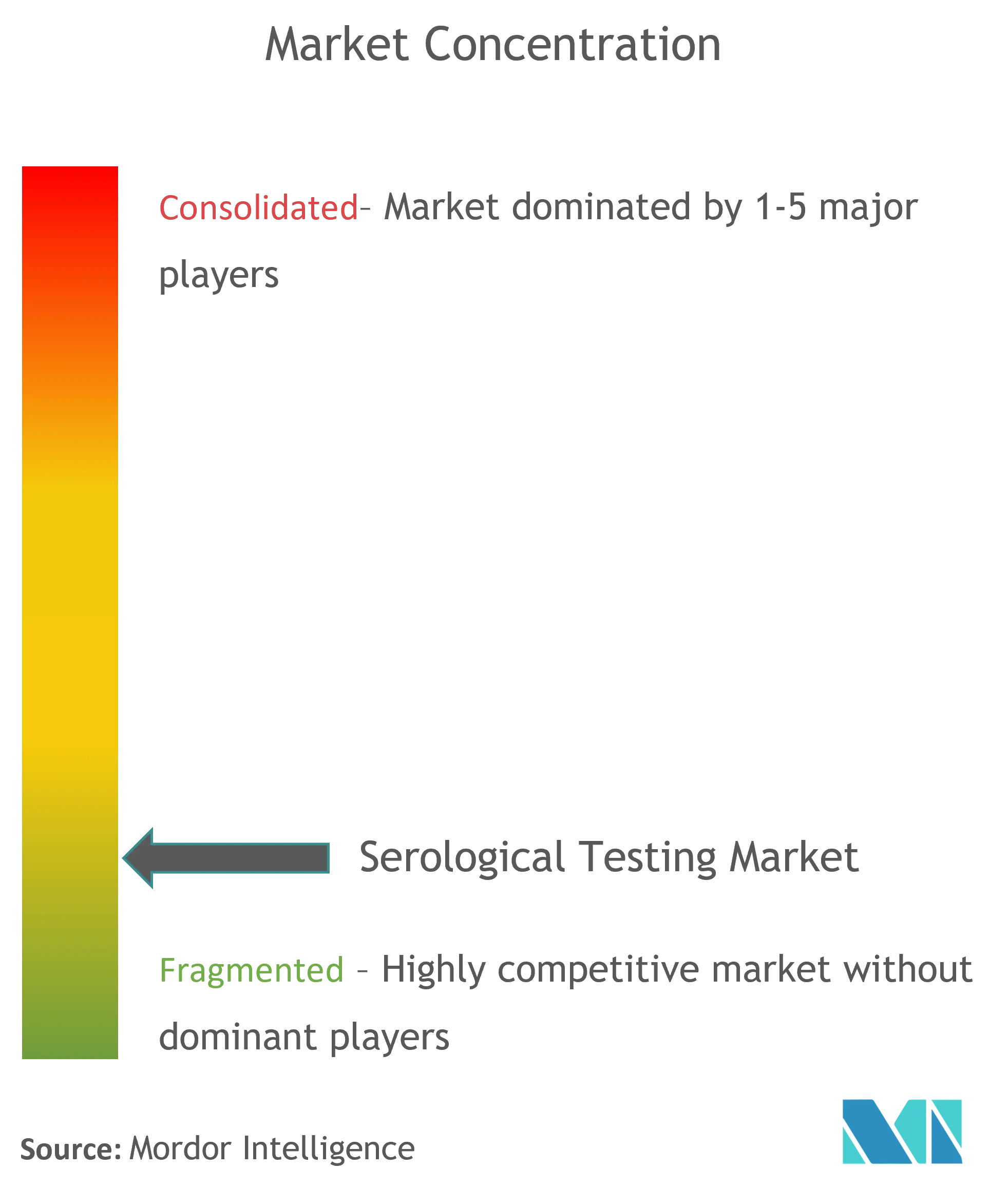
The Serological Testing market is fragmented in nature due to the presence of a several companies operating globally as well as regionally. The competitive landscape includes an analysis of a few international as well as local companies which hold the market shares and are well known. include Cellex Inc., Advanced Diagnostics, Inc., Randox Laboratories Ltd., Eurofins Scientific, Abbott, Becton, Dickinson and Company (BD), Quest Diagnostics, F. Hoffmann La-Roche Ltd, BioMedomics Inc.,Beckman Coulter, Inc. and Thermo Fisher Scientific, Inc..
Global Serological Testing Industry Leaders
F. Hoffmann La-Roche Ltd
Thermo Fisher Scientific, Inc.
Beckman Coulter, Inc.
ELITechGroup
Becton, Dickinson and Company (BD)
- *Disclaimer: Major Players sorted in no particular order
Recent Industry Developments
- In April 2020, Accelerate Diagnostics and BioCheck had a collaboration agreement to distribute MS-FAST Chemiluminescence Immunoassay Analyzer and SARS-CoV-2 Antibody Tests.
- In March 2020, Chembio Diagnostic Systems Inc. had launched the DPP COVID-19 Serological Point-of-Care Test.
Global Serological Testing Market Report Scope
As per the scope of the report, Serological testing is a type of medical testing that works on blood samples by testing the presence of antibodies against viruses. The blood of a person who has been exposed should be full of antibodies against the virus. The serological testing market is segmented by Technology (ELISA, NAT, CLIA/CMIA and Others), Product (Instruments and Consumables & Reagents), Application (Hepatitis, Coronavirus (COVID-19), Human Immunodeficiency Virus (HIV), Rotavirus infections, and Others), End-User (Diagnostic Laboratories, Hospitals and Other End Users), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, and South America). The market report also covers the estimated market sizes and trends for 17 different countries across major regions, globally. The report offers the value (in USD million) for the above segments.
| ELISA |
| NAT |
| CLIA/CMIA |
| Others |
| Instruments |
| Consumables and Reagents |
| Hepatitis |
| Coronavirus (COVID-19) |
| Human Immunodeficiency Virus (HIV) |
| Rotavirus infections |
| Others |
| Diagnostic Laboratories |
| Hospitals |
| Other End Users |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Technology | ELISA | |
| NAT | ||
| CLIA/CMIA | ||
| Others | ||
| By Product | Instruments | |
| Consumables and Reagents | ||
| By Application | Hepatitis | |
| Coronavirus (COVID-19) | ||
| Human Immunodeficiency Virus (HIV) | ||
| Rotavirus infections | ||
| Others | ||
| By End-User | Diagnostic Laboratories | |
| Hospitals | ||
| Other End Users | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||
Key Questions Answered in the Report
What is the current Global Serological Testing Market size?
The Global Serological Testing Market is projected to register a CAGR of 8.8% during the forecast period (2025-2030)
Who are the key players in Global Serological Testing Market?
F. Hoffmann La-Roche Ltd, Thermo Fisher Scientific, Inc., Beckman Coulter, Inc., ELITechGroup and Becton, Dickinson and Company (BD) are the major companies operating in the Global Serological Testing Market.
Which is the fastest growing region in Global Serological Testing Market?
Asia-Pacific is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Global Serological Testing Market?
In 2025, the North America accounts for the largest market share in Global Serological Testing Market.
What years does this Global Serological Testing Market cover?
The report covers the Global Serological Testing Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Global Serological Testing Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
Global Serological Testing Market Report
Statistics for the 2025 Global Serological Testing market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Global Serological Testing analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.




