Clinical Diagnostics Market Size and Share
Clinical Diagnostics Market Analysis by Mordor Intelligence
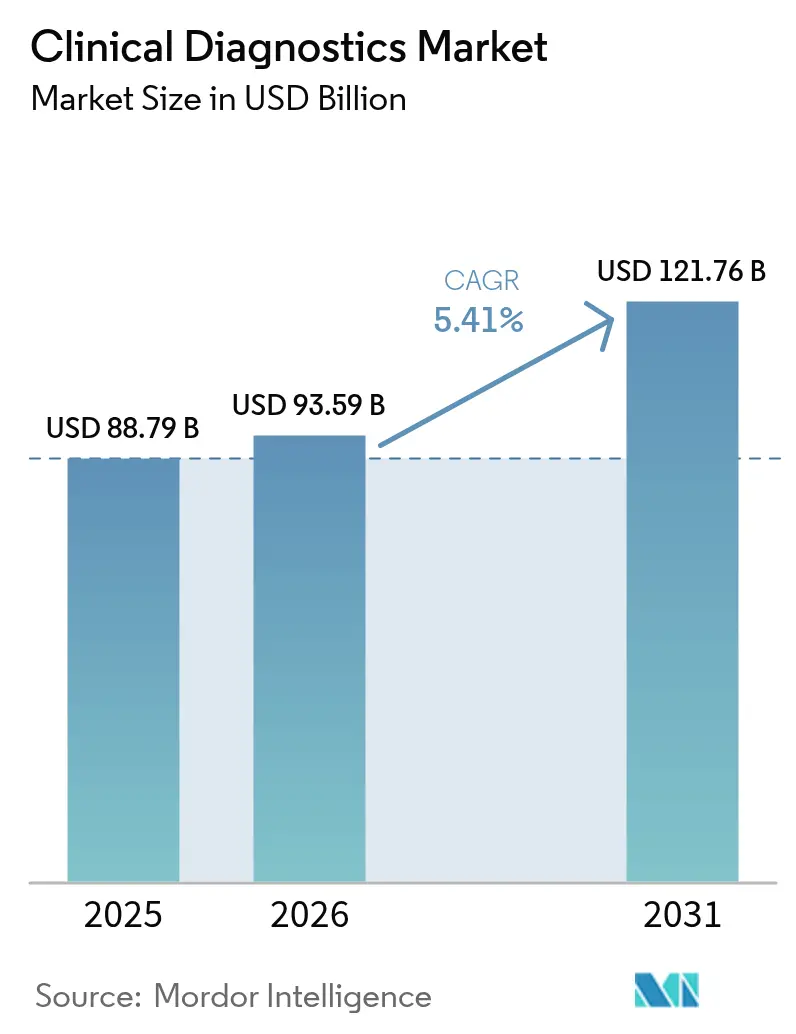
The clinical diagnostics market size is expected to grow from USD 88.79 billion in 2025 to USD 93.59 billion in 2026 and is forecast to reach USD 121.76 billion by 2031 at 5.41% CAGR over 2026-2031. This outlook signals a shift from pandemic-driven volatility to steady growth as laboratories converge automation, artificial intelligence, and precision-medicine capabilities. Heightened chronic-disease prevalence keeps routine Complete Blood Count (CBC) volumes high, yet oncology biomarker panels scale faster as health systems embrace personalized care models. Reagent pricing pressure intensifies as instrument automation trims per-test consumption, while data-management software moves from “nice-to-have” to “mission-critical” status for quality-assurance and interoperability. Emerging economies funnel infrastructure investments toward decentralized and home-based testing formats, expanding the clinical diagnostics market beyond its traditional institutional base.
Key Report Takeaways
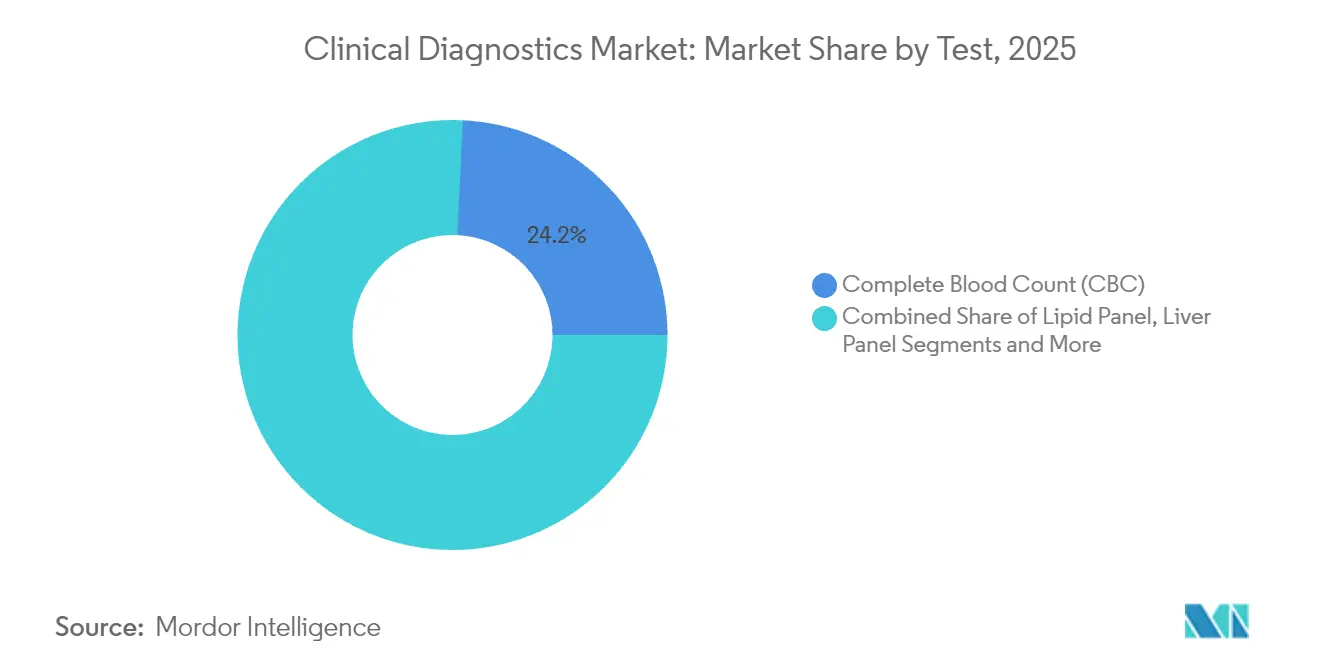
- By test, CBC held 24.24% of 2025 clinical diagnostics market share, whereas oncology & tumor marker testing is projected to grow at 10.39% CAGR through 2031.
- By product, reagents & kits led with 64.98% share in 2025; data-management software & services is set to post the highest 10.62% CAGR to 2031.
- By technology, immunoassay & immunochemistry captured 32.25% share in 2025, while molecular diagnostics will expand at a 12.05% CAGR.
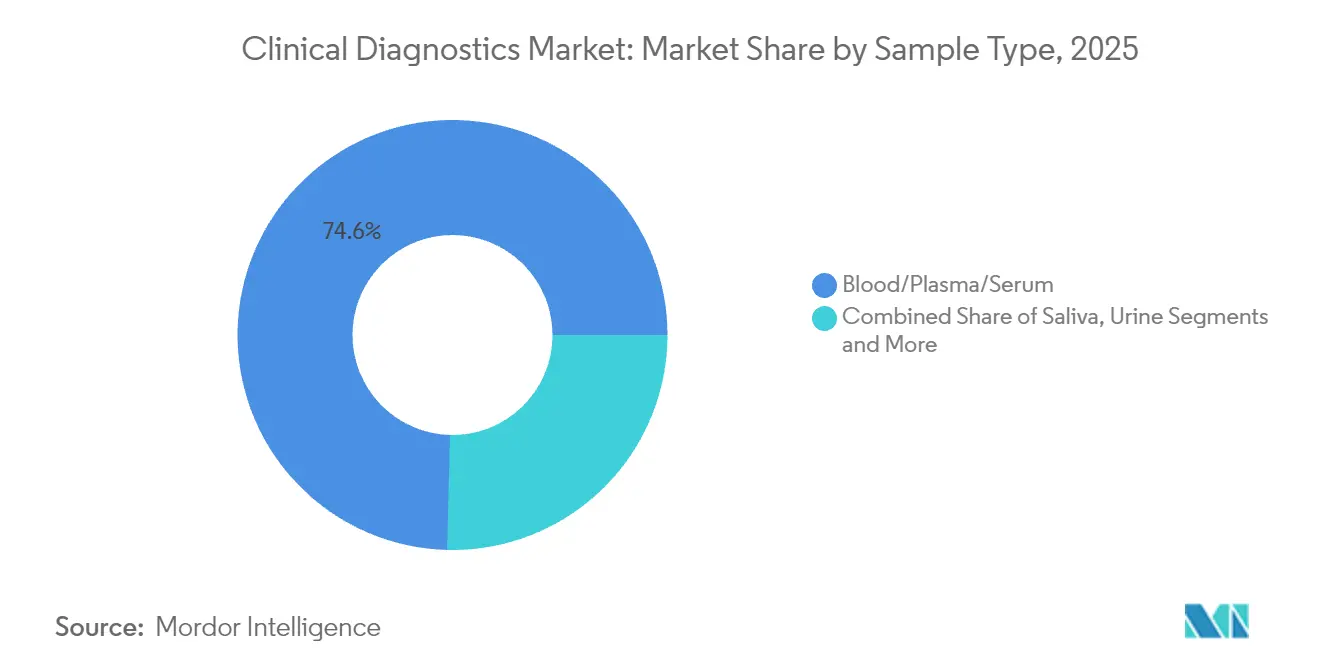
- By sample type, blood/plasma/serum accounted for 74.60% share of the 2025 clinical diagnostics market size; saliva testing is on track for a 10.32% CAGR.
- By setting, centralized clinical laboratories commanded 61.40% share in 2025, yet home-based testing is poised for the strongest 13.48% CAGR.
- By end user, hospital laboratories held 57.65% share in 2025, whereas point-of-care settings show an 11.55% CAGR outlook.
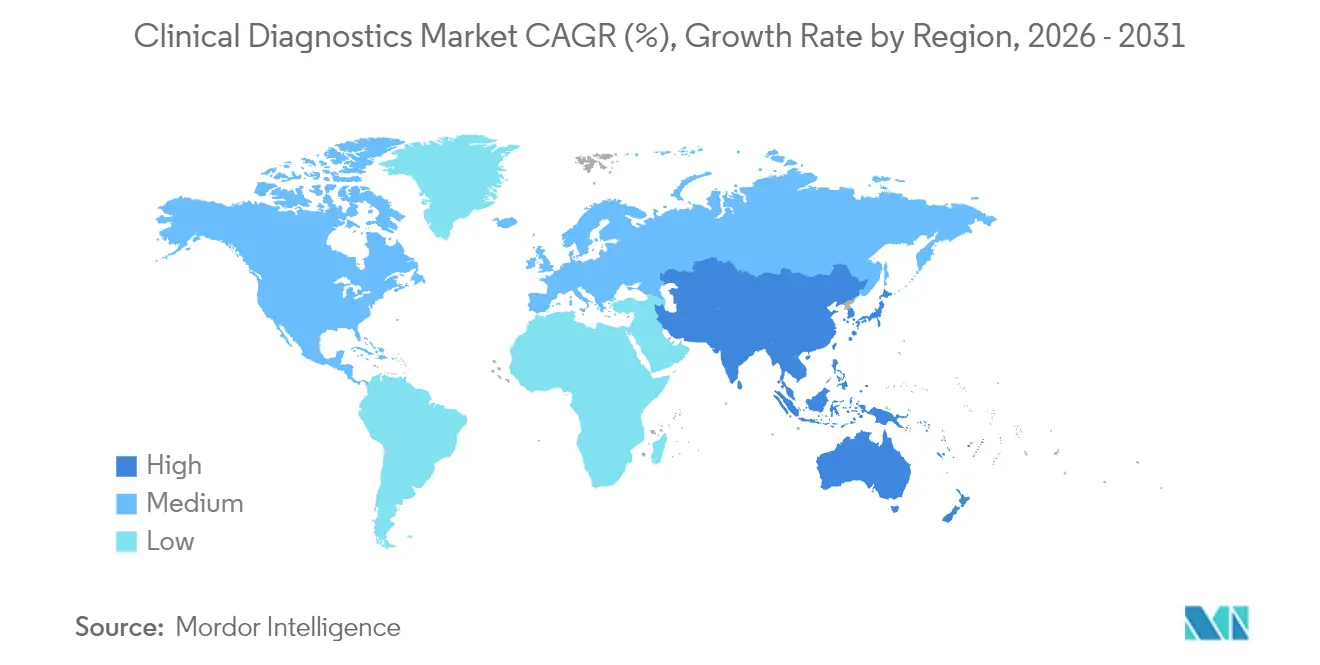
- By geography, North America dominated with 37.98% share in 2025; Asia-Pacific is projected to be the fastest-growing region at 10.14% CAGR.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Clinical Diagnostics Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising Prevalence Of Chronic & Infectious Diseases | +1.2% | Global, with highest impact in Asia-Pacific and Middle East | Long term (≥ 4 years) |
| Adoption Of High-Throughput Automated Analyzers | +0.8% | North America & Europe core, expanding to APAC | Medium term (2-4 years) |
| Expansion Of Decentralized Poc Testing In Emerging Markets | +1.1% | APAC, Latin America, and Sub-Saharan Africa | Medium term (2-4 years) |
| AI-Driven Clinical Decision Support Integration | +0.9% | Global, led by North America and Europe | Short term (≤ 2 years) |
| Multi-Omics & Precision Diagnostics Expand Test Menus | +1.0% | Global, concentrated in developed markets initially | Long term (≥ 4 years) |
| Hospital-At-Home Models Fuel Rapid Specimen-To-Answer Demand | +0.7% | North America and Europe, pilot programs in APAC | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
AI-Driven Clinical Decision Support Integration
Artificial-intelligence modules now sift millions of anonymized laboratory records to surface subtle diagnostic patterns that human review often misses. Quest Diagnostics’ alliance with Google Cloud cut complex-case error rates by nearly 30% while trimming turnaround times for critical values, prompting providers to view AI capacity as standard infrastructure rather than an add-on[1]Quest Diagnostics, “Google Cloud Collaboration Accelerates AI-Powered Diagnostics,” questdiagnostics.com. Early adopters further gain referral share as physicians gravitate to faster and more confident result-delivery pathways.
Multi-Omics & Precision Diagnostics Expand Test Menus
Guardant Health’s tumor-profiling assay illustrates how layered genomic, proteomic, and metabolomic data sharpen therapy selection and reduce repeated biopsies. Laboratories justify higher up-front costs through consolidated sampling schedules and improved adherence, supporting the shift toward value-based reimbursement where diagnostic precision demonstrably lowers downstream treatment expense.
Hospital-At-Home Models Fuel Rapid Specimen-to-Answer Demand
Ontario Health reports that acute-care-at-home programs cut inpatient cost per episode by 30% yet hinge on assays that match central-lab accuracy within 30 minutes[2]Ontario Health, “Annual Business Plan 2024/25,” ontariohealth.ca. Device makers respond with microfluidic cartridges and Bluetooth-enabled readers capable of multi-analyte panels, allowing clinicians to escalate or de-escalate care without facility transfer delays.
Expansion of Decentralized Point-of-Care Testing in Emerging Markets
The World Health Organization underscores demand for rugged, battery-powered analyzers that tolerate high ambient temperatures and intermittent electricity. Suppliers pivot toward low-margin, high-volume strategies supported by public-private partnerships, integrating mobile-health portals for result transmission across bandwidth-constrained geographies.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High Capital Cost Of Advanced Analyzers | -0.6% | Global, most pronounced in emerging markets | Long term (≥ 4 years) |
| Constrained Reimbursement & Cost-Containment Policies | -0.8% | North America and Europe primarily | Medium term (2-4 years) |
| Post-COVID Inventory Glut Slows Instrument Replacement | -0.9% | Global, concentrated in developed markets | Short term (≤ 2 years) |
| Supply-Chain Bottlenecks For Specialty Reagents | -0.5% | Global, with regional variations | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Post-COVID Inventory Glut Slows Instrument Replacement
Hospitals bought redundant molecular analyzers to cope with pandemic surges; many now run at 40-60% capacity, delaying new capital allocation. Abbott Laboratories disclosed lower 2025 diagnostics revenue as customers exhaust existing stocks instead of upgrading platforms[3]Abbott Laboratories, “Q1 2025 Earnings Commentary,” abbott.com. Price competition grows, compressing margins and elongating replacement cycles by 18-24 months at larger systems.
Constrained Reimbursement & Cost-Containment Policies
UnitedHealthcare’s 2025 prior-authorization rules and the French government’s 10% routine-test reimbursement cut exemplify mounting payer scrutiny of test value. Laboratories prioritize assays with robust outcome evidence, and manufacturers channel R&D toward health-economic studies to defend premium tests amid tightening coverage criteria.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Test: Oncology Panels Outpace Routine Screens
Oncology & tumor marker assays are forecast to post a 10.39% CAGR, reflecting pharmaceutical alignment with companion-diagnostic mandates. CBC maintained a 24.24% 2025 clinical diagnostics market share, sustaining base-volume stability in acute and chronic-care pathways.
Expanding multi-parameter oncology panels improve workflow economics by consolidating biomarkers, while lipid profiles face substitution risk from handheld devices that satisfy primary-care turnaround criteria. Infectious-disease menus normalize after pandemic highs yet remain critical in antimicrobial-resistance surveillance programs.
Note: Segment shares of all individual segments available upon report purchase
By Product: Software Becomes the Value Needle-Mover
Reagents & kits delivered 64.98% of 2025 revenue, yet data-management software is on track for a 10.62% CAGR as laboratories digitize quality control and regulatory audit trails. Instruments now ship with open APIs that allow middleware to orchestrate sample routing, reagent allocation, and result release in real time, extending asset life amid capital-spending caution. Competitive bidding squeezes reagent margins, prompting vendors to bundle informatics subscriptions that lock in customer loyalty through workflow efficiencies rather than physical consumables.
By Technology: Molecular Platforms Lead the Innovation Curve
Immunoassay & immunochemistry held 32.25% share of the 2025 clinical diagnostics market size, but molecular diagnostics is forecast to climb at a 12.05% CAGR on AI-assisted pathogen detection and next-generation sequencing cost decline. Automation of hematology cell-imaging and digital coagulation endpoints reduces manual review, while mass-spectrometry niches such as toxicology command premium pricing in reference labs.
By Sample Type: Saliva and Other Non-Invasive Specimens Gain Traction
Blood-based testing still represents 74.60% of current volume, yet saliva assays exhibit a 10.32% CAGR as home-collection kits improve compliance and pediatric acceptance. Urine and tissue biopsies retain relevance for nephrology and oncology workflows, while micro-sweat and breath condensate sampling emerge in metabolic disorder screening pilots that could broaden the clinical diagnostics market in underserved cohorts.
Note: Segment shares of all individual segments available upon report purchase
By Setting: Home Testing Redraws the Logistics Map
Centralized labs leverage economies of scale for complex panels, but home-based kits are set to grow 13.48% annually as reimbursement frameworks evolve to support remote monitoring. Point-of-care carts inside emergency departments now integrate hematology, chemistry, and molecular modules, cutting admission decisions by up to 60 minutes and redirecting lower-acuity cases to outpatient channels.
By End User: Point-of-Care Gains at the Expense of Inpatient Labs
Hospital laboratories accounted for 57.65% of 2025 revenue but face share leakage to point-of-care sites, projected to expand 11.55% per year. Independent labs differentiate through specialty test portfolios, whereas physician-office units secure patient convenience advantages in chronic-disease follow-ups.
Geography Analysis
North America retained 37.98% 2025 share on high per-capita spending, but Asia-Pacific’s 10.14% CAGR underscores widening access and rising chronic-disease incidence. Asia-Pacific is projected to add more than USD 15.62 billion in incremental revenue between 2026 and 2031, buoyed by public-hospital expansion, universal-health-coverage rollouts, and local manufacturing incentives that reduce test cost per capita. Government subsidies encourage decentralized platforms that mitigate specialist shortages in rural districts, allowing the clinical diagnostics market to tap first-time users and drive double-digit unit growth. Multinationals partner with provincial authorities to establish reagent-filling facilities aimed at circumventing import tariffs and shortening lead times.
North America, while mature, remains a technology bellwether. AI-enabled molecular panels and home-specimen logistics have moved from pilot programs to system-wide protocols at integrated-delivery networks. Yet reimbursement constraints and prior-authorization mandates temper volume growth. Laboratories respond by pairing precision oncology tests with real-world-evidence dossiers that justify value under outcome-based contracts. Consolidation persists as regional health systems outsource routine work to large reference labs that optimize scale and invest in next-generation informatics.
Europe faces divergent trajectories: northern countries channel preventive-care budgets into cardiovascular and metabolic-disease screening, whereas southern nations grapple with fiscal austerity that limits adoption of high-priced molecular assays. The European Union’s In Vitro Diagnostic Regulation (IVDR) further elevates compliance requirements, prompting smaller manufacturers to exit sub-scale product lines. Still, aging demographics assure steady baseline volume, and cross-border collaborations on rare-disease diagnostics sustain specialized test demand.
Competitive Landscape
Global competition remains moderately concentrated as the top five vendors account for a significant share of 2024 revenue. Abbott, Roche, Danaher-owned Beckman Coulter, and others defend incumbency through continual menu expansion, automation upgrades, and software bundling. Danaher’s DXC500i analyzer, cleared by the FDA in March 2025, delivers faster throughput and lower sample-carryover, reinforcing its chemistry franchise. Roche extends its cobas line with an AI-assisted viral-load algorithm that reduces invalid-run rates.
Strategic partnerships proliferate. Quest Diagnostics aligns with Google Cloud to embed machine-learning pipelines within its national laboratory information network, targeting stroke risk-prediction algorithms that could be white-labeled for hospital clients. Labcorp teams with Ultima Genomics to accelerate low-cost whole-genome sequencing, while bioMérieux acquires SpinChip Diagnostics to strengthen rapid-test capabilities at the patient bedside.
New entrants concentrate on narrow but high-growth niches such as liquid biopsy, exosome sequencing, and multi-cancer early detection. Capital flows favor platforms that marry biomarker discovery with vertically integrated informatics, minimizing dependency on incumbent analyzers. However, scaling challenges remain: reimbursement hurdles, clinical-utility evidence, and distribution in established hospital formularies slow disruptive share capture. Incumbents increasingly adopt “buy-or-ally” tactics, acquiring promising startups to fill technology gaps while maintaining brand trust and global regulatory infrastructure.
Clinical Diagnostics Industry Leaders
Bio-Rad Laboratories Inc.
Danaher Corporation (Beckman Coulter, Cepheid)
F. Hoffmann-La Roche AG
Abbott Laboratories
bioMerieux SA
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- March 2025: Danaher Corporation received FDA clearance for the DXC500i chemistry analyzer, boosting throughput and broadening its mid-volume lab portfolio.
- January 2025: bioMérieux closed the acquisition of SpinChip Diagnostics, adding microfluidic point-of-care technology to its rapid-testing lineup.
Global Clinical Diagnostics Market Report Scope
As per the scope of the report, a clinical diagnostic test is a type of medical test performed to aid in the diagnosis or detection of any disease. These tests play a vital role in the prevention, control, and surveillance of any disease. Clinical diagnostics help improve patient care, contribute to consumer safety, and reduce healthcare spending.
The clinical diagnostics market is segmented by test, product, end user, and geography. The test segment is further segmented into lipid panel, liver panel, renal panel, complete blood count, electrolyte testing, infectious disease testing, and other tests. The product segment is further divided into instruments, reagents, and other products. The end user is further bifurcated into hospital laboratory, diagnostic laboratory, point-of-care testing, and other end users. The geography is further segmented into North America, Europe, Asia-Pacific, Middle East and Africa, and South America. The market report also covers the estimated sizes and trends for 17 countries across major regions globally. The report offers values (in USD Billion) for the above segments.
| Lipid Panel |
| Liver Panel |
| Renal Panel |
| Complete Blood Count (CBC) |
| Electrolyte Testing |
| Infectious Disease Testing |
| Oncology & Tumor Marker Testing |
| Companion Diagnostics |
| Other Tests |
| Instruments/Analyzers |
| Reagents & Kits |
| Data-Management Software & Services |
| Clinical Chemistry |
| Immunoassay & Immunochemistry |
| Molecular Diagnostics |
| Hematology |
| Coagulation & Hemostasis |
| Microbiology |
| Urinalysis |
| Others (Mass-Spec, Flow Cytometry) |
| Blood/Plasma/Serum |
| Urine |
| Saliva |
| Tissue/Biopsy |
| Other Specimens |
| Centralized Clinical Laboratories |
| Point-of-Care Testing Sites |
| Home-based Testing |
| Hospital Laboratories |
| Independent Diagnostic Laboratories |
| Point-of-Care Settings |
| Physician Office Laboratories |
| Other End Users |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| South Korea | |
| Australia | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Test | Lipid Panel | |
| Liver Panel | ||
| Renal Panel | ||
| Complete Blood Count (CBC) | ||
| Electrolyte Testing | ||
| Infectious Disease Testing | ||
| Oncology & Tumor Marker Testing | ||
| Companion Diagnostics | ||
| Other Tests | ||
| By Product | Instruments/Analyzers | |
| Reagents & Kits | ||
| Data-Management Software & Services | ||
| By Technology | Clinical Chemistry | |
| Immunoassay & Immunochemistry | ||
| Molecular Diagnostics | ||
| Hematology | ||
| Coagulation & Hemostasis | ||
| Microbiology | ||
| Urinalysis | ||
| Others (Mass-Spec, Flow Cytometry) | ||
| By Sample Type | Blood/Plasma/Serum | |
| Urine | ||
| Saliva | ||
| Tissue/Biopsy | ||
| Other Specimens | ||
| By Setting | Centralized Clinical Laboratories | |
| Point-of-Care Testing Sites | ||
| Home-based Testing | ||
| By End User | Hospital Laboratories | |
| Independent Diagnostic Laboratories | ||
| Point-of-Care Settings | ||
| Physician Office Laboratories | ||
| Other End Users | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| South Korea | ||
| Australia | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
How large is the clinical diagnostics market in 2026?
The clinical diagnostics market size is USD 93.59 billion in 2026, with a projected value of USD 121.76 billion by 2031.
Which testing segment is growing the fastest?
Oncology and tumor marker assays are forecast to expand at a 10.39% CAGR through 2031.
What product category shows the highest growth rate?
Data-management software and services lead with a 10.62% CAGR as laboratories digitize workflows.
Why is Asia-Pacific viewed as the key growth region?
Government healthcare investments, rising chronic-disease incidence, and expanding middle-class access drive a 10.14% CAGR in the region.
How is AI influencing diagnostic laboratories?
AI reduces complex-case error rates, accelerates turnaround times, and now forms part of core laboratory infrastructure rather than optional add-ons.
What is the outlook for home-based diagnostic testing?
Home-based testing is expected to grow 13.48% per year as reimbursement models and remote-care programs gain traction.
Page last updated on:











