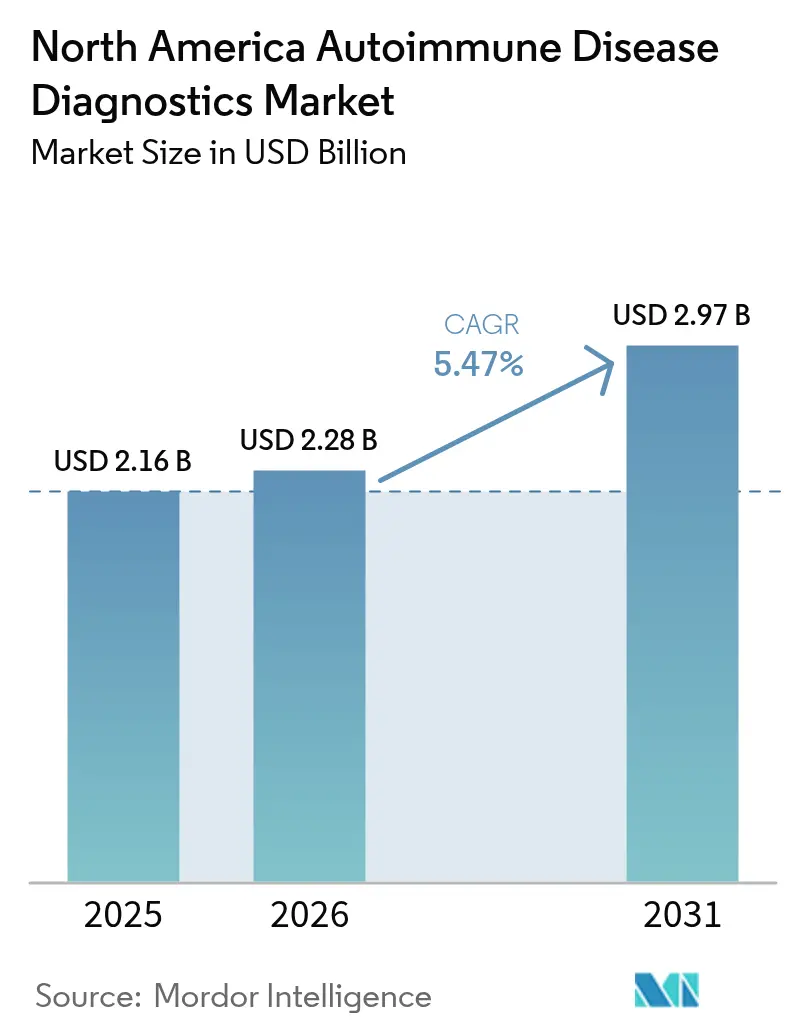
North America Autoimmune Disease Diagnostics Market Analysis by Mordor Intelligence
The North America Autoimmune Disease Diagnostics Market size was valued at USD 2.16 billion in 2025 and estimated to grow from USD 2.28 billion in 2026 to reach USD 2.97 billion by 2031, at a CAGR of 5.47% during the forecast period (2026-2031).
The rising prevalence of autoimmune diseases and growing public awareness of autoimmune disorders are the primary factors driving the market growth. For instance, study data published by the University of Oxford in May 2023 reported that about one in 10 individuals in the United Kingdom was affected by autoimmune disorders in 2022. These autoimmune conditions include 19 diseases, including rheumatoid arthritis, Type 1 diabetes, and multiple sclerosis. It also stated that 10% of the overall population was affected by these autoimmune diseases, a significant percentage. Hence, the high burden of autoimmune diseases is expected to increase the demand for diagnostics, boosting market growth during the forecast period.
Moreover, strategic activities such as product launches by major regional market players are expected to boost market growth, enabling patients to have diversified access to various products. For instance, in March 2024, AMPEL BioSolutions launched LuGENE, a pioneering blood test, in the United States. This test is specifically designed to predict flares in systemic lupus erythematosus (SLE) and provide detailed insights into the unique nuances of each patient's condition. This, in turn, facilitates swifter, evidence-driven treatment, which is expected to be in significant demand over the coming years. Hence, the new product launches will likely drive market growth during the forecast period.
Therefore, the factors above, such as the high prevalence of autoimmune disorders and the new product launches by major market players, are expected to boost market growth. However, the slow turnaround time for autoimmune disease diagnostic test results and the high frequency of false positive results are expected to impede the market growth during the forecast period.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
North America Autoimmune Disease Diagnostics Market Trends and Insights
The Immunologic Assays Segment is Expected to Hold a Significant Market Share During the Forecast Period
Immunologic reactions are particular, as they can take place only in the presence of proper immunologic agents. Compared to conventional tests, immunologic assays have been proven to provide highly accurate results even with tiny samples. Factors responsible for the segment's growth include cost-effectiveness, the rising prevalence of autoimmune diseases coupled with the increasing geriatric population, growth in the biotechnology and pharmaceutical sectors, and high sensitivity and specificity of immunologic assays.
For instance, according to an article published by BMC in December 2022, immunologic assays such as indirect immunofluorescence assay using human epithelial type-2 cells (HEp-2 IFA) have high sensitivity and represent a commonly used screening assay for antinuclear antibodies, which are associated with many autoimmune diseases such as systemic lupus erythematosus (SLE), primary Sjögren's syndrome (pSS), and systemic sclerosis (SSc). Thus, the high efficiency of immunologic assays for diagnosing autoimmune diseases is expected to boost segment growth.
Also, the rising prevalence of autoimmune diseases is the primary factor driving the segment's growth. For instance, according to an article published by BMJ Journals in October 2022, a study was conducted using the last 20 years' data till 2022, which showed that the estimated pooled prevalence of systemic lupus erythematosus (SLE) was 3.66 in Canada, 6.5 in Mexico, and 12.13 in the United States in 2022. Thus, the high prevalence of autoimmune disorders such as SLE is expected to boost the adoption of immunologic assays in the region.
Moreover, according to the data published by Crohn's and Colitis Canada and Pfizer Canada in November 2022, it was estimated that over 300,000 Canadians were living with inflammatory bowel disease in 2022. This number is expected to rise to 403,000 by 2030, equivalent to 1 in 100 people. Hence, the factors above, such as the rising prevalence of autoimmune disorders and the geriatric population, are expected to increase the demand for immunologic assays, thereby boosting segment growth during the forecast period.
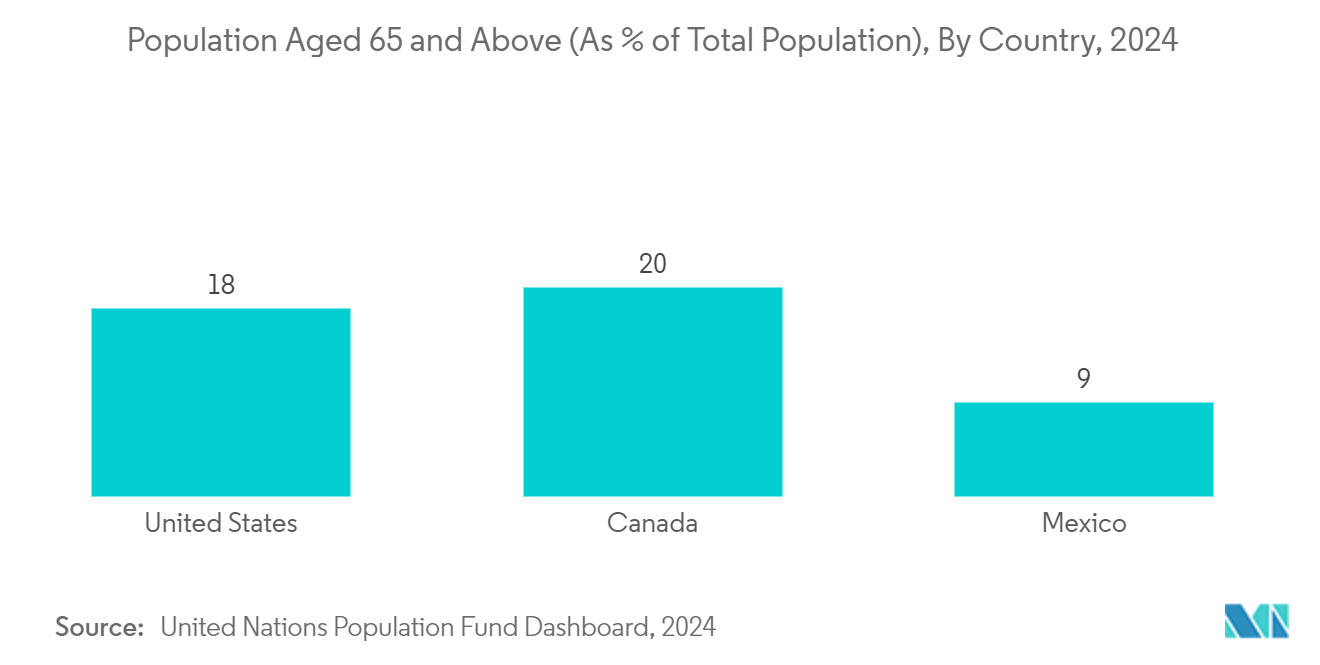
The United States is Expected to Hold a Significant Market Share During the Forecast Period
The United States is expected to hold a significant share of the North American autoimmune disease diagnostics market owing to the increasing developments by major market players in the country. For instance, in April 2022, Quanterix received a breakthrough device designation by the US FDA for the ultra-sensitive blood test that measures a biomarker of nerve damage in people with multiple sclerosis (MS).
Additionally, the high burden of autoimmune diseases likely drives the country's market growth. For instance, according to data updated by the National Institute of Environmental Health Sciences in May 2022, autoimmune diseases are estimated to affect more than 24 million people in the United States. Also, an additional 8 million people are said to have autoantibodies, blood molecules that indicate a person's chance of developing autoimmune disease. Thus, the rising prevalence of autoimmune disorders will boost the country's market growth.
Hence, the factors above, such as the rising prevalence of autoimmune disorders and the increasing research and development on autoimmune diseases, are expected to boost market growth in the United States during the forecast period.
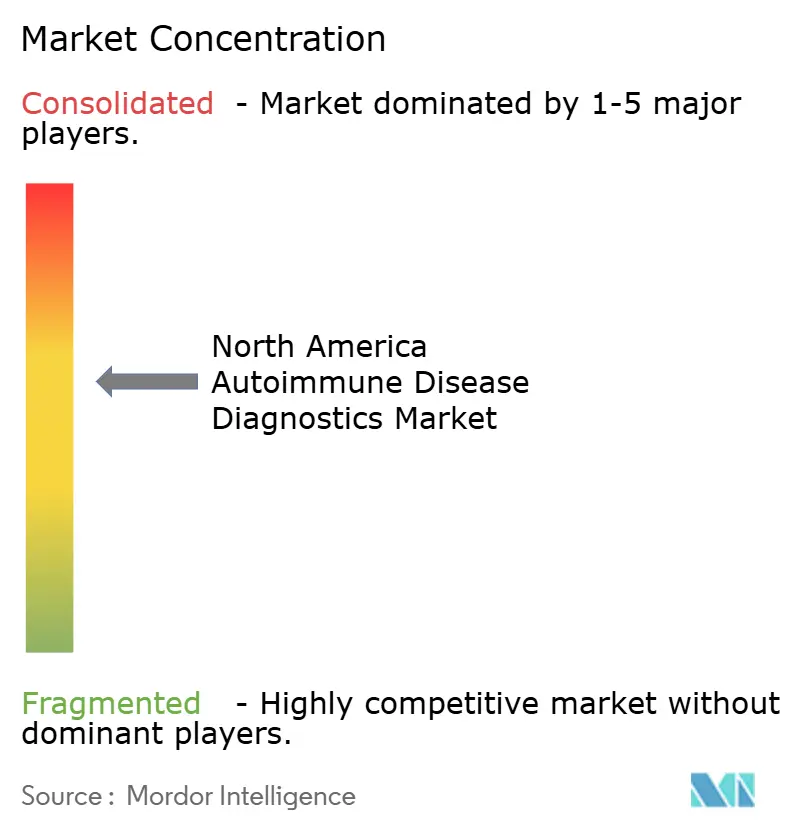
Competitive Landscape
The North American autoimmune disease diagnostics market is fragmented. It comprises several major market players focusing on R&D activities and significant growth strategies. A few diagnostic companies entered into strategic partnerships with hospitals. This trend will continue over the next few years to meet the high patient needs across different regions. Some key North American autoimmune disease diagnostics market players are Abbott Laboratories, Biomérieux, Bio-Rad Laboratories, Euroimmun AG, and Trinity Biotech.
North America Autoimmune Disease Diagnostics Industry Leaders
Abbott Laboratories
Biomérieux
Bio-Rad Laboratories Inc.
Trinity Biotech
Thermo Fisher Scientific
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- May 2024: Mayo Clinic Laboratories and Progentec Diagnostics launched systemic lupus erythematosus advanced biomarker testing services. The DX Lupus Disease Activity Index and the DX Lupus Flare Risk Index were made accessible to all clients of Mayo Clinic Laboratories.
- February 2023: Edesa Biotech received approval from Health Canada for a phase II clinical trial of its EB06 monoclonal antibody candidate to treat vitiligo, a life-altering autoimmune disease.
North America Autoimmune Disease Diagnostics Market Report Scope
As per the report's scope, autoimmunity involves the loss of normal immune homeostasis as the organism produces an abnormal response to its tissue. Autoimmune diseases generally involve the presence of self-reactive T cells, autoantibodies, and inflammation. The diagnosis can be useful in managing patients suffering from autoimmune diseases and help provide a prognosis or indicate the severity of organ involvement or damage.
The North American autoimmune disease diagnostics market is segmented by disease type, diagnostic test, and geography. The market is segmented by disease type into systemic autoimmune disease and localized autoimmune disease. The systemic autoimmune diseases include rheumatoid arthritis, psoriasis, systemic lupus erythematosus (SLE), multiple sclerosis, and other diseases. The localized autoimmune diseases include inflammatory bowel disease, Type 1 diabetes, thyroid, and other localized autoimmune diseases. By diagnosis tests, the market is segmented into regular laboratory tests, inflammatory markers, immunologic assays, antibody tests, and other tests. By geography, the market is segmented into the United States, Canada, and Mexico. The report offers the value (in USD) for the above segments.
| Systemic Autoimmune Disease | Rheumatoid Arthritis |
| Psoriasis | |
| Systemic Lupus Erythematosus (SLE) | |
| Multiple Sclerosis | |
| Other Disease Types | |
| Localized Autoimmune Disease | Inflammatory Bowel disease |
| Type 1 Diabetes | |
| Thyroid | |
| Other Localized Autoimmune Diseases |
| Regular Laboratory Tests |
| Inflammatory Markers |
| Immunologic Assays |
| Antibody Tests |
| Other Tests |
| United States |
| Canada |
| Mexico |
| By Disease Type | Systemic Autoimmune Disease | Rheumatoid Arthritis |
| Psoriasis | ||
| Systemic Lupus Erythematosus (SLE) | ||
| Multiple Sclerosis | ||
| Other Disease Types | ||
| Localized Autoimmune Disease | Inflammatory Bowel disease | |
| Type 1 Diabetes | ||
| Thyroid | ||
| Other Localized Autoimmune Diseases | ||
| By Diagnostic Test | Regular Laboratory Tests | |
| Inflammatory Markers | ||
| Immunologic Assays | ||
| Antibody Tests | ||
| Other Tests | ||
| Geography | United States | |
| Canada | ||
| Mexico | ||


Key Questions Answered in the Report
How big is the North America Autoimmune Disease Diagnostics Market?
The North America Autoimmune Disease Diagnostics Market size is expected to reach USD 2.28 billion in 2026 and grow at a CAGR of 5.47% to reach USD 2.97 billion by 2031.
What is the current North America Autoimmune Disease Diagnostics Market size?
In 2026, the North America Autoimmune Disease Diagnostics Market size is expected to reach USD 2.28 billion.
Who are the key players in North America Autoimmune Disease Diagnostics Market?
Abbott Laboratories, Biomérieux, Bio-Rad Laboratories Inc., Trinity Biotech and Thermo Fisher Scientific are the major companies operating in the North America Autoimmune Disease Diagnostics Market.
What years does this North America Autoimmune Disease Diagnostics Market cover, and what was the market size in 2025?
In 2025, the North America Autoimmune Disease Diagnostics Market size was estimated at USD 2.28 billion. The report covers the North America Autoimmune Disease Diagnostics Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the North America Autoimmune Disease Diagnostics Market size for years: 2026, 2027, 2028, 2029, 2030 and 2031.
Page last updated on:








