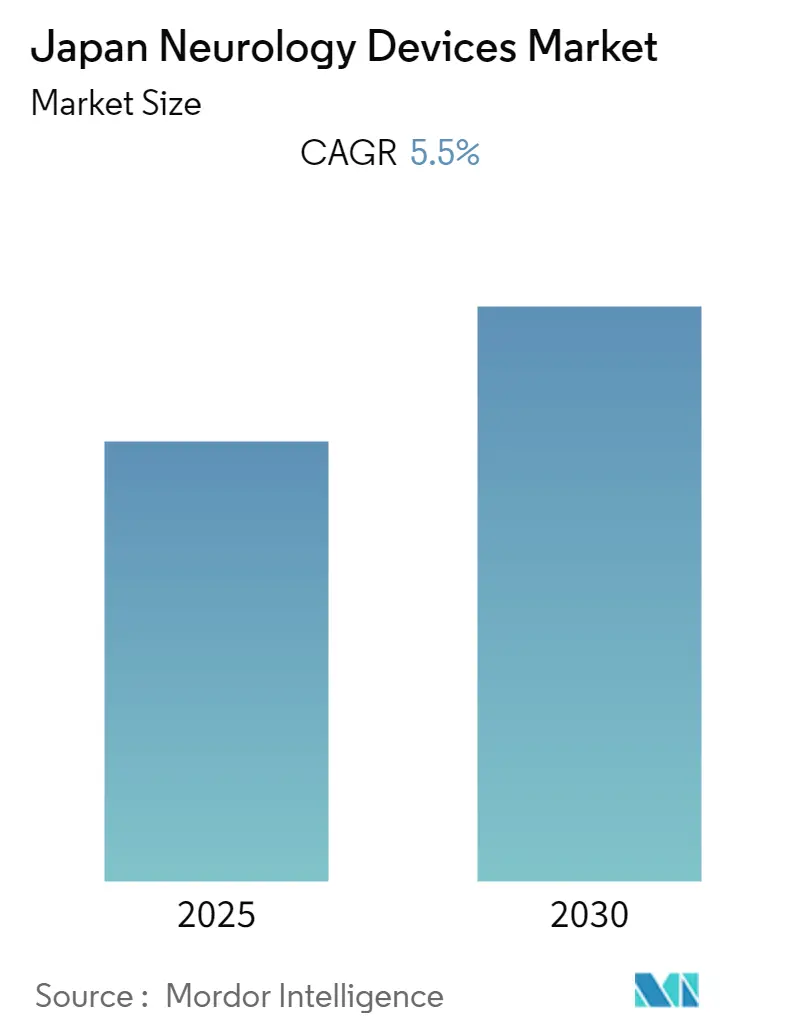
Japan Neurology Devices Market Analysis by Mordor Intelligence
The Japan Neurology Devices Market is expected to register a CAGR of 5.5% during the forecast period.
COVID-19 had a significant impact on the Japanese neurology devices market and the overall healthcare industry. According to the study published in Spine Surgery and Related Research in February 2022, the initial COVID-19 semi-lockdown in Japan resulted in fewer elective surgeries and more urgent surgeries that had affected progressive neurological deterioration for some spine patients, even in a nonepidemic region. Moreover, according to the study published in Surgery Today in November 2022, during the COVID-19 pandemic, overall, fewer surgeries were performed, and the rate of decline was about 10% to 15%. Hence, COVID-19 had a significant impact on the Japanese neurology devices market in the initial phase of the pandemic. However, currently, the market is growing at a stable pace owing to a resumption of neurological surgeries, diagnosis procedures, a resumption of manufacturing activities, and the widespread availability of products across Japan, and it is expected to witness a similar trend over the coming years.
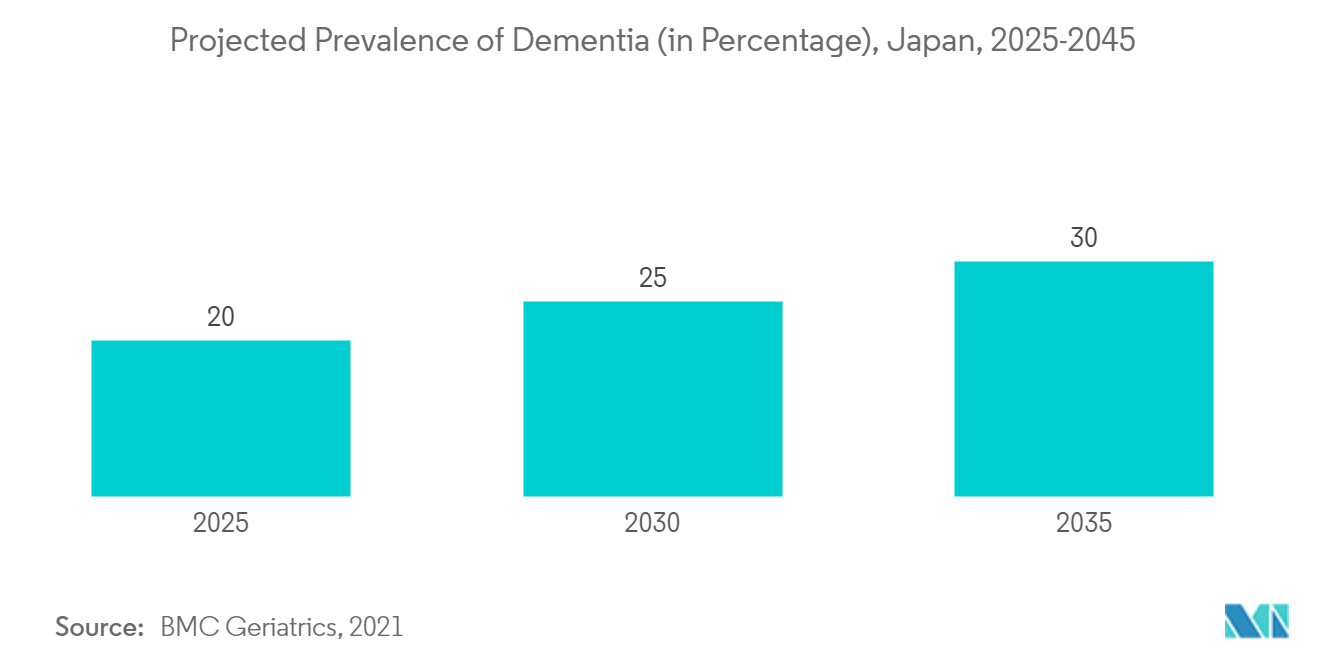
The key factors propelling the growth of this market are an increase in the incidence of neurological disorders, huge investments by private players in neurology devices, and an increase in R&D in the field of neuro therapies. For instance, according to the study published in the Journal of Alzheimer's Diseases in March 2021 in Japan, it was predicted that there will be between 6.5 million to 7 million and 8.5 million to 11.5 million people with dementia in 2025 and 2060, respectively. Likewise, in Japan, 50-75% of dementia cases are caused by Alzheimer's disease (ADD). In addition, according to the World Alzheimer Report 2022, about 43% of those with mild dementia, 67% of those with moderate dementia, and 90% of those with severe dementia were institutionalized in the country. Thus, a significant burden of neurological diseases is expected to boost demand for neurology devices, thereby boosting the market's growth.
Furthermore, increased investment by the market players in introducing advanced neurological devices is expected to boost the market over the forecast period. For instance, in January 2022, GrayMatters Health (GMH), a developer of digital self-neuromodulation therapeutics for mental disorders, announced the successful closing of its USD 10 million Series A financing round. Otsuka Medical Devices Co., Ltd. (Tokyo, Japan) was the lead investor in this round. With the funding, GrayMatters will introduce the first digital therapeutic device that directly targets and regulates specific brain biomarkers associated with mental disorders.
Thus, all the aforementioned factors, such as the significant burden of neurological diseases and investments in neurology devices, are expected to boost market growth in the near future. However, the high cost of equipment may restrain market growth over the forecast period.
Japan Neurology Devices Market Trends and Insights
Cerebrospinal Fluid Management Device is Expected to Hold Significant Share Over the Forecast Period
Cerebrospinal Fluid Management (CSF) devices play a major role in the treatment of traumatic brain injury (TBI). Shunts are devices used in the CSF procedure that ensure that any cerebrospinal fluid accumulated near the brain gets drained out. The major factors contributing to the growth of the market for cerebrospinal fluid (CSF) management devices include the increasing prevalence of neurological disorders and the increasing geriatric population.
The growing burden of neurological diseases in the country is expected to propel the growth of the market over the forecast period. For instance, according to the study published in the Journal of Movement Disorders in October 2021, the annual prevalence of Parkinson's disease (PD) in Japan is reported as 50-80 per 100,000, and it is increasing rapidly because aging is a major risk factor for PD. Thus, a significant burden of Parkinson's disease is expected to increase the demand for its treatment, thereby boosting the market for CSF devices over the forecast period.
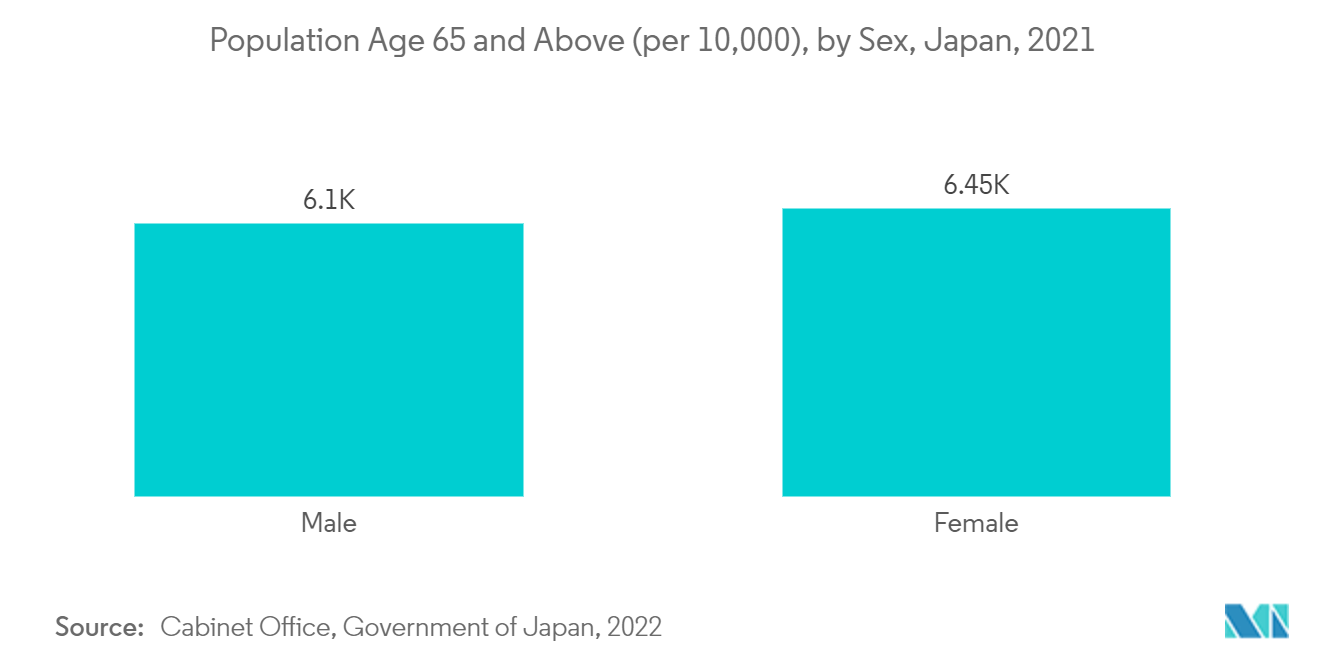
According to World Bank data updated in July 2022, Japan has the highest proportion of the elderly population in the world. In addition, as per the World Population Prospectus 2022 published by the United Nations, in 2021, Japan and a few other countries had the highest life expectancies at birth (about 85 years) and age 65 among the countries and areas with at least half a million people. Thus, a growing geriatric population that is more susceptible to neurological diseases is expected to drive the demand for CSF devices.
Thus, all the aforementioned factors, such as the growing geriatric population, are expected to boost segment growth over the forecast period.


Deep Brain Stimulation Devices is Expected to Hold Significant Share Over the Forecast Period
Deep brain stimulation (DBS), a neurosurgical procedure, is used to treat movement disorders like Parkinson's disease, essential tremor, and dystonia, as well as other conditions like obsessive-compulsive disorder (OCD) and epilepsy by implanting a medical device called a neurostimulator that sends electrical impulses to specific targets in the brain (the brain nucleus).
The segment is driven by the rising prevalence of neurological diseases and rising research activities on deep brain stimulation devices. For instance, according to the study published in Neuroengineering and Rehabilitation in November 2022, Neodymium (NdFeB) magnets, which are small and powerful, are used in transcranial static magnetic field stimulation (tSMS) to temporarily inhibit brain activity below the magnet. Due to its competitive benefits, including safety, simplicity, and low cost, it is a promising non-invasive brain stimulation technique. The tSMS system has the capacity to control brain activity and create an effective magnetic field in deep tissues. For non-invasive deep brain stimulation, it is applicable. Thus, technological advancement in the DBS is expected to propel the growth of the segment.
Thus, all the aforementioned factors are expected to boost segment growth over the forecast period.


Competitive Landscape
The Japanese neurology devices market is moderately competitive, with the presence of several regional and global players. The competitive landscape includes an analysis of a few international as well as local companies which hold market shares and are well known. The major companies include B. Braun Melsungen AG, Stryker Corporation, Boston Scientific Corporation, Medtronic PLC, Abbott Laboratories, among others.
Japan Neurology Devices Industry Leaders
B. Braun Melsungen AG
Stryker Corporation
Boston Scientific Corporation
Medtronic PLC
Abbott Laboratories
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- September 2022: Shionogi & Co., Ltd. and Pixie Dust Technologies, Inc. announced that they have entered into a business partnership agreement (hereafter 'the Partnership Agreement') to launch a business involving brain activation through sound stimulation.
- December 2020: Terumo Corporation (Japan) launched the WEB Embolization System, an intravascular aneurysm treatment device, in Japan. With this commercial launch, Terumo provides new treatment options for brain aneurysm cases that were difficult to treat in currently approved methods.


Japan Neurology Devices Market Report Scope
As per the scope of the report, neurological devices are medical devices that help to diagnose, prevent, and treat a variety of neurological disorders and conditions, such as Alzheimer's disease, Parkinson's disease, major depression, and traumatic brain injury. The Japan Neurology Devices Market is Segmented by Type of Device (Cerebrospinal Fluid Management Device, Interventional Neurology Device (Interventional/Surgical Simulators, Neurothrombectomy Devices, and Carotid Artery Stents, and Others), Neurosurgery Devices (Neuroendoscopes, Stereotactic Systems, and Other Neurosurgery Devices), Neurostimulation Device (Spinal Cord Stimulation Devices, Deep Brain Stimulation Devices, Sacral Nerve Stimulation Devices, and Other Neurostimulation Devices), and Other Type of Devices). The report offers the value (in USD million) for the above segments.
| Cerebrospinal Fluid Management Devices | |
| Interventional Neurology Devices | Interventional/Surgical Simulators |
| Neurothrombectomy Devices | |
| Carotid Artery Stents | |
| Others | |
| Neurosurgery Devices | Neuroendoscopes |
| Stereotactic Systems | |
| Other Neurosurgery Devices | |
| Neurostimulation Devices | Spinal Cord Stimulation Devices |
| Deep Brain Stimulation Devices | |
| Sacral Nerve Stimulation Devices | |
| Other Neurostimulation Devices | |
| Other Types of Devices |
| By Type of Device | Cerebrospinal Fluid Management Devices | |
| Interventional Neurology Devices | Interventional/Surgical Simulators | |
| Neurothrombectomy Devices | ||
| Carotid Artery Stents | ||
| Others | ||
| Neurosurgery Devices | Neuroendoscopes | |
| Stereotactic Systems | ||
| Other Neurosurgery Devices | ||
| Neurostimulation Devices | Spinal Cord Stimulation Devices | |
| Deep Brain Stimulation Devices | ||
| Sacral Nerve Stimulation Devices | ||
| Other Neurostimulation Devices | ||
| Other Types of Devices | ||


Key Questions Answered in the Report
What is the current Japan Neurology Devices Market size?
The Japan Neurology Devices Market is projected to register a CAGR of 5.5% during the forecast period (2025-2030)
Who are the key players in Japan Neurology Devices Market?
B. Braun Melsungen AG, Stryker Corporation, Boston Scientific Corporation, Medtronic PLC and Abbott Laboratories are the major companies operating in the Japan Neurology Devices Market.
What years does this Japan Neurology Devices Market cover?
The report covers the Japan Neurology Devices Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Japan Neurology Devices Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
Japan Neurology Devices Market Report
Statistics for the 2025 Japan Neurology Devices market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Japan Neurology Devices analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.



