Alzheimers Diagnosis And Drugs Market Size and Share
Alzheimers Diagnosis And Drugs Market Analysis by Mordor Intelligence
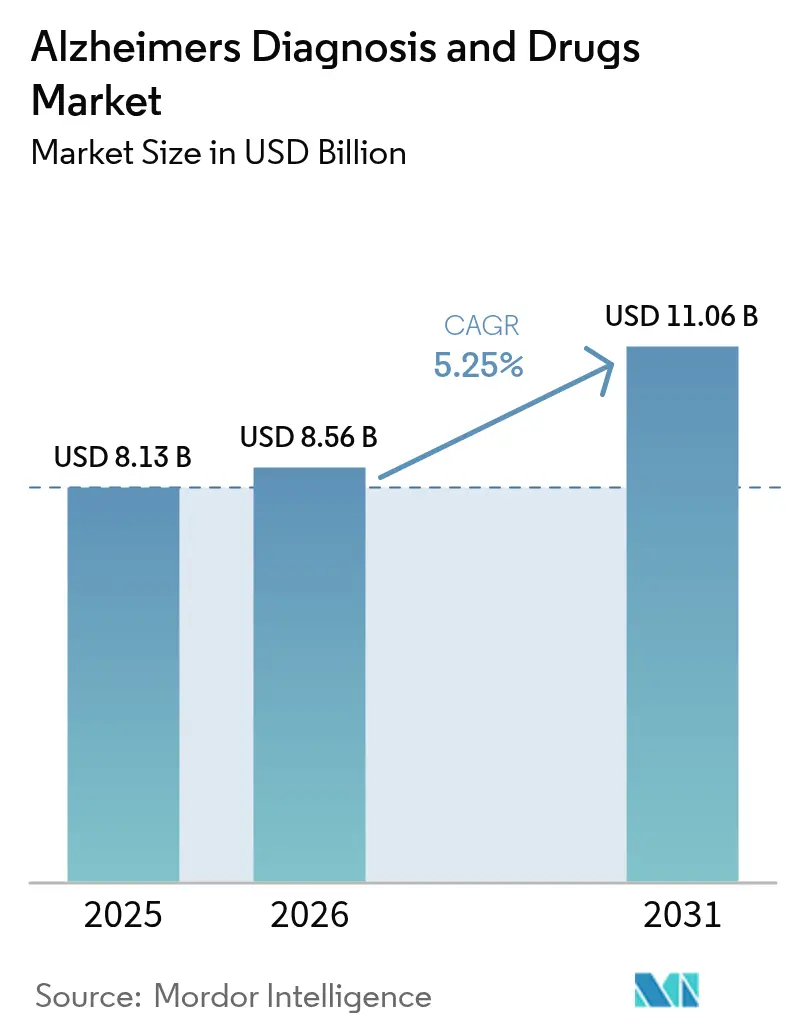
The Alzheimers Diagnosis And Drugs Market size is expected to increase from USD 8.13 billion in 2025 to USD 8.56 billion in 2026 and reach USD 11.06 billion by 2031, growing at a CAGR of 5.25% over 2026-2031.
Growth stems from the first wave of disease-modifying antibodies, widening biomarker reimbursement, and AI-enabled imaging platforms that shorten diagnostic timelines. Anti-amyloid monoclonal antibodies have re-energized investor sentiment, while blood-based tests are solving capacity bottlenecks created by limited PET scanners and cerebrospinal fluid labs. Governments in North America and parts of Europe are adding value-based payment rules that link reimbursement to real-world outcomes, a step that should reduce payer push-back on expensive biologics. Asia-Pacific health systems are spending heavily on neurology training and telehealth, positioning the region for double-digit gains. Meanwhile, venture funding is gravitating toward AI-driven diagnostic start-ups and combination-therapy programs that hedge the historically high Phase III failure rate.
Key Report Takeaways
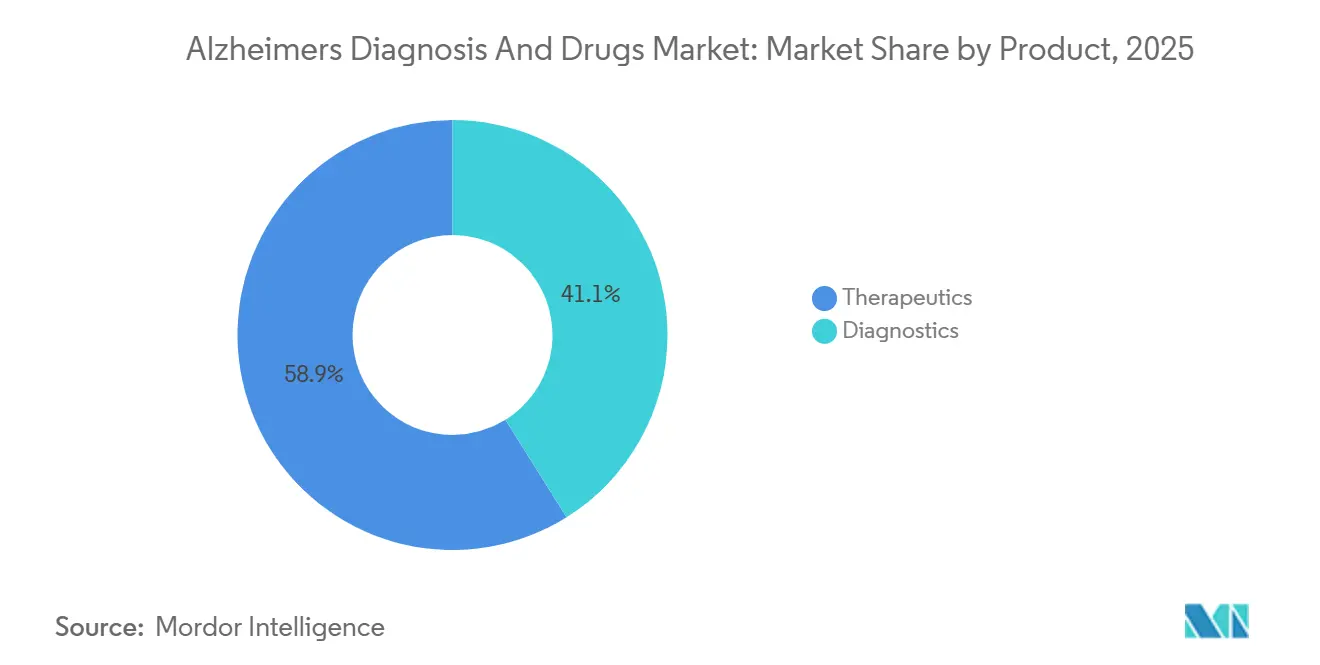
- By product category, therapeutics accounted for 58.90% of the Alzheimer's diagnosis and drugs market share in 2025, while diagnostics are advancing at a 11.95% CAGR through 2031.
- By end user, hospitals and specialty clinics controlled 54.85% of the 2025 revenue pool, but home-care and remote testing providers are expanding at a 13.75% CAGR through 2031.
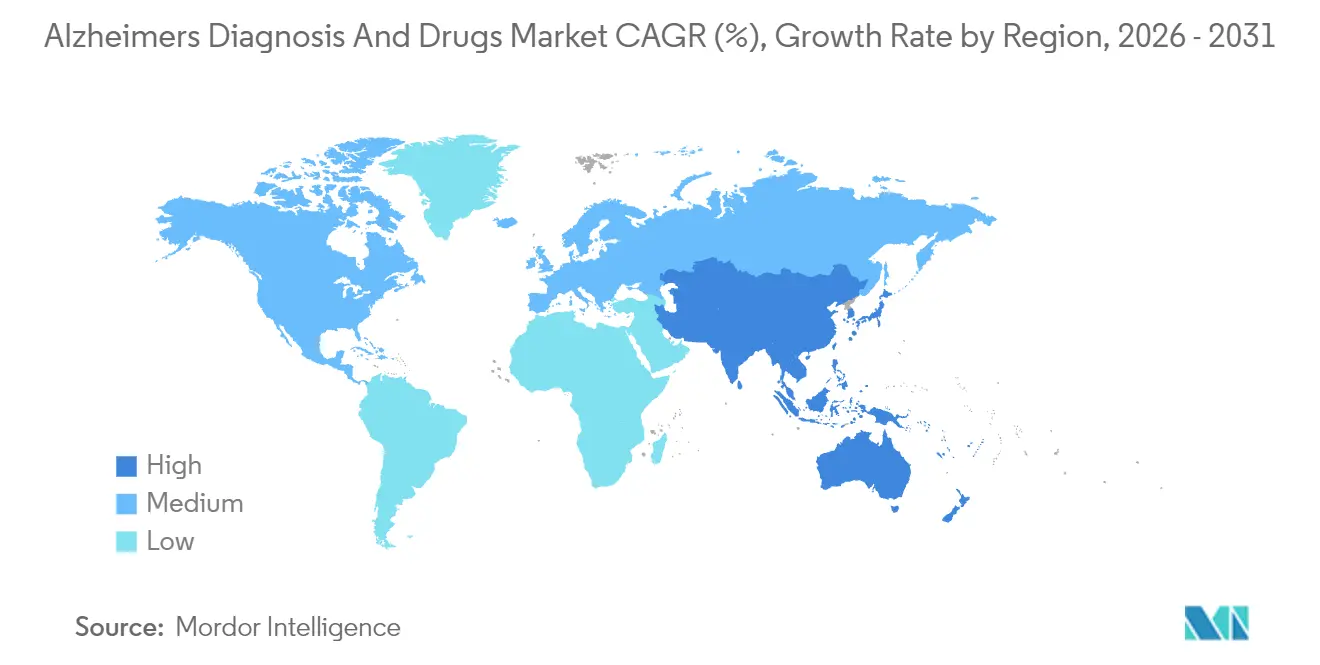
- By geography, North America dominated with 45.10% revenue share in 2025; Asia-Pacific is set to grow the fastest at 10.55% CAGR.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Alzheimers Diagnosis And Drugs Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising biomarker-based early diagnosis uptake | +1.2% | North America, EU leading | Medium term (2-4 years) |
| Accelerating approvals of anti-amyloid monoclonal antibodies | +1.8% | North America, EU primary; APAC emerging | Short term (≤2 years) |
| Growing geriatric population and disease prevalence | +1.5% | Global; APAC highest growth | Long term (≥4 years) |
| Expansion of blood-based diagnostic test reimbursement | +0.9% | North America leading | Medium term (2-4 years) |
| AI-enabled neuro-imaging workflow efficiencies | +0.7% | Developed markets | Medium term (2-4 years) |
| Regional public-private consortia for dementia R&D | +0.6% | North America, EU, select APAC | Long term (≥4 years) |
| Source: Mordor Intelligence | |||
Accelerating Approvals of Anti-Amyloid Monoclonal Antibodies
Full FDA approval of lecanemab in July 2024 and conditional EMA authorization two months later established a commercial pathway for disease-modifying therapy, prompting Medicare to relax access rules through coverage with evidence development.[1]“FDA Grants Accelerated Approval for Alzheimer’s Disease Treatment,” FDA, fda.gov Donanemab’s FDA nod in August 2024 intensified competition, forcing manufacturers to engage in value-based pricing discussions earlier in the product life cycle. Hospital networks are already expanding infusion suites, while specialty pharmacies negotiate risk-sharing agreements that tie discounts to cognitive-score maintenance. The approvals have also raised the regulatory bar, with future candidates expected to demonstrate plaque removal plus clinically meaningful slowing of decline. Asia-Pacific agencies are mirroring Western regulators, with Japan granting lecanemab priority review within 6 months of the U.S. decision, reinforcing global momentum behind disease-modifying biologics.
Rising Biomarker-Based Early Diagnosis Uptake
The FDA’s 2024 breakthrough-device designations for plasma phospho-tau assays capped a decade-long quest for minimally invasive screening, shrinking dependence on PET and lumbar puncture.[2]Heather Snyder, “Revised Diagnostic Guidelines for Alzheimer’s Disease,” Alzheimer’s Association, alz.org Updated clinical guidelines now recommend blood biomarkers as first-line tests, which has multiplied testing volumes at Quest Diagnostics and LabCorp. Primary-care physicians are adopting screening workflows that add just five minutes to routine visits, enabling earlier therapeutic intervention. Health-plan actuaries are recalculating cost-offsets, noting that each year of delay in institutional care saves USD 17,000 per patient in U.S. Medicaid outlays. Emerging economies are piloting mobile phlebotomy vans that collect samples in rural areas, broadening diagnostic reach without significant investment in bricks-and-mortar infrastructure.
Growing Geriatric Population & Disease Prevalence
The UN projects the global 65-plus cohort to reach 95 million by 2030, with APAC accounting for over half of that growth. Alzheimer’s prevalence will hit 7.1 million in the United States alone, raising the economic burden to USD 360 billion per year. Governments are embedding dementia strategies into broader healthy-aging agendas, allocating funds for memory clinic networks and specialist training slots. China’s five-year dementia program mandates biomarker capacity in every tier-2 hospital, while India is rolling out community health worker curricula that include cognitive screening modules. Venture investors view these demographics as a structural tailwind, justifying larger Series B rounds for platform biotech and digital therapeutics aimed at early-stage disease.
Expansion of Blood-Based Diagnostic Test Reimbursement
Medicare assigned national coverage for amyloid and tau blood tests in January 2024, triggering commercial payer adoption by Aetna, Humana, and Blue Cross plans by mid-2024. Germany’s G-BA granted provisional coverage at EUR 320 per test in September 2024 and demanded 24-month real-world evidence submissions. NICE draft guidance in October 2024 limited biomarker use to patients with CDR scores of 0.5-1.0 pending asymptomatic validation. This staggered adoption pattern positions diagnostics suppliers to capture revenue sooner in the United States while lobbying European agencies for broader inclusion, creating regional tailwinds for the Alzheimer's diagnosis and drugs market.
Restraint Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Late-stage drug failure rates and sunk R&D costs | -0.8% | Global | Long term (≥4 years) |
| Limited specialist workforce for therapy monitoring | -0.6% | Global; acute in rural markets | Medium term (2-4 years) |
| Diagnostic biomarker performance variability across ethnicities | -0.4% | Diverse populations | Medium term (2-4 years) |
| Payer hesitancy on high-cost biologics | -0.9% | North America, EU | Short term (≤2 years) |
| Source: Mordor Intelligence | |||
Late-Stage Drug Failure Rates & Sunk R&D Costs
Phase III attrition above 90% continues to discourage big-ticket bets, underscored by Roche’s 2024 exit from gantenerumab after USD 2 billion in spend. Investors price higher risk, upping demanded equity stakes and milestone contingencies. Mid-cap biotech is turning to platform approaches, aiming to recycle failed assets into combination regimens rather than scrapping them outright. Policy makers fear innovation droughts and are experimenting with tax credits that trigger only on successful proofs of concept, thereby sharing downside risk. Academic consortia are lobbying for dedicated NIH lines that cover “bridge-to-pivot” studies, extending the life of promising molecules that miss single-endpoint trials.
Limited Specialist Workforce for Disease-Modifying Therapy Monitoring
At USD 26,500 per year, lecanemab forces payers to reckon with stretched neurology budgets; NICE withheld full UK coverage pending real-world data, reflecting a broader European skepticism.[3]“Lecanemab Health Technology Assessment,” NICE, nice.org.uk Private U.S. insurers require amyloid positivity plus mild cognitive impairment staging, trimming the eligible pool by an estimated 30%. Coverage with Evidence Development compels physicians to upload cognitive-outcome metrics, creating administrative drag that deters smaller clinics. Some payers are trialing subscription models modeled on hepatitis-C contracts, but uptake remains limited. Delayed reimbursement cycles strain specialty pharmacies' cash flow, nudging them to demand higher wholesaler discounts or abandon the category. Patient advocacy groups fear a two-tier system in which only large urban centers provide disease-modifying biologics.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Product: Diagnostics Outpace Therapeutics on Reimbursement Momentum
Diagnostics revenue is forecast to compound 11.95% yearly through 2031, more than double the Alzheimer's diagnosis and drugs market CAGR, as blood biomarker tests supplant imaging and CSF taps. Declining CSF volumes down 2.1% per year reflect patient preference and faster blood-test turnarounds. AI-driven MRI post-processing further reduces dependence on costly PET scans.
Therapeutics retained 58.90% of 2025 sales, driven by generic cholinesterase inhibitors, which accounted for USD 2.8 billion. Anti-tau agents lag: TauRx’s hydromethylthionine missed its phase 3 endpoint in September 2024, delaying launch to 2027. Roche’s semorinemab entered phase 3 in January 2024, with results expected in 2027, signaling a second wave of innovation beyond amyloid clearance. The Alzheimer's disease diagnosis and therapeutics market is projected to grow modestly at 5.25% per year, as diagnostics capture a rising share of new spending.
By End User: Home-Care Providers Gain as Point-of-Care Tests Proliferate
Hospitals and specialty clinics generated 54.85% of 2025 revenue by controlling infusion suites and MRI scanners. Their dominance will erode as subcutaneous dosing and portable blood-test kits move treatment to lower-cost sites.
Home-care and remote testing providers are forecast to expand 13.75% annually through 2031, the fastest pace among end users. Eisai’s at-home lecanemab autoinjector, filed for FDA priority review in January 2025, exemplifies the shift. Rural patients gain access without traveling to memory centers, broadening geographic penetration and diversifying revenue streams inside the Alzheimer's diagnosis and drugs market.
Geography Analysis
North America retained 45.10% revenue in 2025, propelled by early access to FDA-cleared biologics and CMS reimbursement for blood biomarkers. Canadian provinces align benefits, though Quebec negotiates independent price caps that shave average antibody prices by 12%. Mexico leverages medical tourism, drawing Latin American patients for PET scans and tapping cross-border insurance partnerships that package lodging with diagnostic bundles. Canada’s provincial formularies added lecanemab during 2024, contingent on amyloid confirmation and APOE genotyping. Sustained payer backing underpins a steady 5.8% regional CAGR through 2031.
Asia-Pacific is the fastest-growing bloc, charting a 10.55% CAGR. China’s dementia plan mandates that amyloid-PET and blood-biomarker testing be available at every prefecture-level hospital by 2028. The Alzheimer's disease and drug market share in APAC could reach 28.60% by 2031 as Japan accelerates reimbursement for AI imaging and South Korea rolls out nationwide cognitive-screening programs at community clinics. Australia’s expedited-review pathway shaves six months off regulatory timelines, making the country a beachhead for Western firms entering Asia. India is piloting public-private elder-care hubs that combine day care, telehealth, and diagnostics under a single roof, financed by municipal bonds.
Europe offers a mature yet fragmented landscape. Germany’s sickness funds cover blood-biomarker tests ahead of most EU peers, but France still ties reimbursement to PET confirmation, slowing routine uptake. The Horizon Europe program injects USD 350 million into dementia consortia, widening the R&D pool for mid-sized biotech. Eastern European members lag in therapeutic adoption due to constrained specialty-care budgets, though they gain from EU structural funds that upgrade imaging infrastructure.
Competitive Landscape
Competition intensified once lecanemab and donanemab demonstrated that plaque clearance yields clinical benefit. Biogen and Eisai co-promote Leqembi, leveraging Biogen’s U.S. neurology sales force and Eisai’s global manufacturing. Eli Lilly, facing capacity bottlenecks, signed a contract-manufacturing pact with Samsung Biologics to double antibody output by 2026. Roche redirects resources to a tau-targeted small-molecule after its antibody setback, while Novartis invests in gene-editing spin-outs exploring APOE4 knock-down strategies. C2N Diagnostics and Quanterix engage in cross-licensing of antibody clones to broaden assay menus and secure hospital-lab stickiness.
Digital entrants complicate the field. Google’s DeepMind teams up with the University of Oxford to develop multimodal AI that integrates speech pattern analytics with MRI, inching toward a software-as-medical-device submission for 2026. Apple embeds cognitive-assessment modules into watchOS, signaling a future in which consumer electronics feed clinical decision support. Patents around phospho-tau epitopes form a dense thicket; USPTO filings related to Alzheimer’s biomarkers jumped 28% in 2024. Mid-tier players hedge risk through option-based partnerships: Alector grants AbbVie regional marketing rights for a TREM2 agonist contingent on reaching Phase II endpoints. Overall, intellectual-property strength, AI enablement, and manufacturing scalability form the three pillars of durable advantage.
Alzheimers Diagnosis And Drugs Industry Leaders
AstraZeneca PLC
Eli Lilly and Company
F. Hoffmann-La Roche AG
Johnson &Johnson Services, Inc.
Bristol-Myers Squibb Company
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- January 2026: The FDA accepted Eisai’s priority-review filing for an autoinjector formulation of lecanemab, expanding at-home dosing options.
- July 2025: The FDA approved a titration-dose label update for donanemab, halving ARIA-E incidence while preserving amyloid clearance.
- May 2025: Sanofi agreed to acquire Vigil Neuroscience for its oral TREM2 agonist VG-3927, reinforcing its early-stage neurodegeneration pipeline.
- May 2025: The FDA cleared the Lumipulse G plasma p-tau217/β-amyloid 1-42 ratio test, the first blood assay authorized to aid Alzheimer’s diagnosis.
Global Alzheimers Diagnosis And Drugs Market Report Scope
As per the scope of the report, Alzheimer's disease is a progressive and neurodegenerative disorder that attacks the brain's nerve cells or neurons, resulting in loss of memory, thinking, and language skills, and behavioral changes. There is a rapid growth in the number of people living with Alzheimer's disease, and only around one in four people with the disease get diagnosed.
The alzheimers diagnosis and drugs market is segmented by product, which is sub-segmented as therapeutics -cholinesterase inhibitors, NMDA receptor antagonists, and other therapeutics, and diagnostics - brain imaging, CSF test, blood-based biomarkers test, and genetic testing. By end user, the market is segmented into hospitals & specialty clinics, diagnostic laboratories, research & academic institutes, and home care / remote testing providers. By geography, the market is segmented into North America, Europe, Asia-Pacific, the Middle East, Africa, and South America. The report also covers estimated market sizes and trends for 17 countries across the major global regions. The report provides the value (in USD) for all the above segments.
| Therapeutics | Cholinesterase Inhibitors |
| NMDA Receptor Antagonists | |
| Anti-amyloid mAbs | |
| Anti-tau & other DMTs | |
| Diagnostics | Brain Imaging |
| CSF Biomarker Tests | |
| Blood-based Biomarker Tests | |
| Genetic Testing |
| Hospitals & Specialty Clinics |
| Diagnostic Laboratories |
| Research & Academic Institutes |
| Home-care / Remote Testing Providers |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product | Therapeutics | Cholinesterase Inhibitors |
| NMDA Receptor Antagonists | ||
| Anti-amyloid mAbs | ||
| Anti-tau & other DMTs | ||
| Diagnostics | Brain Imaging | |
| CSF Biomarker Tests | ||
| Blood-based Biomarker Tests | ||
| Genetic Testing | ||
| By End User | Hospitals & Specialty Clinics | |
| Diagnostic Laboratories | ||
| Research & Academic Institutes | ||
| Home-care / Remote Testing Providers | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the forecast revenue for the Alzheimers diagnosis and drugs market by 2031?
It is projected to reach USD 11.06 billion, reflecting a 5.25% CAGR over 2026-2031.
Which product class is growing the fastest?
Diagnostics, especially blood-based biomarker assays, are expanding at a 11.95% CAGR through 2031.
Which region will post the highest growth rate?
Asia-Pacific is on track for a 10.55% CAGR, driven by aging demographics and increased healthcare spending.
What share did hospitals and specialty clinics hold in 2025?
They captured 54.85% of global revenue thanks to infusion and imaging infrastructure.
Which companies dominate disease-modifying therapy?
Biogen, Eisai, and Eli Lilly lead with approved or near-approved anti-amyloid antibodies.
Page last updated on:









