Genome Editing Market Size and Share
Genome Editing Market Analysis by Mordor Intelligence
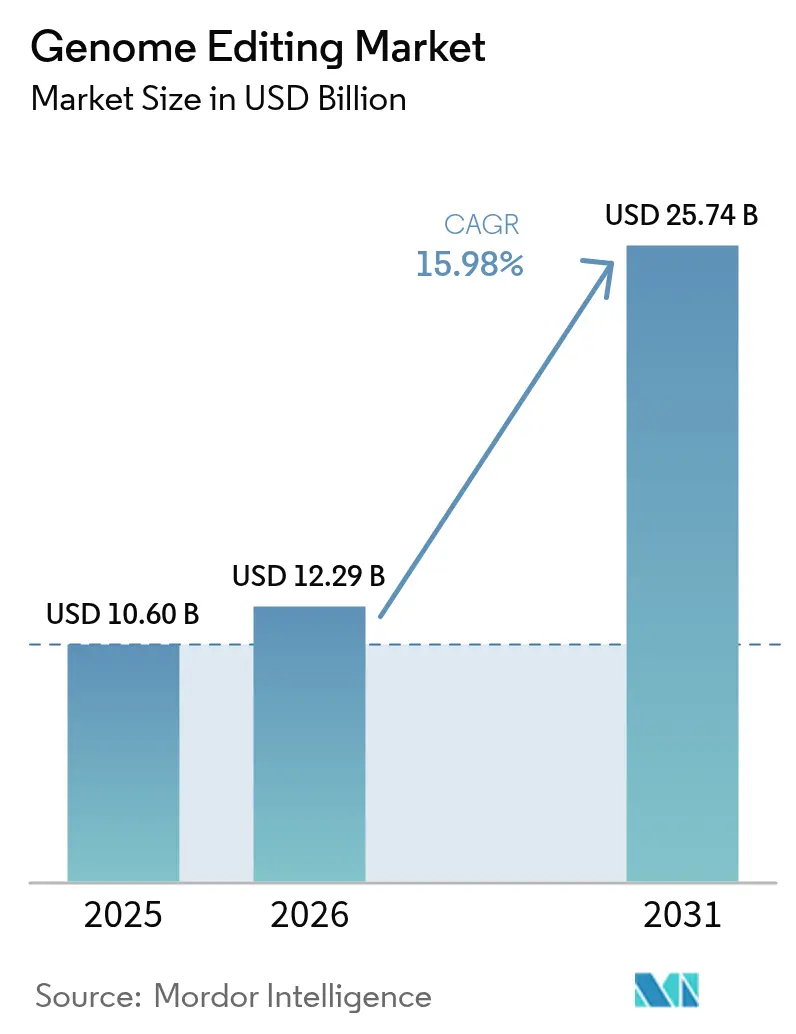
The Genome Editing Market size was valued at USD 10.60 billion in 2025 and estimated to grow from USD 12.29 billion in 2026 to reach USD 25.74 billion by 2031, at a CAGR of 15.98% during the forecast period (2026-2031).
Clinical validation of CRISPR-Cas9, rising demand for climate-resilient crops, and abundant venture capital all converge to accelerate commercial adoption. Growing regulatory confidence is evident in the wave of review designations granted to CRISPR therapeutics, while agricultural regulators in several countries now treat many gene-edited plants as conventionally bred. Competitive strategies focus on expanding GMP-compliant capacity, integrating AI into nuclease design, and signing platform licensing deals that lock in intellectual-property advantages. Intensifying collaboration between large pharmaceutical companies and agile start-ups broadens the therapeutic pipeline and speeds time-to-market, even as manufacturing scale-up, trade policy misalignment, and skilled-labor shortages temper the outlook.
Key Report Takeaways
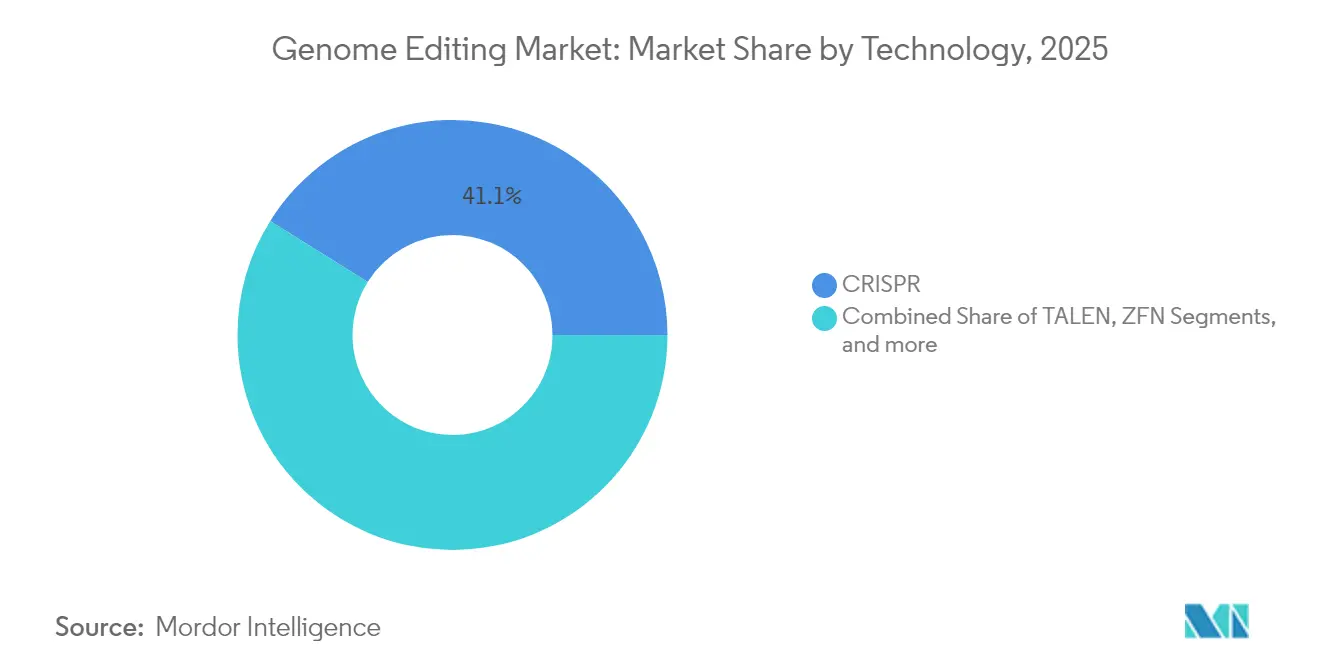
- By technology, CRISPR platforms led with 41.10% of genome editing market share in 2025; TALEN is projected to advance at a 19.49% CAGR to 2031.
- By delivery method, viral vectors accounted for 46.10% of revenue in 2025, while non-viral physical delivery is forecast to expand at 16.21% CAGR through 2031.
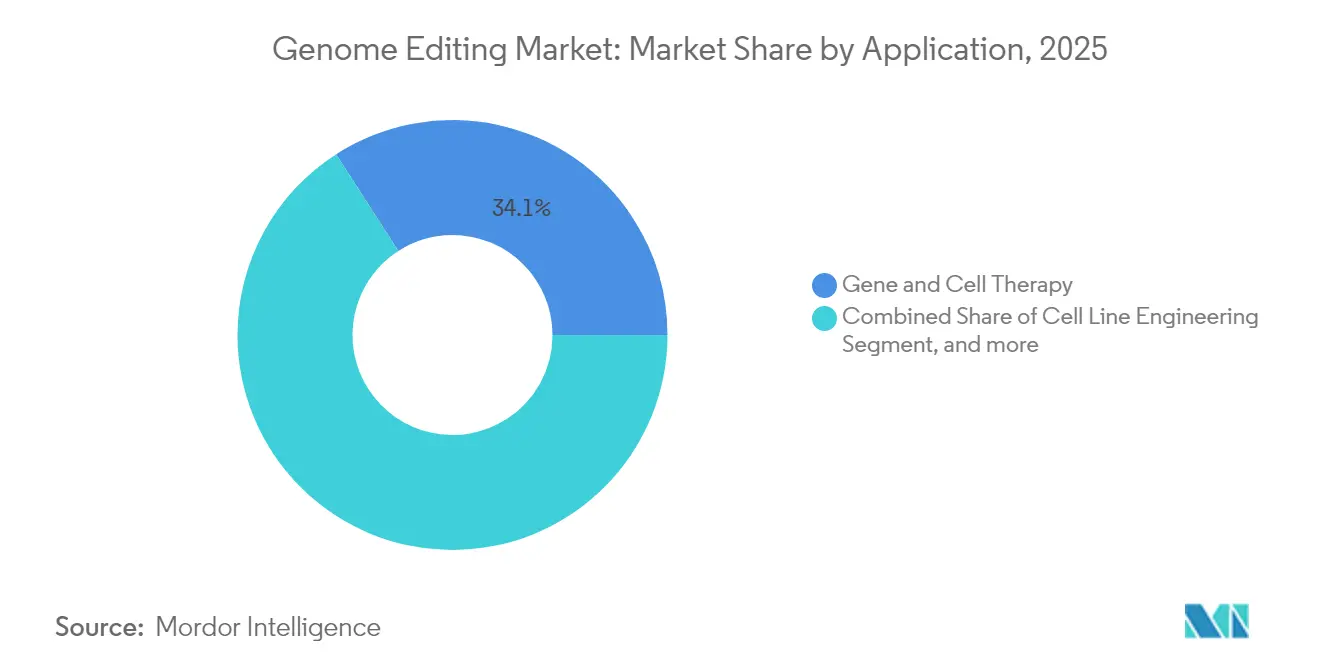
- By application, gene and cell therapy held 34.10% of the genome editing market size in 2025, whereas diagnostics and synthetic biology post the fastest 16.78% CAGR from 2026-2031.
- By end user, pharmaceutical and biotechnology companies captured a 51.85% share of the genome editing market in 2025 and are growing at a 16.65% CAGR to 2031.
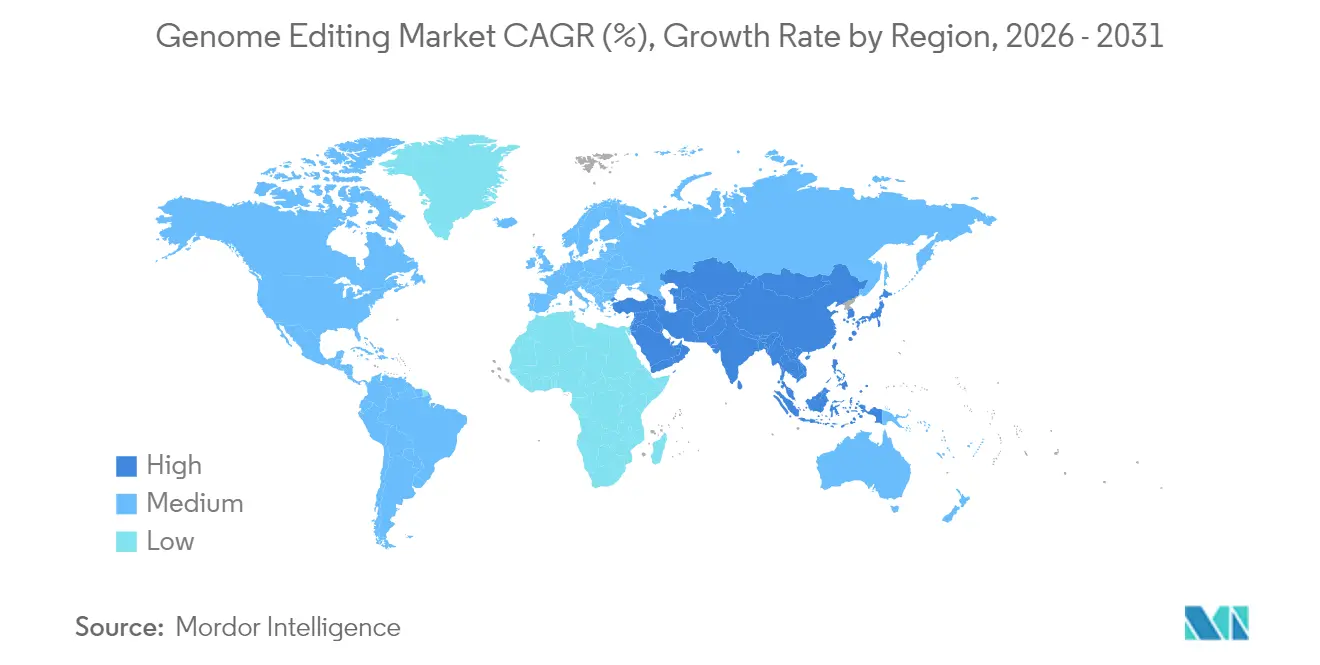
- By region, North America retained 40.80% revenue share in 2025; Asia-Pacific is set to grow at a 19.75% CAGR over the same period.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Genome Editing Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rapid Adoption of CRISPR-Cas Systems in Clinical Trials | +3.2% | Global, with North America & EU leading | Medium term (2-4 years) |
| Expanding Agricultural Biotechnology Demand for Climate-Resilient Crops | +2.8% | Global, with APAC & Latin America core | Long term (≥ 4 years) |
| Surging VC & IPO Funding for Gene-Editing Start-Ups | +2.1% | North America & EU primary, APAC emerging | Short term (≤ 2 years) |
| Mainstreaming In-Vivo Gene-Editing Therapeutics for Rare Diseases | +2.5% | Global, with regulatory advantages in US | Medium term (2-4 years) |
| Automation & AI-Assisted High-Throughput Screening Platforms | +1.8% | Global, concentrated in biotech hubs | Medium term (2-4 years) |
| Government Bio-Economy Roadmaps in Emerging Countries | +1.4% | APAC, Latin America, MEA | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Rapid Adoption of CRISPR-Cas Systems in Clinical Trials
FDA approval of the first CRISPR therapy unlocked broader clinical exploration, enabling multi-arm umbrella trials that cut development cycles by up to two years.[1]US Food and Drug Administration, “FDA Approves First CRISPR-Based Therapy,” fda.gov Positive read-outs from hematology, ophthalmology, and solid-tumor studies underscore platform versatility and attract sustained capital inflows.[2]Alexis Rappaport, “CRISPR Applications Expand Beyond Monogenic Disorders,” nature.com Universities and biotech firms are scaling CRISPR-TIL protocols that achieved complete responses in gastrointestinal cancers, moving beyond monogenic indications.
Expanding Agricultural Biotechnology Demand for Climate-Resilient Crops
Genome-edited rice, drought-tolerant corn, and heat-resistant livestock exemplify solutions that mitigate climate shocks while maintaining yield.[3]US Department of Agriculture, “Biotechnology Risk Assessment Research Grants,” usda.gov Regulatory exemptions in 16 jurisdictions where no foreign DNA persists accelerate commercialization, allowing small breeders to enter the genome editing market without onerous GMO hurdles. Public–private programs channel CRISPR tools toward nutritional fortification in staple crops, broadening market opportunities in developing regions.
Surging VC & IPO Funding for Gene-Editing Start-Ups
Despite a cyclical dip in 2024, high-quality platforms still close nine-figure rounds and craft alliances that blend big-pharma capital with nimble R&D. Strategic investments funnel resources into ultracompact CRISPR enzymes, AI-guided design suites, and programmable integrases, reinforcing technology pipelines.
Mainstreaming In-Vivo Gene-Editing Therapeutics for Rare Diseases
Single-dose in vivo programs deliver durable protein knockdown, with lipid nanoparticle systems achieving organ-specific editing beyond the liver. FDA guidance prioritizes expedited review for rare-disease candidates, prompting sponsors to stack filing queues with in-vivo products slated for launch later in the decade.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Uncertain Long-Term Off-Target Safety Profile in Humans | -2.3% | Global, with stricter EU oversight | Long term (≥ 4 years) |
| High CAPEX For GMP-Compliant Gene-Editing Manufacturing Suites | -1.9% | Global, acute in emerging markets | Medium term (2-4 years) |
| Cross-Border Regulatory Fragmentation for Edited Seeds | -1.6% | Global trade, EU-US divergence critical | Medium term (2-4 years) |
| Talent Shortage in Advanced Molecular Biology Skillsets | -1.2% | Global, concentrated in biotech hubs | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Uncertain Long-Term Off-Target Safety Profile in Humans
Ultra-deep sequencing continues to reveal hundreds of potential off-target sites, prompting regulators to demand expanded biodistribution and durability studies before late-stage trials can proceed. High-fidelity Cas variants and prime-editing approaches lessen risk, but limited long-term human data keep post-marketing surveillance requirements stringent, especially in the EU.
High CAPEX For GMP-Compliant Gene-Editing Manufacturing Suites
Green-field plants require USD 200-500 million and specialist clean-room designs. Although podular construction reduces overrun risk, capital intensity limits entry by smaller firms and fuels consolidation around CDMOs with existing footprints. Automation lowers per-batch cost yet demands skilled operators in short supply, extending time-to-capacity ramp-up.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Technology: CRISPR Leads While TALEN Accelerates
CRISPR delivered 41.10% genome editing market share in 2025, but TALEN exhibits a 19.49% CAGR that is reshaping the competitive mix. Evolving AI tools dramatically cut guide-RNA optimization cycles and expand the nuclease design space, spawning derivatives such as Open-CRISPR with novel sequence homology. Base- and prime-editing modalities gain traction as safety-oriented alternatives and may carve dedicated submarkets, especially in indication areas sensitive to double-strand break risks.
At the same time, zinc finger nucleases preserve niches in ultra-specific applications where intellectual-property freedom outweighs throughput. Meganucleases and oligonucleotide-directed mutagenesis round out the toolkit for industrial synthetic-biology workflows. Consequently, platform providers increasingly bundle editing modalities with predictive AI software and screening libraries, anchoring broader solution sales across therapeutic, agricultural, and industrial customers.

By Delivery Method: Viral Vectors Dominate, Non-Viral Innovation Surges
Viral vectors generated 46.10% of 2025 revenue through predictable transduction efficiency and a well-understood regulatory path, ensuring continued demand in high-value ex-vivo therapies. Yet non-viral physical approaches will expand at 16.21% CAGR as electroporation, microfluidic squeezing, and tissue-selective lipid nanoparticles solve the immunogenicity and payload-size limits inherent to viral systems.
Robotic microinjection platforms quadruple embryo-editing throughput, spurring agricultural breeders to adopt non-viral protocols at scale. Ribonucleoprotein complexes deliver transient editing that fades before adaptive immunity triggers, appealing to chronic-disease programs requiring repeat dosing. Chemical carriers remain relevant for screening and research but cede ground in clinical settings to more efficient physical tools.
By Application: Therapeutics Still Command Yet Diagnostics Accelerate
Gene and cell therapy maintained 34.10% of 2025 revenue and anchor most near-term commercial launches. Diagnostics, however, will post a 16.78% CAGR as CRISPR-Cas13-based RNA detection platforms enter infectious-disease and oncology screening markets. Agricultural crop engineering continues to expand as climate policy encourages resilient varieties, though regulatory fragmentation slows cross-border trade. High-throughput drug discovery and functional-genomics screens draw steady capital investment, leveraging automated CRISPR libraries to elucidate disease mechanisms.
Technological convergence with single-cell multi-omics refines disease-pathway mapping, allowing therapeutics developers to stratify patients more accurately and boost clinical-trial success rates. Synthetic-biology initiatives use genome editing to elevate lipid, enzyme, or nutraceutical yields, underpinning next-generation bio-manufacturing ecosystems.

By End User: Pharma Leadership Coupled with Academic Pioneering
Pharmaceutical and biotechnology enterprises held 51.85% of the 2025 demand and are forecast to expand at a 16.65% CAGR as precision-medicine pipelines multiply. The genome editing industry benefits from academic institutes that seed fundamental discoveries and spin out fresh ventures. CROs capture workflow outsourcing from firms that lack internal editing expertise, while plant-breeding companies scale up to meet climate-resilience targets.
Commercial rollout of treatment centers for approved CRISPR therapies illustrates the logistical lift required to translate innovation into patient access. Big-pharma partnerships inject capital and global reach into emerging platforms, exemplified by nine-figure deals targeting cardiovascular and metabolic disorders. Workforce development programs attempt to close the molecular-biology talent gap, yet tight labor markets persist in biotech hubs.
Geography Analysis
North America captured 40.80% of 2025 revenue, anchored by deep venture funding, leading clinical-trial infrastructure, and supportive FDA policies. Federal bio-economy initiatives allocate multi-billion-dollar budgets to genomics R&D and advanced manufacturing, reinforcing domestic supply chains. Nonetheless, construction-cost inflation and scarcity of GMP personnel place upward pressure on operating expenses for new facilities.
Europe combines world-class academic research with fragmented regulations that slow agricultural commercialization. The EU’s GMO designation for most gene-edited crops conflicts with more permissive stances in the United Kingdom and Switzerland, prompting calls for policy harmonization. Meanwhile, biotech investment funds and Horizon Europe grants channel resources into therapeutic programs, sustaining R&D despite trade-policy uncertainty.
Asia-Pacific is the fastest-growing region at 19.75% CAGR, propelled by Chinese industrial-policy incentives, Japanese regulatory streamlining for genome-edited foods, and Australia’s RNA Blueprint aiming for an USD 8 billion contribution to GDP. India’s risk-tiered guidelines create an expedited path for low-risk edits, catalyzing start-up formation. Technology-transfer limits and patent-pool negotiations will shape the trajectory of foreign market entrants.
Competitive Landscape
The competitive field is moderately fragmented. Thermo Fisher Scientific leverages its Life Sciences Solutions unit, which accounts for a significant share of corporate revenue, to supply reagents and instruments across research, diagnostics, and bioprocessing segments. Merck KGaA’s Sigma-Aldrich complements similar tool portfolios and maintains strong customer lock-in through consumable subscriptions.
Platform innovators are reshaping market boundaries. CRISPR Therapeutics transitioned to revenue generation following CASGEVY’s launch and now advances cardiovascular and oncology assets while holding USD 1.86 billion in cash. Intellia Therapeutics prioritizes in vivo editing and expects its cash runway to extend beyond 2027 after a 27% workforce realignment. Partnering activity intensifies, typified by Regeneron’s alliance with Mammoth Biosciences, which combines ultracompact nucleases with antibody-targeting. Patent portfolios remain decisive; access to foundational CRISPR IP offers incumbents defensive moats that smaller entrants must navigate via cross-licensing or novel enzyme discovery.
Midsized CDMOs diversify into cell-and-gene therapy suites, but high CAPEX and variable utilization have already sparked consolidation. Select service providers experiment with continuous-manufacturing lines that could halve per-dose costs once regulatory acceptance solidifies
Genome Editing Industry Leaders
GenScript USA Inc.
Integrated DNA Technologies Inc.
New England Biolabs Inc.
Sangamo Biosciences Inc.
Thermo Fisher Scientific Inc.
GenScript Biotech Corporation
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- May 2025: CRISPR Therapeutics reported Phase 1 success for CTX310, achieving 82% triglyceride and 81% LDL reduction; over 65 CASGEVY treatment centers are now active.
- May 2025: Intellia Therapeutics presented two-year data for nexiguran ziclumeran, showing sustained 90% TTR knockdown and neuropathy improvement.
- May 2025: University of Minnesota investigators achieved a complete response in a gastrointestinal-cancer patient using CRISPR-modified TIL therapy.
- April 2025: Regeneron and Mammoth Biosciences launched an in-vivo CRISPR collaboration focused on multi-tissue delivery platforms.
- January 2025: Intellia Therapeutics restructured to concentrate on NTLA-2002 and nexiguran ziclumeran, reducing headcount by 27% and extending cash runway to 2027.
Research Methodology Framework and Report Scope
Market Definitions and Key Coverage
Our study defines the genome editing market as revenue generated from reagents, instruments, services, and delivery vectors that enable targeted addition, deletion, or correction of DNA or RNA in living organisms for life-science, medical, agricultural, and industrial uses.
Scope exclusion: We exclude traditional sequencing consumables and stand-alone gene-synthesis orders that are not tied to editing workflows.
Segmentation Overview
- By Technology
- Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)
- Transcription Activator-Like Effector Nuclease (TALEN)
- Zinc Finger Nuclease (ZFN)
- Meganucleases
- Oligonucleotide-directed Mutagenesis (ODM)
- Other Technologies
- By Delivery Method
- Viral Vectors
- Non-viral Physical Methods
- Non-viral Chemical Methods
- By Application
- Cell Line Engineering
- Gene & Cell Therapy
- Drug Discovery & Functional Genomics
- Agricultural Crop Engineering
- Diagnostics & Synthetic Biology
- By End User
- Pharmaceutical & Biotechnology Companies
- Academic & Government Research Institutes
- Contract Research Organizations
- Agriculture & Food Companies
- By Geography
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific
- China
- Japan
- India
- Australia
- South Korea
- Rest of Asia-Pacific
- Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
- South America
- Brazil
- Argentina
- Rest of South America
- North America
Detailed Research Methodology and Data Validation
Primary Research
Our team spoke with tool vendors, therapy sponsors, core-facility directors, and regulators across three continents, and these dialogues anchored adoption curves, average selling prices, and clinical attrition rates.
Desk Research
We reviewed NIH trial registries, FDA biologics releases, EUROSTAT biotechnology data, USDA genome-edited crop adoption surveys, ClinicalTrials.gov statistics, and WIPO patent filings. Public company 10-Ks, investor decks, and news in Dow Jones Factiva supplied pricing and capacity signals, while D&B Hoovers provided revenue splits. The named sources illustrate our wider literature set.
Market-Sizing & Forecasting
We construct a top-down model beginning with global R&D and clinical spending, genome-edited acreage, and GMP vector output. We then cross-check it with sampled supplier invoices and channel checks. Influential variables include active CRISPR trials, vector dose per patient, seed royalty per acre, venture-capital deal value, regulatory green-lights, and kit ASP shifts. Forecasts draw on multivariate regression with scenario analysis, and missing bottom-up cells are bridged with conservative proxies.
Data Validation & Update Cycle
We triangulate outputs against import-export codes, patent citation velocity, and quarterly earnings, and any outliers trigger senior analyst re-work before sign-off. Report baselines refresh yearly with interim updates after material events.
Why Our Genome Editing Baseline Stands Up to Scrutiny
According to Mordor Intelligence, published figures vary because publishers choose different revenue buckets, geographies, and success probabilities. We retain a broad yet auditable scope and refresh currency and price decks quarterly.
Differences often arise when others count only product sales, apply static price curves, or assume every Phase II trial succeeds, whereas we blend tools, services, and probability-weighted therapy revenue.
Benchmark comparison
| Market Size | Anonymized source | Primary gap driver |
|---|---|---|
| USD 10.60 B (2025) | Mordor Intelligence | |
| USD 4.66 B (2024) | Global Consultancy A | Kits focus and limited Asia data |
| USD 10.80 B (2025) | Trade Journal B | Aggressive pipeline success, single scenario |
| USD 9.40 B (2024) | Industry Portal C | Omits services and uses static currency |
Our comparison shows that Mordor's disciplined scope choices, dual-path validation, and frequent updates create a dependable baseline for strategic decisions.


Key Questions Answered in the Report
What is the current value of the genome editing market?
The genome editing market is valued at USD 12.29 billion in 2026.
How fast will the genome editing market grow through 2031?
Revenue is projected to rise at a 15.98% CAGR, reaching USD 25.74 billion by 2031.
Which technology holds the largest genome editing market share?
CRISPR platforms led with 41.10% share in 2025, ahead of other nuclease systems.
Why is Asia-Pacific the fastest-growing regional market?
Supportive policies in China, Japan, Australia, and India foster rapid adoption, producing a forecast 19.75% CAGR to 2031.
What is the greatest risk facing commercial expansion?
Cross-border regulatory fragmentation, particularly for gene-edited crops, remains the primary drag on global market access.
Which companies are setting the pace for innovation?
CRISPR Therapeutics, Intellia Therapeutics, Mammoth Biosciences, Thermo Fisher Scientific, and Merck KGaA are among the leaders, each advancing distinctive platforms or integrated solutions.
Page last updated on:









