Pyrogen Testing Market Size and Share
Pyrogen Testing Market Analysis by Mordor Intelligence
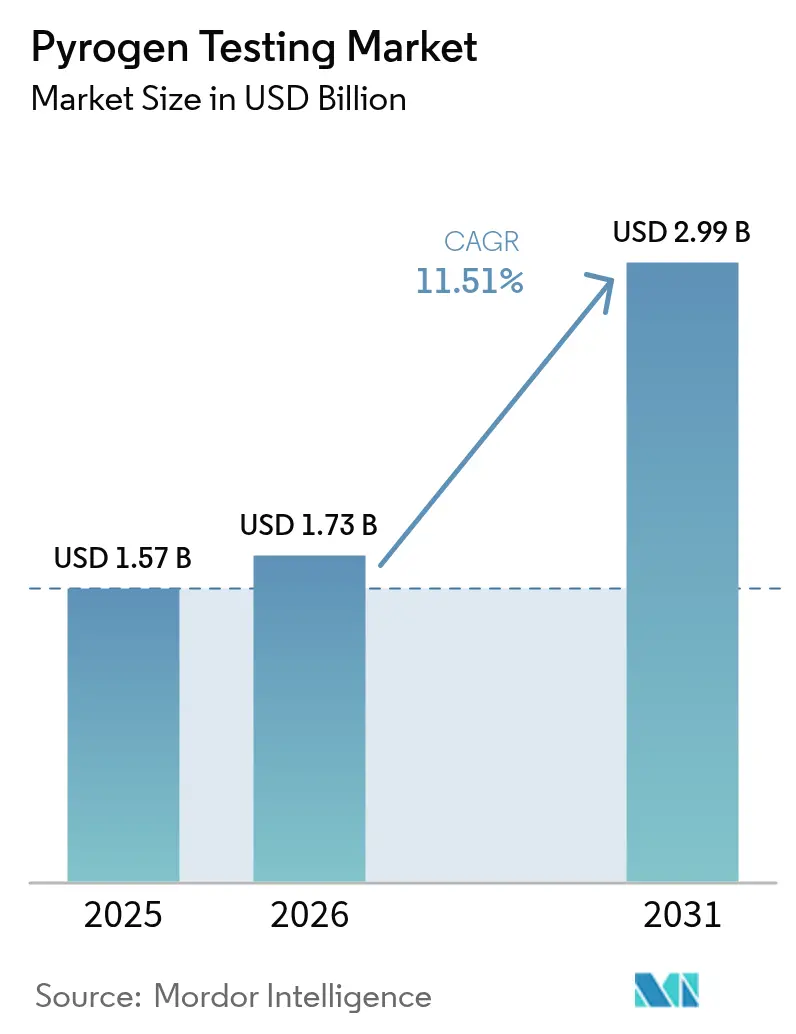
The Pyrogen Testing Market size is expected to grow from USD 1.57 billion in 2025 to USD 1.73 billion in 2026 and is forecast to reach USD 2.99 billion by 2031 at 11.51% CAGR over 2026-2031.
Growth is anchored in simultaneous United States Pharmacopeia and European Pharmacopoeia revisions that legitimize recombinant Factor C (rFC) and the Monocyte Activation Test (MAT), while eliminating the rabbit-pyrogen method in Europe. Cell-based and recombinant platforms are gaining momentum as biopharma firms pursue animal-free, supply-secure options, and global approvals of injectable biologics continue to rise. Automation-ready microfluidic analyzers are shortening turnaround times from 24 hours to less than four, supporting high-throughput lot release. Regional diversification of contract development and manufacturing organizations (CDMOs) further amplifies demand as outsourcing spreads across North America, Europe, India, and China. Headline risks include episodic horseshoe-crab lysate shortages, low endotoxin recovery (LER) events in surfactant-rich formulations, and the high validation cost SMEs face when converting to MAT or rFC platforms.
Key Report Takeaways
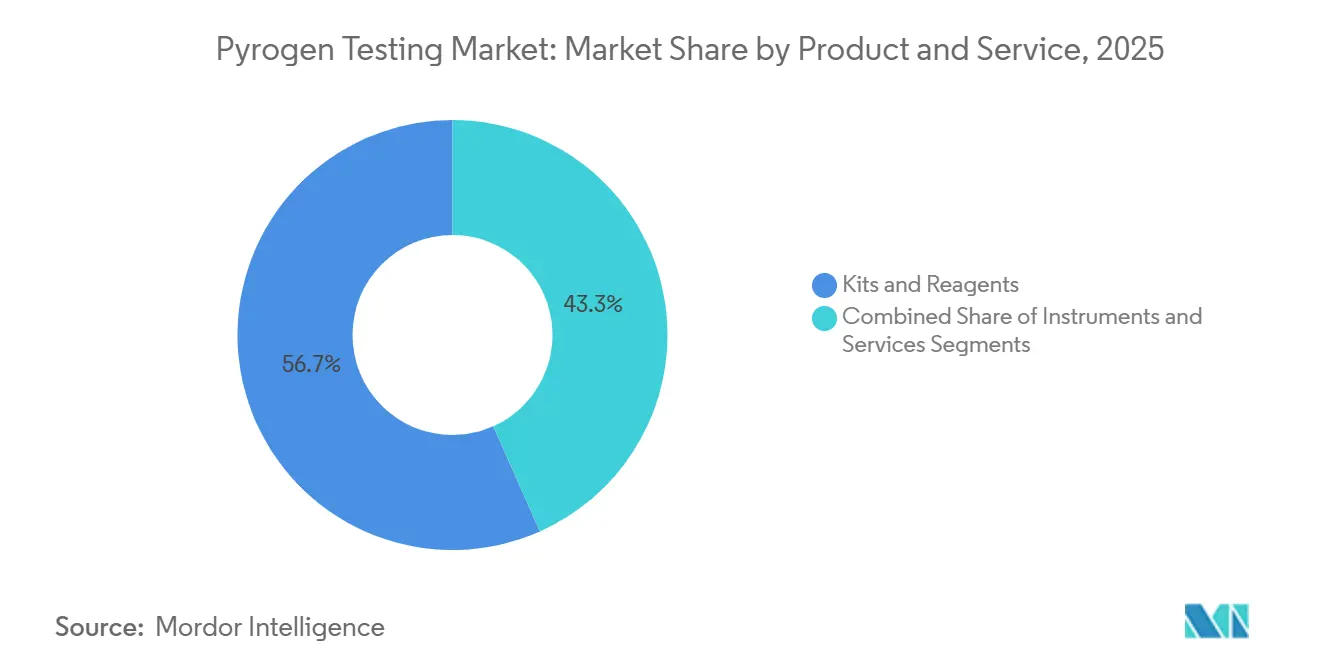
- By product & service, kits and reagents accounted for 56.67% of the pyrogen testing market share in 2025 and instruments are expected to grow at a 13.62% CAGR through 2031.
- By detection technology, animal-based assays commanded 72.52% of revenue in 2025 and recombinant and cell-based methods are on track for a 14.77% CAGR to 2031.
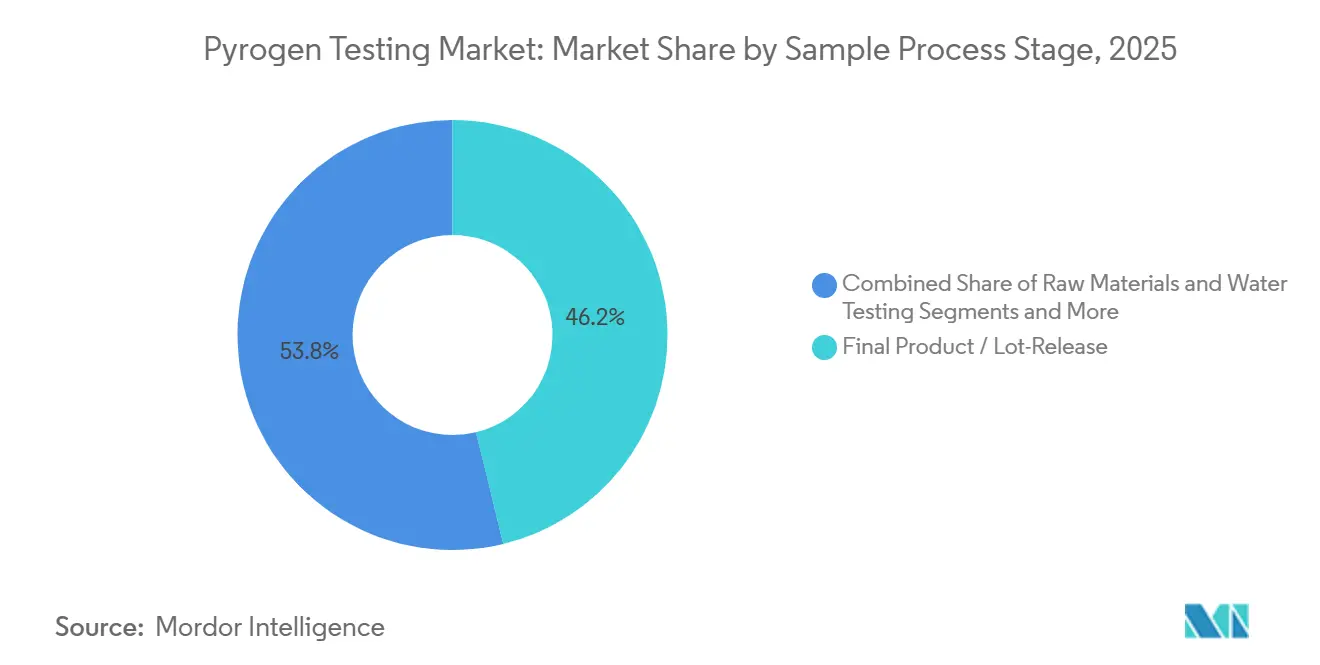
- By sample stage, final product and lot-release testing represented 46.21% of revenue in 2025 and in-process testing is projected to rise at a 14.24% CAGR over the same horizon.
- By test type, LAL tests held 53.14% of revenue in 2025 and the Monocyte Activation Test is forecast to register a 15.72% CAGR to 2031.
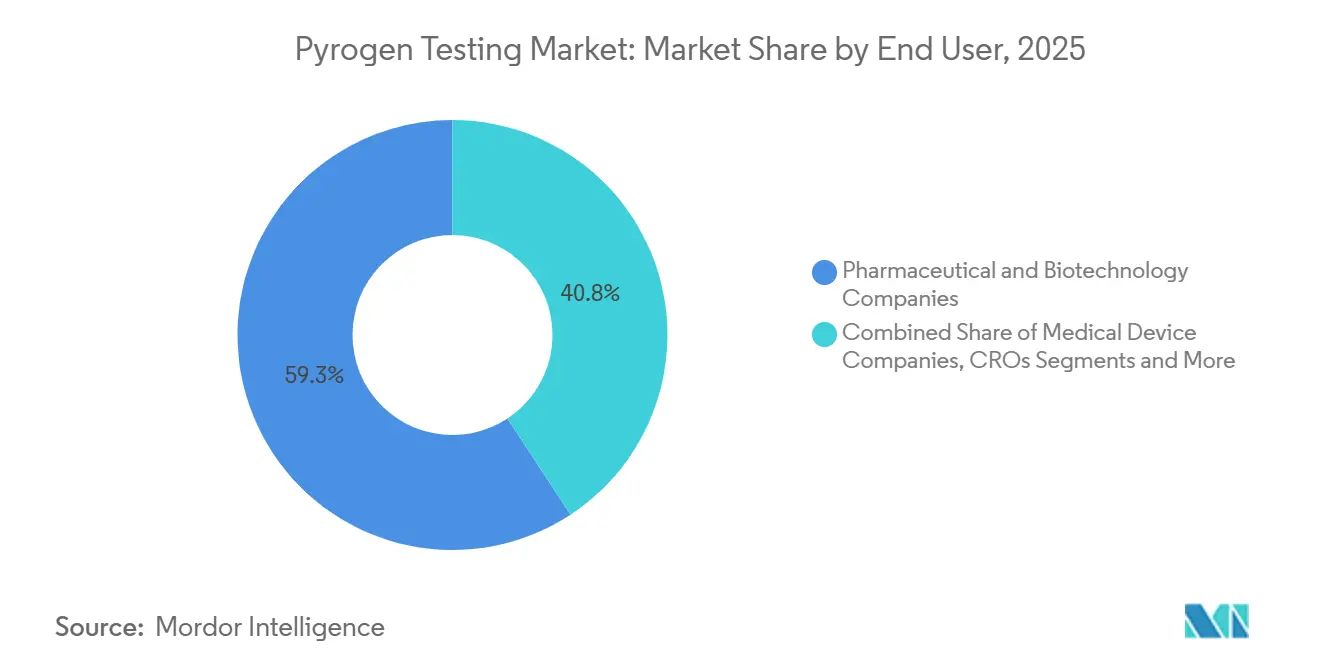
- By end user, pharmaceutical and biotechnology companies generated 59.25% of revenue in 2025 and medical device manufacturers are poised for a 13.44% CAGR through 2031.
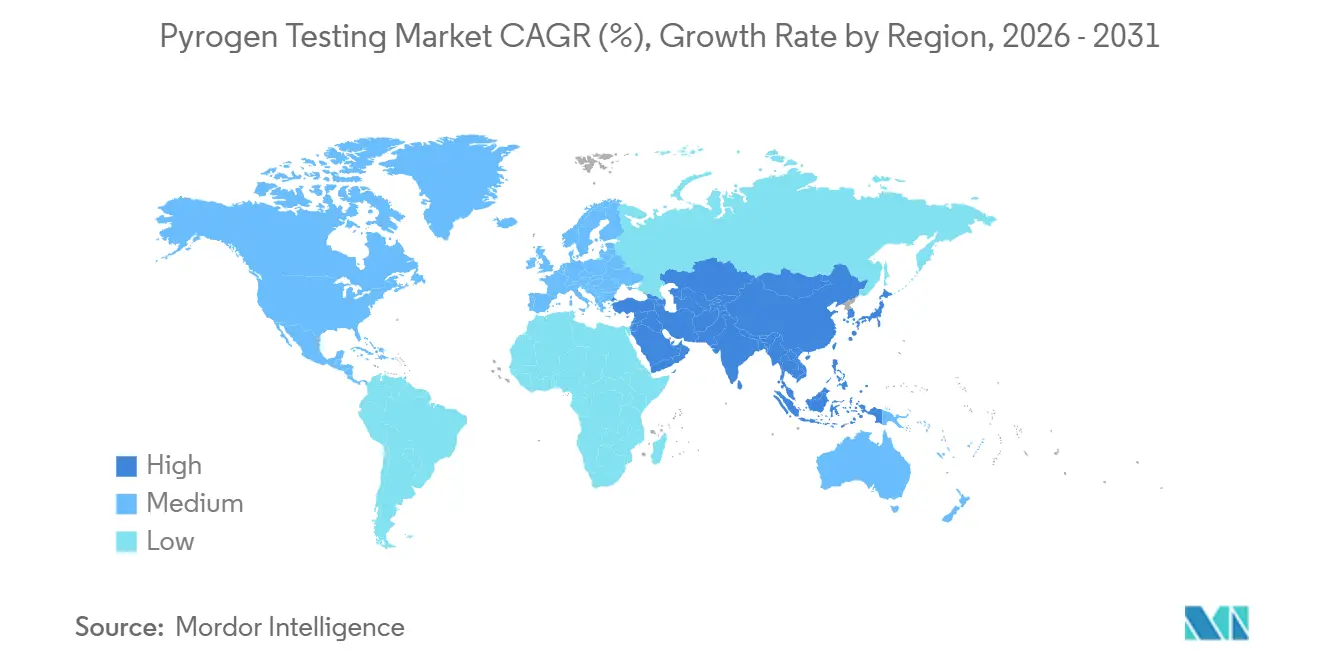
- By geography, North America delivered 39.14% of revenue in 2025 and Asia-Pacific is predicted to post a 13.63% CAGR to 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Pyrogen Testing Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Biologics & vaccine pipeline expansion | +2.3% | North America, Europe, China | Long term (≥ 4 years) |
| Endotoxin limits in revised USP <85>/Ph. Eur. 2.6.14 | +2.1% | North America, Europe | Short term (≤ 2 years) |
| Outsourcing to CROs & CDMOs | +1.8% | Global, fastest in India and China | Medium term (2-4 years) |
| Automation-ready microfluidic analyzers | +1.6% | North America, Europe, Japan | Medium term (2-4 years) |
| EU phase-out of rabbit tests | +1.9% | Europe, spill-over to North America and APAC | Short term (≤ 2 years) |
| Chinese rFC cost advantages | +1.4% | APAC core, spill-over Middle East & Africa | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Biologics & Vaccine Pipeline Expansion
Global regulators cleared 46 novel drugs and 18 biosimilars in 2025, nearly all parenteral, enlarging quality-control sample volumes at every bioprocess stage.[1]U.S. Food and Drug Administration, “CDER Novel Drug Approvals 2025,” FDA, fda.gov Multiple COVID-19 strain-updated mRNA vaccines incorporated new fill-finish lines that demanded fresh component qualifications. Each monoclonal antibody or viral-vector therapy must meet strict ±0.05 EU/mL limits for upstream harvest, downstream pools, and final vials, driving recurring pyrogen testing market demand. Large-scale capital projects by Eli Lilly, AstraZeneca, and Johnson & Johnson are adding sterile capacity that will begin generating validation batches in 2027, locking in sustained testing volumes. Emerging-market producers seeking mutual recognition with ICH jurisdictions are similarly upgrading QC infrastructures, broadening the global footprint of the pyrogen testing market.
Mandatory Endotoxin Limits in New USP 85 and Ph. Eur. 2.6.14
USP’s July 2025 update formally recognizes rFC as equivalent to LAL once equivalence is proven, enabling method substitution without full dossier revalidation. Revision Recognizes Recombinant Factor C,” USP, usp.org">[2]United States Pharmacopeia, “USP Chapter <1085> Revision Recognizes Recombinant Factor C,” USP, usp.org In parallel, Europe’s deletion of the rabbit-pyrogen method effective July 2025 compels immediate MAT adoption for products headed to the EU.[3]European Directorate for the Quality of Medicines & HealthCare, “European Pharmacopoeia Deletes Rabbit Pyrogen Test,” EDQM, edqm.eu These harmonized policies eliminate historical ambiguity that held back recombinant and cell-based platforms and compress compliance timelines for legacy products. Reagent makers have responded with next-generation MAT kits offering 0.05 EU/mL sensitivity, aligning with new pharmacopeial thresholds. Collectively, the rule changes strengthen the regulatory foundation of the pyrogen testing market.
Outsourcing to CROs & CDMOs
Global sponsors are migrating lot-release testing to contract labs to trim fixed costs and leverage specialized talent. Charles River’s 2024 addition of gene-therapy endotoxin services and Thermo Fisher’s GMP laboratory expansion in Wisconsin exemplify capacity scaling. Outsourcing also spreads adoption of fully automated analyzers that smaller biotech firms could not justify in-house, helping to enlarge both service and equipment sub-segments of the pyrogen testing market.
Automation-Ready Microfluidic Analyzers
bioMérieux’s ENDONEXT system processes 120 samples per hour with 95% automation, slashing hands-on time and error rates. Cartridge-based designs from Merck remove reagent preparation steps and permit in-suite testing in fill-finish areas. Integration with LIMS satisfies 21 CFR Part 11, reinforcing data-integrity compliance. The throughput and compliance benefits position automated analyzers as the fastest-growing hardware niche within the broader pyrogen testing market.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High validation cost for MAT & rFC at SMEs | -0.8% | Emerging markets, small biotech hubs | Medium term (2-4 years) |
| Horseshoe-crab harvest limits tightening LAL supply | -0.9% | North America, Europe | Short term (≤ 2 years) |
| Persistent Low Endotoxin Recovery (LER) events | -0.6% | Biosimilar-heavy portfolios worldwide | Medium term (2-4 years) |
| Divergent data-integrity rules complicate automation ROI | -0.5% | Multinational labs in China, India, and highly regulated markets | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
High Validation Cost for SMEs
Adapting a single biologic line to MAT or rFC can exceed USD 500,000 in qualification and documentation spend, a burden that smaller firms often absorb by outsourcing rather than capitalizing new labs. The cost headwind slows the pyrogen testing market transition in emerging regions where access to low-cost validation support remains limited.
Horseshoe-Crab Harvest Restrictions
The Atlantic States Marine Fisheries Commission caps annual harvests at 500,000–700,000 crabs, and 15%–30% bleed mortality creates episodic supply squeezes. Resulting lysate price spikes push users toward rFC yet simultaneously threaten real-time production schedules for manufacturers unable to re-validate quickly—adding volatility to the pyrogen testing market.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Product & Service: Instruments Drive the Next Wave of Expansion
Kits and reagents supplied 56.67% of 2025 revenue, underscoring the razor-and-blade model that anchors the pyrogen testing market size for consumables. However, instruments are poised for a 13.62% CAGR through 2031 as manufacturers install microfluidic systems to combat labor shortages and compress release timelines. Automated readers integrate seamlessly with LIMS, satisfying data-integrity expectations and supporting continuous bioprocessing workflows. Proprietary cartridges ensure recurring revenue, reinforcing supplier lock-in. Services—chiefly CRO lot-release testing—remain attractive to early-stage biotech clients that lack in-house QC, funneling incremental volumes into the pyrogen testing market.
Labor availability and audit-trail scrutiny accelerate capital outlays for fully integrated analyzers, exemplified by Merck’s cartridge-based platform that allows non-specialists to test directly in fill-finish suites. While consumables will continue as the largest revenue pool, hardware adoption raises average selling prices and broadens the pyrogen testing market share of high-margin instrument vendors.

By Detection Technology: Recombinant and Cell-Based Methods Gain Ground
Animal-based assays held 72.52 % of the 2025 pyrogen testing market share, yet recombinant Factor C and MAT platforms are forecast to post a 14.77 % CAGR through 2031. USP Chapter <1085>, effective February 2026, now allows rFC substitution once equivalence is proven, removing the last major barrier to animal-free methods. Europe’s deletion of the rabbit test mandates MAT for all new and legacy products shipped into the EU, compressing timelines for cell-based validation.
Chinese suppliers sell rFC kits 30–40 % below Western LAL prices, widening adoption across APAC facilities and increasing the pyrogen testing market size for recombinant solutions. LAL retains primacy in legacy BLAs but faces episodic supply shocks whenever horseshoe-crab quotas tighten. Rapid cartridge devices now deliver quantitative results in 30 minutes, demonstrating that speed and regulatory clarity together are tipping the technology mix away from animal lysate.
By Sample / Process Stage: In-Process Testing Surges With Continuous Bioprocessing
Final product and lot-release represented 46.21 % of 2025 revenue, underscoring its centrality to batch disposition. However, in-process testing is poised to climb at a 14.24 % CAGR through 2031 as multi-column chromatography and perfusion reactors multiply sample nodes per batch, enlarging the pyrogen testing market size tied to upstream and downstream pools. Automated readers that handle 120 samples per hour enable real-time monitoring rather than end-of-run confirmation, aligning with quality-by-design principles.
Raw-material screening is expanding as single-use bag and tubing vendors must certify low endotoxin loads before shipment. ISO 10993-1:2025 highlights pyrogenicity as a systemic biological hazard, prompting device and biologic makers alike to adopt earlier checkpoints. Medical-device rinse tests remain the smallest slice but are climbing as newer implant standards tighten acceptable endotoxin limits. Overall, migration toward in-process sampling elevates testing volumes well before the final vial stage.

By Test Type: MAT Leads Growth, LAL Holds Legacy Ground
LAL delivered 53.14 % of 2025 revenue and remains deeply embedded in historical submissions. Yet MAT is forecast to grow 15.72 % annually, the fastest rate in the segment, after Europe’s rabbit-test ban took effect in July 2025. MilliporeSigma’s PyroMAT kit achieves 0.05 EU/mL sensitivity, satisfying new pharmacopeial thresholds and assuring broad pyrogen coverage.
Recombinant Factor C adoption accelerates where price and ethical considerations converge, especially in APAC. Conversely, rabbit assays shrink everywhere except markets without explicit prohibitions, but even there, ethical scrutiny is mounting. Together, these shifts reallocate the pyrogen testing market share toward animal-free platforms while leaving LAL as a declining yet significant baseline.
By End User: Pharma & Biotech Dominate, Medical Devices Accelerate
Pharmaceutical and biotechnology companies generated 59.25 % of 2025 revenue, anchored by monoclonal antibodies, mRNA vaccines, and viral vectors that drive high-frequency lot release. Medical-device manufacturers are projected to record a 13.44 % CAGR through 2031, propelled by ISO 10993 revisions that impose stricter systemic-toxicity and pyrogenicity controls.
CROs and CDMOs scale capacity fastest because outsourcing trims fixed costs for smaller sponsors. Charles River’s 2024 viral-vector testing acquisition broadened its endotoxin service portfolio, reinforcing the trend toward specialized third-party quality-control hubs. Academic labs hold a minor share but gain volume from government-funded vaccine research that now requires GMP-level documentation. Collectively, pharma and biotech remain the volume anchors, while device firms inject fresh momentum into growth.

Geography Analysis
North America held 39.14% of 2025 revenue, buoyed by the United States’ dense biopharma pipeline and frequent parenteral approvals. USP’s formal rFC recognition will likely catalyze method substitutions, enlarging the region’s software-integration and equipment opportunities. Canada and Mexico show steady growth as near-shoring of sterile fill-finish expands.
Europe follows as the second-largest region. The July 2025 rabbit-test sunset forces continent-wide MAT adoption, triggering equipment and kit purchases. Germany, the United Kingdom, and France continue to anchor demand, while Eastern Europe gains share through biosimilar outsourcing.
Asia-Pacific is projected to lead growth at 13.63% CAGR through 2031, fueled by China’s domestic rFC capacity and India’s rising sterile-drug export ambitions. Japan and South Korea complement the regional picture with automation upgrades to offset aging technical workforces. The Middle East and Africa plus South America remain nascent but attractive targets as multinational firms and local governments collaborate on GMP facility build-outs, planting future seeds for the pyrogen testing market.
Competitive Landscape
Competition is moderately fragmented. Legacy lysate suppliers—Associates of Cape Cod, Charles River, Lonza—compete on sensitivity and regulatory pedigree, while bioMérieux, Merck, and Sartorius aim to dominate the automation layer with analytics-ready hardware. Chinese newcomers ACROBiosystems and Hzymes Biotech leverage cost advantages to penetrate APAC and emerging markets, forcing incumbents to bundle consumables, software, and multi-year service agreements. Strategic M&A continues, highlighted by Charles River’s 2024 viral-vector testing acquisition that deepened its gene-therapy QC footprint. Vendors able to satisfy both FDA 21 CFR Part 11 and China’s T/CNHAW 0015-2025 electronic-record criteria are best positioned to win multi-site global contracts, underscoring compliance as a critical differentiator across the pyrogen testing market.
Pyrogen Testing Industry Leaders
-
Merck KGaA
-
Lonza Group
-
Charles River Laboratories International Inc.
-
Associates of Cape Cod Inc.
-
Thermo Fisher Scientific Inc.
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- January 2026: BETMAT Biotechnology launched a premium sustainable endotoxin-testing service portfolio targeting next-generation QC laboratories.
- December 2025: The European Pharmacopoeia approved a revision that fully integrates recombinant Factor C into chapter 2.6.14 and removes rabbit-pyrogen testing effective January 2026.
Research Methodology Framework and Report Scope
Market Definitions and Key Coverage
Our study defines the pyrogen testing market as the revenue generated by in-vitro kits, reagents, instruments, and contract services that detect endotoxin or other fever-inducing contaminants in parenteral drugs, vaccines, biologics, and high-risk medical devices.
Environmental or food-sector endotoxin assays, animal-based research reagents, and routine sterility tests are outside this scope.
Segmentation Overview
-
By Product & Service
- Kits & Reagents
- Instruments
- Services
-
By Detection Technology
- Animal-Based Tests
- Recombinant / Cell-Based Tests
- Instrument-Integrated Rapid Systems
-
By Sample / Process Stage
- Raw Materials & Water Testing
- In-Process (Upstream & Downstream)
- Final Product / Lot-Release
- Medical Device Surface & Rinse Tests
-
By Test Type
- Limulus Amebocyte Lysate (LAL) Tests
- Recombinant Factor C (rFC) Assays
- Monocyte Activation Test (MAT)
- Rabbit Pyrogen Test
-
By End User
- Pharmaceutical & Biotechnology Companies
- Medical Device Companies
- Contract Research Organizations (CROs)
- Academic & Research Institutes
-
By Geography
-
North America
- United States
- Canada
- Mexico
-
Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Rest of Europe
-
Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia-Pacific
-
Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
-
South America
- Brazil
- Argentina
- Rest of South America
-
North America
Detailed Research Methodology and Data Validation
Primary Research
Mordor analysts interviewed quality-control heads at drug makers, validation leads at contract testing labs, and regional regulatory consultants across North America, Europe, and Asia-Pacific. These conversations validated LAL kit price dispersion, rFC adoption rates, and service throughput assumptions, closing gaps that literature alone could not bridge.
Desk Research
We first screened open datasets, including United States FDA recall archives, EMA EudraGMDP certificates, USP Chapter <86> compendia updates, and World Health Organization TRS notes, to map regulatory test demand shifts. Statistics from bodies such as the International Federation of Pharmaceutical Manufacturers & Associations and the World Customs Organization helped size global injectable output and trade. Supplementary insights came from company 10-Ks, SEC filings, and media retrieved through Dow Jones Factiva, as well as financial snapshots on D&B Hoovers. This multi-angle groundwork sketched the demand pool and typical selling prices. The sources listed are illustrative, not exhaustive; many additional references informed data collection and clarification.
Market-Sizing & Forecasting
A hybrid top-down view, linking injectable production volumes and average pyrogen-test frequency per batch, was cross-checked with selective bottom-up roll-ups of major kit suppliers and contract labs to fine-tune totals. Key variables include parenteral batch counts, reagent price bands, penetration of rFC and MAT methods, regional biologics pipeline expansion, and outsourcing intensity. Multivariate regression on these drivers underpins the 2025-2030 forecast, with scenario analysis around rFC uptake guiding upside and downside cases.
Data Validation & Update Cycle
Outputs pass variance checks against external shipment data and historical growth norms, followed by a peer review. Reports refresh annually, and interim revisions trigger when regulatory or supply-chain events materially shift baseline assumptions.
Why Mordor's Pyrogen Testing Baseline Commands Reliability
Published figures often diverge because firms choose different product mixes, currency bases, and refresh cadences.
We anchor our 2025 baseline at USD 1.59 billion, integrating instruments and services that some publishers leave out.
Benchmark comparison
| Market Size | Anonymized source | Primary gap driver |
|---|---|---|
| USD 1.59 bn (2025) | Mordor Intelligence | - |
| USD 1.47 bn (2024) | Global Consultancy A | Omits testing services; 2024 exchange rates |
| USD 1.30 bn (2023) | Industry Journal B | Historical base year and limited geography |
| USD 1.43 bn (2024) | Research Boutique C | Focuses on consumables only |
Taken together, the comparison shows that Mordor's disciplined scope selection, current-year currency normalization, and dual-track validation provide a balanced, transparent baseline that decision-makers can replicate and trust.


Key Questions Answered in the Report
How large is the pyrogen testing market today?
The pyrogen testing market size reached USD 1.73 billion in 2026 and is projected to climb to USD 2.99 billion by 2031.
Which region is growing fastest?
Asia-Pacific is forecast to register a 13.63% CAGR through 2031, the quickest pace worldwide.
What technology is replacing rabbit-pyrogen tests in Europe?
The Monocyte Activation Test has become the mandated alternative, with full enforcement starting July 2025.
Why is recombinant Factor C gaining popularity?
Pharmacopeial revisions in the United States and Europe now recognize rFC as equivalent to LAL, eliminating prior regulatory barriers.
What challenges do small manufacturers face when adopting MAT or rFC?
Validation expenses can top USD 500,000 per product line, a significant hurdle for SMEs with limited capital.
How are suppliers addressing horseshoe-crab lysate shortages?
Vendors are expanding recombinant and cartridge-based platforms, while regulators encourage rFC adoption to ease supply pressure.
Page last updated on:









