Prefilled Syringes Market Size and Share
Prefilled Syringes Market Analysis by Mordor Intelligence
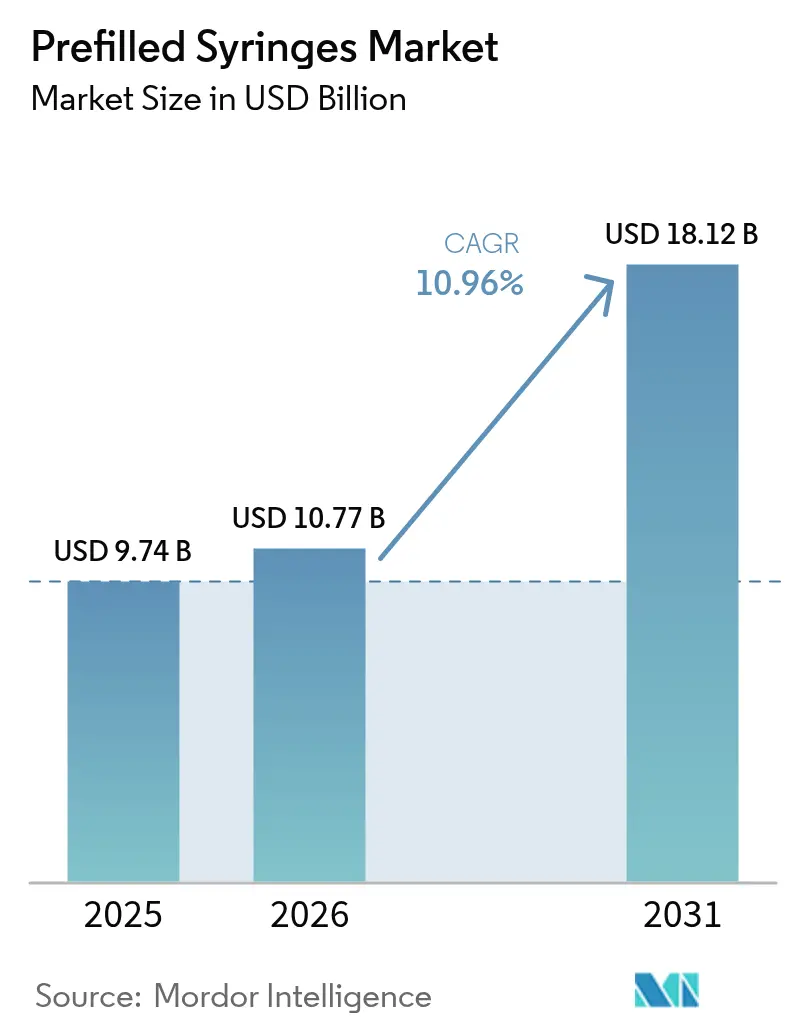
The Prefilled Syringes Market size is expected to increase from USD 9.74 billion in 2025 to USD 10.77 billion in 2026 and reach USD 18.12 billion by 2031, growing at a CAGR of 10.96% over 2026-2031.
Capacity bottlenecks in medical-grade borosilicate glass, the maturation of cyclic-olefin polymer alternatives, and stricter safety regulations are redefining competitive dynamics across the supply chain. Biologic drugs that demand cold-chain integrity are displacing vial-and-syringe workflows and anchoring long-term demand for ready-to-administer devices. Hospitals are purchasing safety-engineered formats to comply with needlestick-injury mandates, while payers reward home-care pathways that rely on user-friendly autoinjectors. Suppliers that bundle nested-tub packaging, automated particle inspection, and connected-device telemetry are winning long-term agreements with tier-1 pharmaceutical companies.
Key Report Takeaways
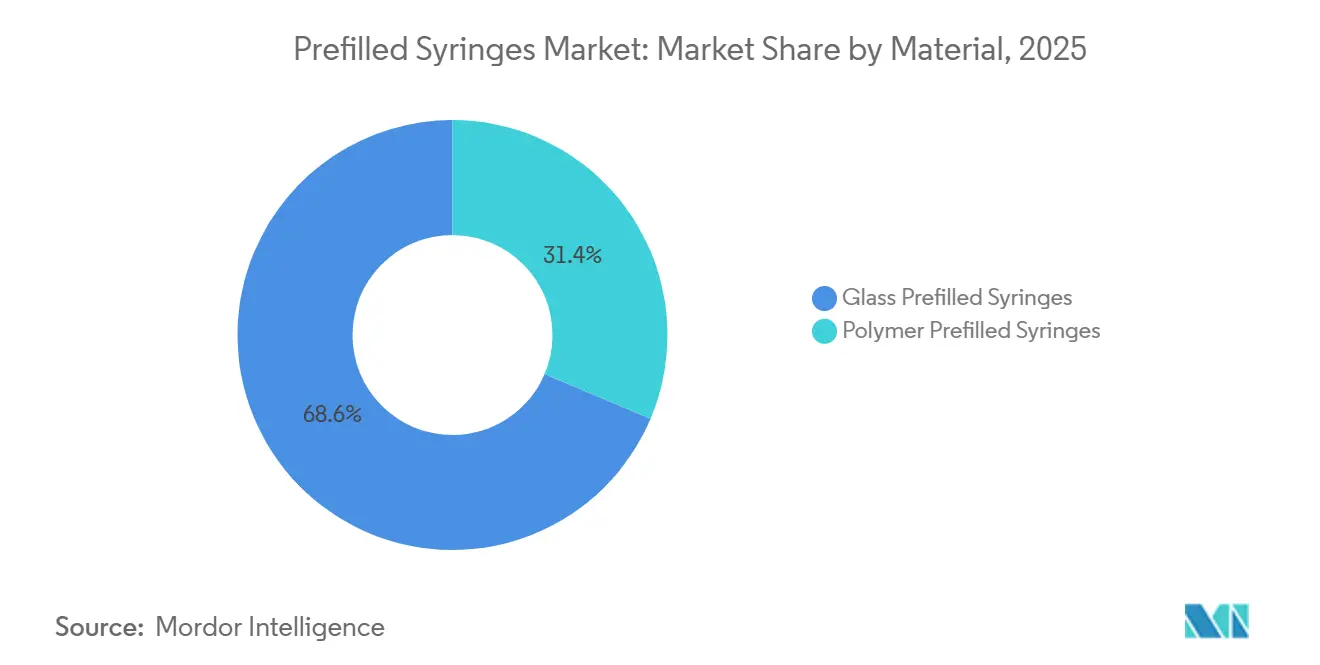
- By material, glass captured 68.63% of 2025 revenue, whereas polymer is the fastest-growing segment, with a 11.06% CAGR to 203.
- By application, diabetes led with 34.76% of the prefilled syringes market share in 2025; vaccines are forecast to expand at a 13.63% CAGR through 2031.
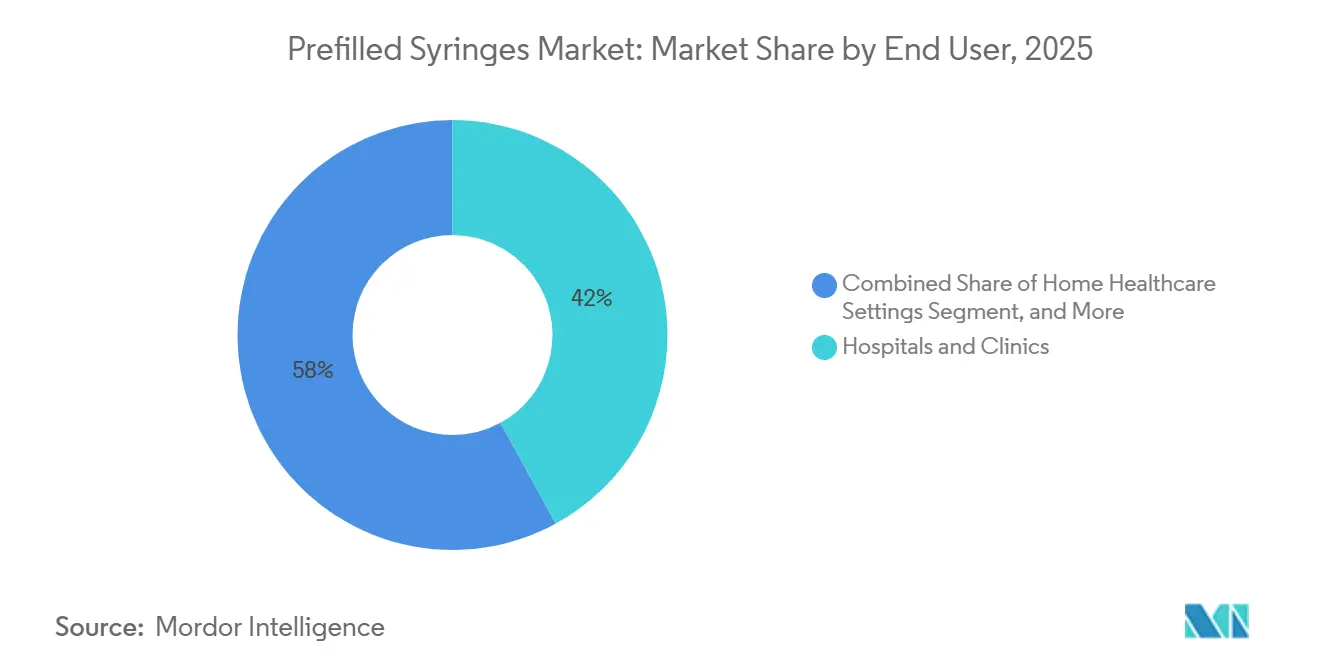
- By end user, hospitals and clinics held a 42.02% share of the prefilled syringes market in 2025, while home healthcare is advancing at a 11.92% CAGR.
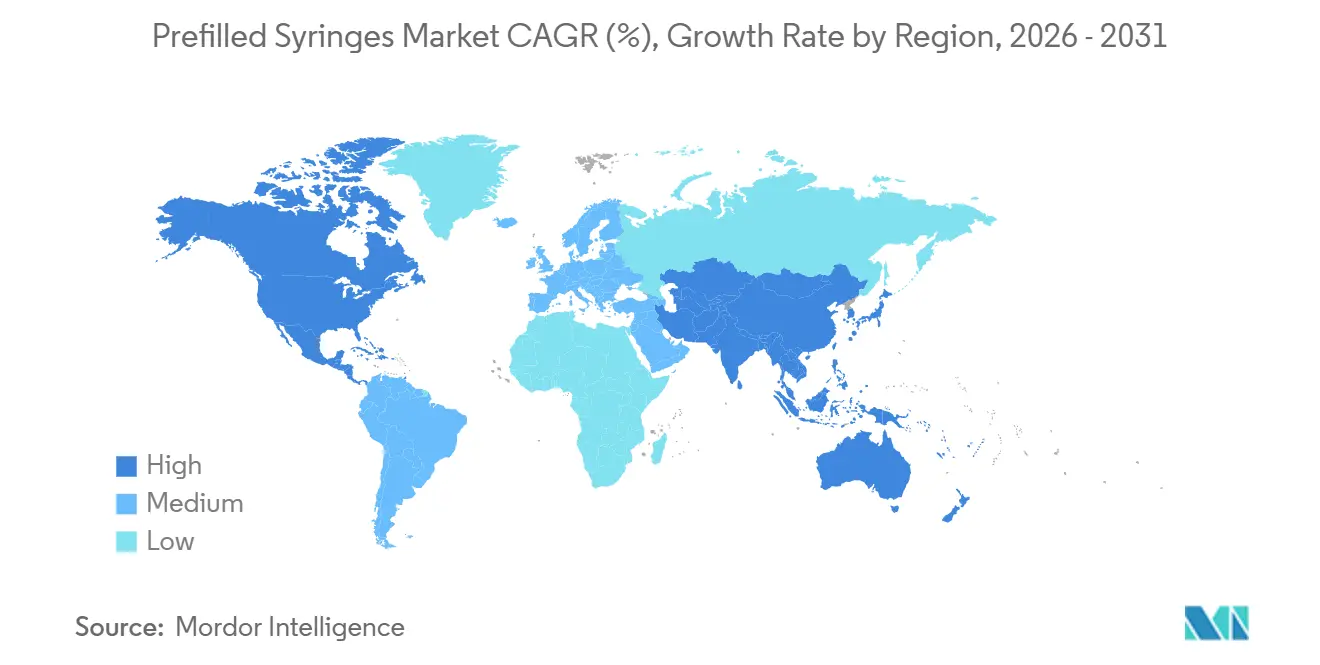
- By geography, North America accounted for 38.41% of 2025 value, whereas Asia-Pacific is projected to post a 12.27% CAGR to 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Prefilled Syringes Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising Prevalence of Chronic Diseases | 1.8% | Global, with highest intensity in North America and Europe | Long term (≥ 4 years) |
| Shift Toward Self-Administration & Home-Healthcare Models | 1.5% | North America and Europe core, expanding to urban APAC | Medium term (2-4 years) |
| Acceleration of Biologics/Biosimilars Pipeline Requiring Ready-To-Fill Containers | 2.0% | Global, led by North America and EU manufacturing hubs | Long term (≥ 4 years) |
| Regulatory Emphasis on Needlestick-Injury Prevention | 0.8% | EU and North America mandated, gradual APAC adoption | Medium term (2-4 years) |
| Emergence of Connected "Smart" Syringes Enabling Dose-Tracking | 0.6% | North America and select EU markets, pilot programs in Japan | Short term (≤ 2 years) |
| Capacity Investments In Polymer PFS Lines to Mitigate Glass Shortages | 1.2% | Global, concentrated in Germany, US, and Japan manufacturing sites | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Rising Prevalence of Chronic Diseases
Diabetes prevalence doubled between 1990 and 2022, climbing to 14.0% of the global adult population and pushing weekly GLP-1 therapies into mainstream use. Prefilled syringes eliminate reconstitution errors and shorten training time, thereby lowering annual treatment costs by USD 8,000–12,000 per rheumatoid arthritis patient compared with infusion-center administration.[1]Global Diabetes Report 2025, World Health Organization, who.int Payer contracts in the United States and Germany already link reimbursement to adherence metrics pulled from Bluetooth-enabled autoinjectors, giving manufacturers a revenue-protection incentive to supply smart formats. Chronic-disease growth, therefore, delivers a structural lift to the prefilled syringes market, especially for long-acting biologics that require precise dosing. Forward pipelines of subcutaneous antibodies suggest a durable demand floor extending past 2031.
Shift Toward Self-Administration & Home-Healthcare Models
Medicare Part B coverage expanded in 2024 to support home-injected biologics, shifting an estimated 25–30% of specialty injectable volume out of hospital outpatient departments by 2028. Telehealth platforms paired with Bluetooth autoinjectors reduced readmissions due to non-compliance by up to 22% in U.S. pilots. European structural funds worth EUR 5 billion are earmarked for home-care infrastructure, accelerating the policy’s export to the EU. Prefilled formats remove the final reconstitution barrier, lowering skill requirements and enabling direct-to-consumer channels. Combined, these factors expand the addressable market for prefilled syringes beyond hospital walls.
Acceleration of Biologics / Biosimilars Pipeline Requiring Ready-To-Fill Containers
The FDA Purple Book listed 48 approved biosimilars by end-2025, most launched in prefilled syringe clones to mirror originator devices and encourage switching.[2]FDA Combination-Product Guidance, fda.gov Monoclonal antibodies are viscous and shear-sensitive; siliconized glass or cyclic-olefin polymer syringes minimize protein aggregation compared with vial transfer. CDMOs have responded with 15–20% capacity growth in 2024–2025, favoring isolator-based lines that support quick changeovers at ≤400 units per minute. Device suppliers offering nested-tub packaging reduce line prep time, giving biologics sponsors a speed-to-launch advantage that deepens the moat for integrated syringe platforms. Consequently, the prefilled syringes market benefits directly from every new biologic or biosimilar license.
Regulatory Emphasis on Needlestick-Injury Prevention
OSHA fines for noncompliance reached USD 15,625 per infraction in 2025, steering U.S. hospitals toward prefilled passive-safety syringes. EU Directive 2010/32/EU carries similar obligations, yet 23% of Eastern European facilities lag in implementation due to budget gaps. Reported needlestick incidents surged by 23% during COVID-19 staffing shortages, reinforcing the need to switch to sharps with automatic needle shields. Hospitals justify the 15–25% device premium by avoiding post-exposure prophylaxis, typically costing USD 500–3,000 per event. Safety regulation, therefore, underpins a predictable tailwind for the prefilled syringes market.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Complex Extractables/Leachables Compliance Elevating CMC Costs | -1.2% | Global, most acute for small biotech firms in North America and EU | Medium term (2-4 years) |
| Supply-Chain Constraints for Medical-Grade Borosilicate Glass | -0.9% | Global, with tightest supply in North America and Europe | Short term (≤ 2 years) |
| Tightening PFAS & Silicone-Oil Regulations Impacting Legacy Lubricants | -0.5% | North America and EU regulatory focus, spillover to APAC | Medium term (2-4 years) |
| Growing Environmental Scrutiny on Single-Use Plastics Disposal | -0.4% | EU leading, with emerging pressure in North America and select APAC markets | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Complex Extractables / Leachables Compliance Elevating CMC Costs
ISO 10993-18:2020 compels exhaustive chemical characterization under worst-case temperature and time scenarios, often adding 6–9 months to CMC timelines.[3]ISO 10993-18:2020, International Organization for Standardization, iso.org FDA guidance sets an analytical threshold of 0.15 µg/day above which toxicology justifications are mandatory. Redundant regional standards force duplicate testing for U.S., EU, and Asian submissions, consuming up to 12% of mid-tier biosimilar development budgets. Cyclic-olefin polymers lack the historical safety database of glass, so sponsors face even more scrutiny, extending review cycles. These delays temper near-term growth for the prefilled syringes market, especially for cash-constrained biotech entrants.
Supply-Chain Constraints for Medical-Grade Borosilicate Glass
Vial demand monopolized furnace capacity through 2025, pushing syringe-grade glass lead times to more than 18 months. New furnaces cost USD 216–324 million and have decade-long lifecycles, limiting rapid expansion. Shortages lifted spot prices by 20–30% above contract levels; firms without long-term agreements weighed the cost of polymer requalification against delayed launches. Concentrated production in Germany and Japan exposes the supply chain to energy-price shocks and labor disputes. These structural weaknesses curb near-term upside for the prefilled syringes market until diversified capacity comes online.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Material: Polymer Gains Ground Amid Glass Fragility
Glass accounted for 68.63% of 2025 revenue, reflecting entrenched regulatory acceptance and mature extractables data. Nonetheless, polymer’s 11.06% CAGR signals a decisive tilt in new capacity adds and future launches, particularly for high-viscosity monoclonal antibodies at fracture risk during autoinjector actuation. Polymer also shields sponsors from glass-particle recalls and borosilicate shortages, prompting parallel validation programs despite the upfront burden of extractables testing. SCHOTT and Gerresheimer are hedging by adding polymer lines while maintaining glass leadership, confirming a dual-track strategy. These moves ensure the prefilled syringes market retains supplier diversity and capacity resilience, even as glass remains indispensable for oxygen-sensitive vaccines requiring long shelf lives.

By Application: Diabetes Dominates, Vaccines Surge
Diabetes accounted for 34.76% of 2025 revenue, as GLP-1 agonists scaled rapidly and insulin analogs remained stable. The launch pace of GLP-1 class line extensions suggests a sustained volume floor, anchoring one-third of the prefilled syringes market share through mid-decade. RSV and endemic COVID-19 campaigns boost vaccine volume, delivering a 13.63% CAGR that will lift the prefilled syringes market size for immunizations by 2031.
Rheumatoid-arthritis biosimilars and anticoagulants provide steady mid-single-digit expansion, while fertility, immunoglobulins, and emergency drugs contribute niche but lucrative margins. Collectively, high-frequency dosing regimens favor connected autoinjectors that feed adherence data into payer dashboards, reinforcing the shift away from vial formats.
By End-User: Home Healthcare Gains Momentum
Hospitals and clinics retained 42.02% of 2025 spending due to complex cold-chain biologics and strict sharps-safety protocols. Yet home healthcare’s 11.92% CAGR indicates a structural hand-off of chronic-disease administration to patients. By 2031, home settings are positioned to overtake ambulatory centers and claim nearly one-third of the prefilled syringes market. Medicare policy changes, telehealth integration, and cost parity between home injection and clinic visits accelerate this trend.
Retail pharmacies, long-term care, and occupational clinics round out the landscape, with influenza and B12 shots anchoring predictable annual volume. Human-factors design—audible clicks, large grips, and clear dose windows remain critical to reduce user error across all sites.

Geography Analysis
North America captured 38.41% of 2025 revenue, buoyed by dense biologics clusters in New Jersey, North Carolina, and California, as well as OSHA safety mandates. Medicare home care coverage is projected to divert up to 30% of specialty injectable volume by 2028, cementing demand for patient-centric devices. Canada’s regulatory reciprocity and Mexico’s CDMO expansions extend the regional supply chain, supporting steady growth in the prefilled syringes market.
Europe’s regulatory drivers include EU Directive 2010/32/EU and aggressive biosimilar adoption, keeping the region a stronghold for glass-to-polymer innovation. Structural funds of EUR 5 billion for home-care programs through 2027 will expand decentralized administration in underserved Eastern regions, though uneven compliance with sharps mandates continues to restrain uptake.
Asia-Pacific is the growth engine at a 12.27% CAGR. India’s Production Linked Incentive scheme, China’s shortened biosimilar approval windows, and Japan’s reimbursement premium for connected devices combine to amplify local manufacturing and consumption. Australia and South Korea, with tight regulatory regimes, serve as beachheads for smart-device pilots that later scale across ASEAN.
Competitive Landscape
The market is moderately consolidated. Becton, Dickinson, and West Pharmaceutical Services leverage vertical integration across glass tubing, elastomers, and inspection systems to secure long-term supply contracts. BD’s Neopak and Effivax lines underpin 5.9% organic growth, supported by capacity expansions in North Carolina and Ireland. West Pharmaceutical Services pairs Daikyo stoppers with SmartDose wearables, capturing high-margin demand for combination products.
CDMOs, notably Vetter Pharma and Catalent, provide turnkey fill-finish solutions that de-risk launches for sponsors without in-house aseptic capability. Their investments in isolator lines and FlexDirect small-batch modules answer the biosimilar wave. Polymer specialists such as SiO2 Materials Science and Stevanato Group push cyclic-olefin innovation, aided by silicon-free technologies that sidestep PFAS scrutiny.
Technology differentiation revolves around nested-tub packaging, automated sub-visible particle inspection, and connected caps. Suppliers able to satisfy ISO 11608 ergonomics while bundling telemetry enjoy superior pricing power. Regional production footprints further influence buyer decisions in an era of supply-chain de-risking.
Prefilled Syringes Industry Leaders
Becton Dickinson and Company
West Pharmaceutical Services, Inc.
Gerresheimer AG
Terumo Corporation
SCHOTT AG
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- April 2025: argenx gained FDA clearance for a patient-self-injected Vyvgart Hytrulo prefilled syringe covering generalized myasthenia gravis and CIDP.
- September 2024: BD launched the Neopak XtraFlow Glass Prefillable Syringe and unveiled new capacity for the wider Neopak platform.
- July 2024: Genentech won FDA approval for the Vabysmo prefilled syringe, the first bispecific antibody in prefilled form for retinal disorders.
- May 2024: Hikma Pharmaceuticals introduced Midazolam Injection, USP, in 2 mg/2 mL and 10 mg/2 mL strengths, debuting the product in US hospitals in a prefilled syringe.
Global Prefilled Syringes Market Report Scope
As per the scope of this report, a prefilled syringe is a single dose packet of the parenteral drug, to which a needle has been fixed by the manufacturer. Prefilled syringes provide greater patient safety by reducing the potential for inadvertent needle sticks and exposure to toxic products that can occur while drawing medication from vials.
The Prefilled Syringes Market Report is Segmented by Material (Glass, Polymer), Application (Diabetes, Rheumatoid Arthritis, Vaccines, Anticoagulants, Other Applications), End-user (Hospitals & Clinics, Ambulatory Surgical Centers, Home Healthcare Settings, Other End Users), and Geography (North America, Europe, Asia-Pacific, Middle East & Africa, South America). The Market Forecasts are Provided in Terms of Value (USD). The market report also covers the estimated market sizes and trends for 17 different countries across major regions globally. The report offers the value (in USD million) for the above segments.
| Glass Prefilled Syringes |
| Polymer Prefilled Syringes |
| Diabetes |
| Rheumatoid Arthritis |
| Vaccines |
| Anticoagulants |
| Other Applications |
| Hospitals & Clinics |
| Ambulatory Surgical Centers |
| Home Healthcare Settings |
| Other End Users |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East & Africa | GCC |
| South Africa | |
| Rest of Middle East & Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Material | Glass Prefilled Syringes | |
| Polymer Prefilled Syringes | ||
| By Application | Diabetes | |
| Rheumatoid Arthritis | ||
| Vaccines | ||
| Anticoagulants | ||
| Other Applications | ||
| By End-user | Hospitals & Clinics | |
| Ambulatory Surgical Centers | ||
| Home Healthcare Settings | ||
| Other End Users | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East & Africa | GCC | |
| South Africa | ||
| Rest of Middle East & Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the projected value of the prefilled syringes market in 2031?
The market is forecast to reach USD 18.12 billion by 2031, growing at a 10.96% CAGR from 2026 to 2031.
Which application is expanding fastest through 2031?
Vaccines lead growth at a 13.63% CAGR as RSV and endemic COVID-19 shots shift to prefilled formats.
Why are polymer syringes gaining share?
They resist breakage, avoid glass-particle contamination, and enable silicone-free designs, supporting 11.06% CAGR adoption.
How is Medicare policy influencing demand?
Part B now reimburses home-injected biologics, redirecting up to 30% of specialty volume from hospitals to patient homes by 2028.
Which region shows the highest future CAGR?
Asia-Pacific is set to expand at 12.27% CAGR, driven by India’s incentives, China’s faster approvals, and Japan’s premium for connected devices.
Page last updated on:









