Orthobiologics Market Size and Share
Orthobiologics Market Analysis by Mordor Intelligence
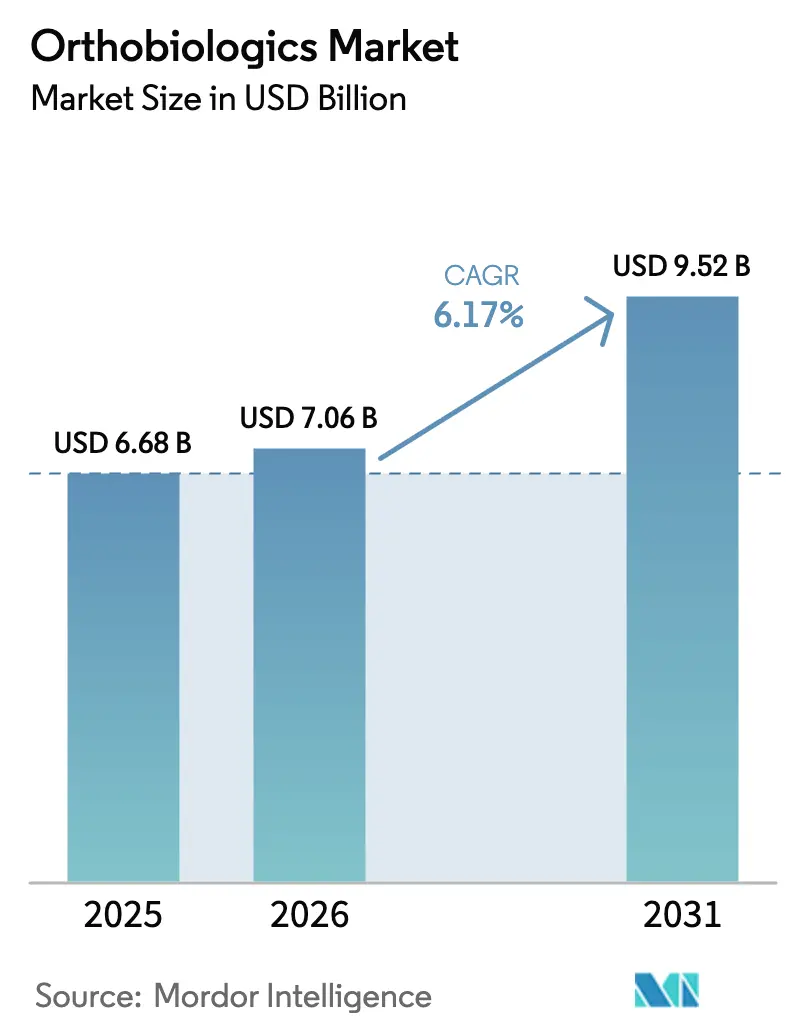
The Orthobiologics Market size is expected to increase from USD 6.68 billion in 2025 to USD 7.06 billion in 2026 and reach USD 9.52 billion by 2031, growing at a CAGR of 6.17% over 2026-2031.
Short-term growth reflects rising clinical demand driven by the prevalence of osteoarthritis, which continues to accelerate as global populations age. Commercial momentum is tempered by patchy reimbursement for platelet-rich plasma (PRP) and stem cell therapies, leaving many interventions as cash-pay procedures despite 18 new PRP preparation devices clearing the FDA’s 510(k) pathway in 2024. Domestic manufacturing capacity expanded in 2025 as tissue processors reshored production to offset Section 232 steel and aluminum tariffs that added 25% to the cost of imported orthopedic hardware. Collectively, these forces position the orthobiologics market for steady rather than explosive growth during the forecast window.
Key Report Takeaways
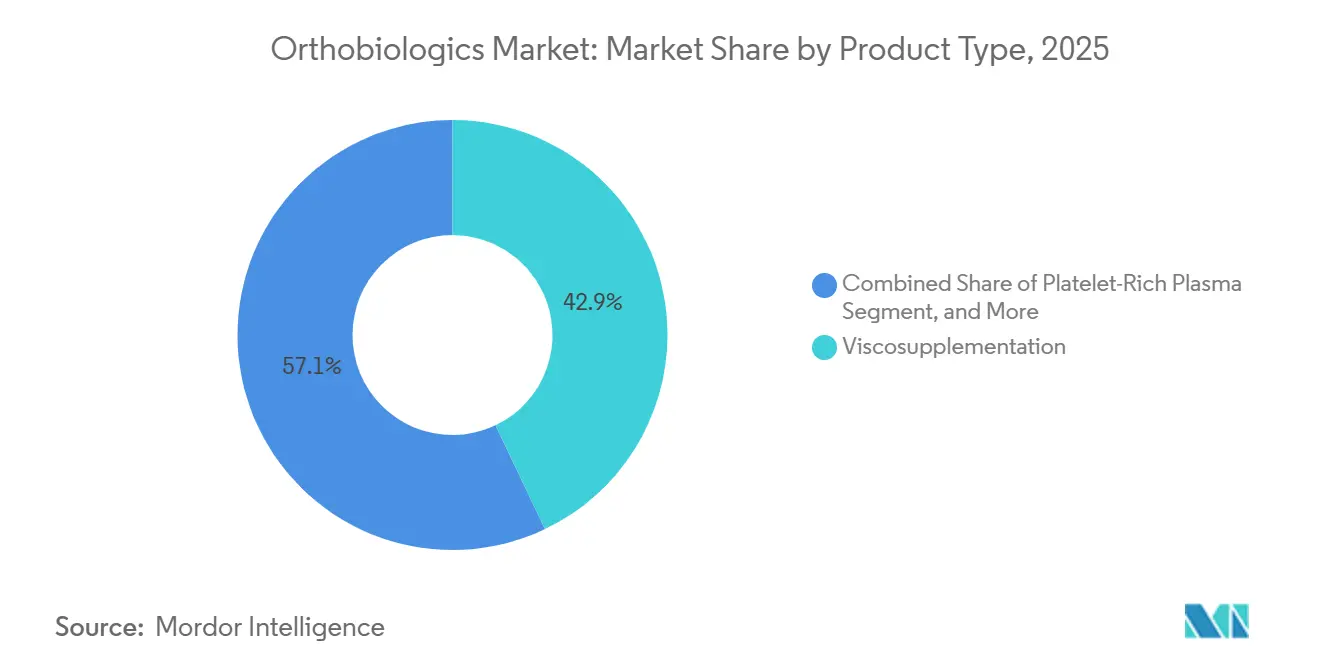
- By product type, viscosupplementation led with 42.92% of the orthobiologics market share in 2025, while platelet-rich plasma is forecast to expand at a 7.09% CAGR through 2031.
- By application, spinal fusion accounted for 52.64% of 2025 revenue, whereas osteoarthritis and joint degeneration are advancing at a 9.63% CAGR to 2031.
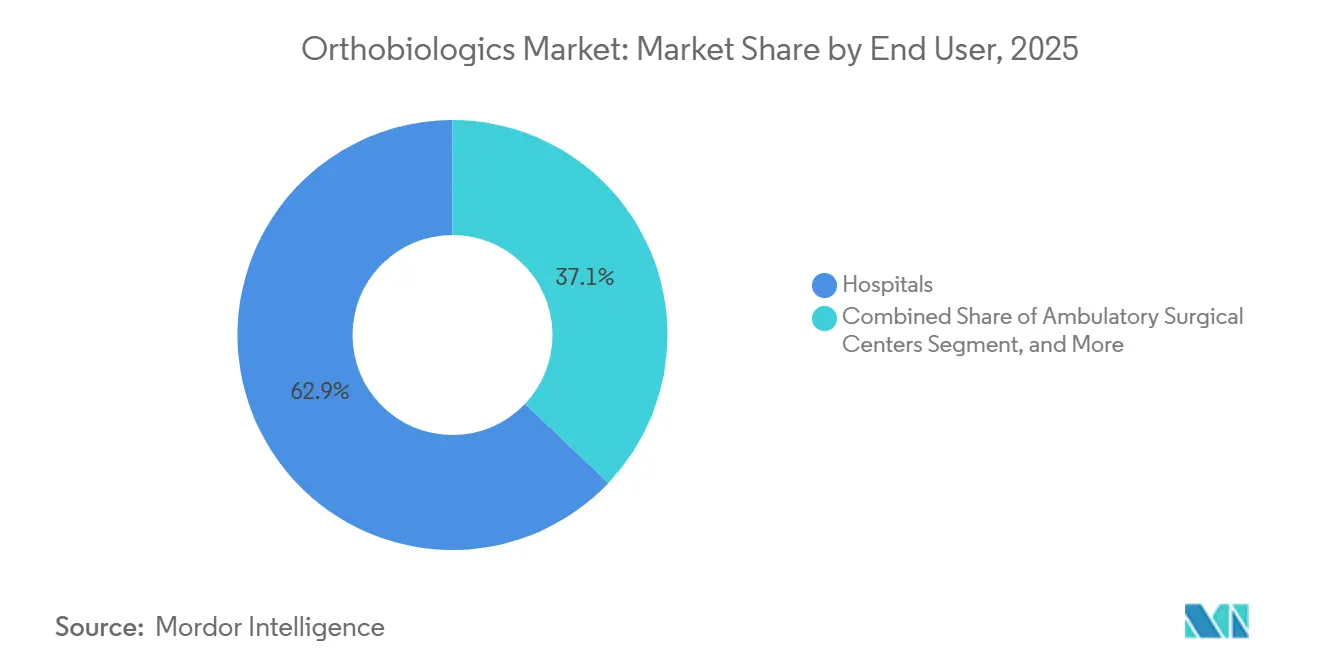
- By end user, hospitals controlled 62.92% of revenue in 2025; ambulatory surgical centers are growing fastest at an 8.18% CAGR over the same period.
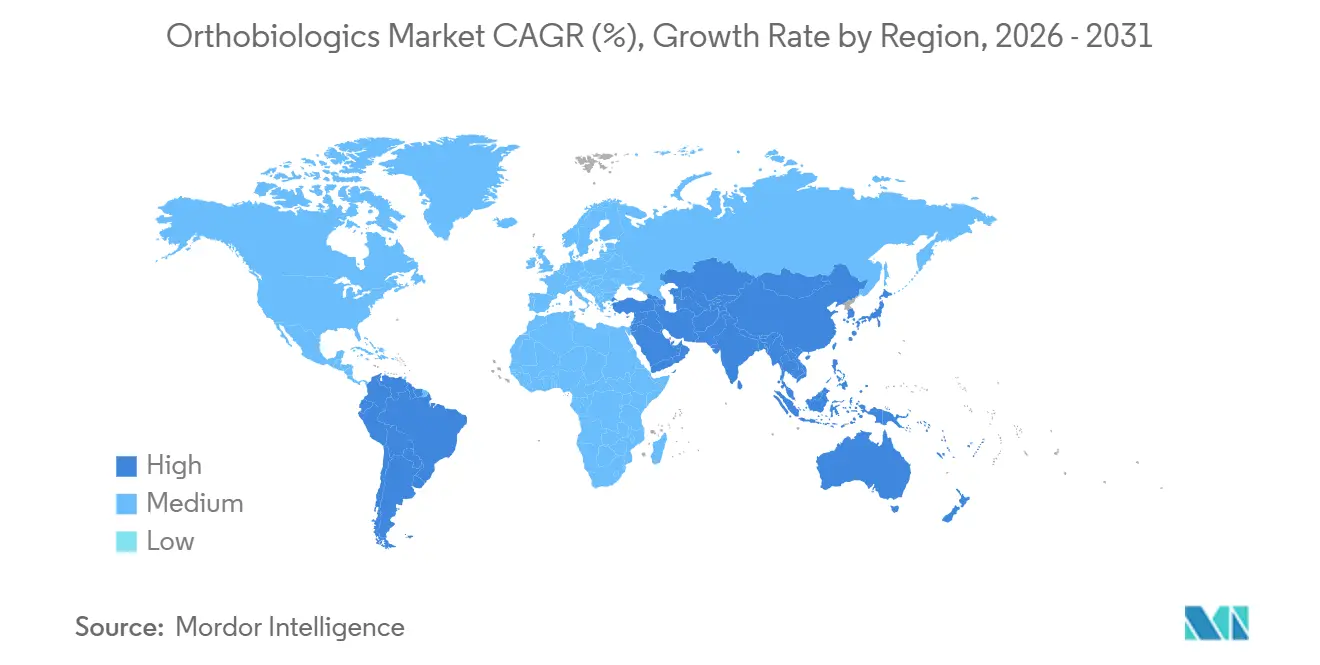
- By geography, North America captured 43.17% of global 2025 revenue, while Asia-Pacific is projected to expand at an 11.27% CAGR to 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Orthobiologics Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising Osteoarthritis Prevalence & Aging Demographics | +1.2% | Global, acute in North America, Europe, East Asia | Long term (≥ 4 years) |
| Shift Toward Minimally-Invasive & Outpatient Orthopedic Procedures | +0.9% | North America, Europe, early adoption in urban Asia-Pacific markets | Medium term (2-4 years) |
| Advances in Regenerative Biomaterials | +0.7% | Global, led by North American and EU research hubs | Medium term (2-4 years) |
| Increasing Sports Injuries Demanding Faster Recovery Solutions | +0.6% | North America, Europe, affluent Asia-Pacific cities | Short term (≤ 2 years) |
| Proliferation of In-Hospital Biologics Labs | +0.5% | North America, Western Europe, select Asia-Pacific urban centers | Medium term (2-4 years) |
| Tariff-Driven Reshoring of Manufacturing | +0.3% | United States with spillover to Canada and Mexico | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Rising Osteoarthritis Prevalence & Aging Demographics
Global osteoarthritis cases are forecast to top 700 million by 2030 as populations live longer and obesity rates climb.[1]World Health Organization, “Osteoarthritis,” who.int In the United States, clinically diagnosed prevalence reached 32.5 million adults in 2024, a 15% jump from 2015. Confronted with long surgical wait times, nine months for knee replacement in metropolitan Japan, surgeons increasingly deploy viscosupplementation and PRP as bridge therapies. Direct osteoarthritis care costs the United States USD 185 billion in 2024, prompting payers to consider biologics that may delay arthroplasty. As a result, the orthobiologics market is tightly linked to demographic trends that expand the candidate pool for non-operative biologic interventions.
Shift Toward Minimally-Invasive & Outpatient Orthopedic Procedures
Ambulatory surgical centers (ASCs) performed 5.2 million orthopedic cases in the United States during 2024, up 12% year on year after CMS added complex spine and joint reconstructions to its covered list.[2]Centers for Medicare & Medicaid Services, “ASC Covered Procedures List,” cms.gov ASCs value single-use PRP kits and ready-to-use allograft putties that fit fast-throughput workflows, as evidenced by Zimmer Biomet’s GPS III PRP system, which delivers a standardized 6 mL concentrate in under 15 minutes. Reimbursement for single-level lumbar fusion averages USD 18,000 in an ASC versus USD 35,000 in hospital outpatient departments, freeing budget for biologics that enhance fusion. Although Europe lags, Germany’s insurers still require hospital admission for most spine work; the outpatient shift is expected to broaden across developed regions, redirecting share within the orthobiologics market toward products optimized for ASC use.
Advances in Regenerative Biomaterials
Peptide-amphiphile nanofibers that mimic native bone extracellular matrix advanced to Phase II trials in 2025, demonstrating 30% faster radiographic union versus autograft in lumbar fusion. New demineralized bone matrix (DBM) lines now pair bone morphogenetic protein-2 at half the original dose, trimming inflammatory complications that once slowed adoption. A 2024 study showed that DBM enriched with platelet-derived growth factors achieved 92% single-level cervical fusion without donor-site morbidity. Organogenesis earned FDA Breakthrough Device designation in 2025 for a 3D-printed collagen-hydroxyapatite construct, signaling regulatory confidence in additive manufacturing for patient-specific implants.[3]Organogenesis, “Breakthrough Device Designation,” organogenesis.com Collectively, these innovations are eroding reliance on iliac-crest harvests, which carry a 10-15% complication rate.
Increasing Sports Injuries Demanding Faster Recovery Solutions
Professional athletics validate biologic efficacy: 42% of National Football League players with Grade II hamstring tears received PRP within 48 hours in 2024, reducing the median return-to-play time from 21 to 14 days. Comparable acceleration was reported for pitchers with ulnar collateral ligament sprains in Major League Baseball. Recreational adoption followed, with U.S. clinics performing about 380,000 PRP procedures in 2024, up 18% from 2023. India’s Regenerative Medicine Society formally endorsed PRP for Achilles tendinopathy in 2025, further validating demand in cash-pay markets. While outcomes vary with platelet concentration and activation protocols, the willingness of athletes and active adults to self-fund therapy sustains a robust growth pocket within the orthobiologics market.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High Therapy Cost & Patchy Reimbursement | -0.8% | Global, acute in North America and Western Europe | Medium term (2-4 years) |
| Stringent Region-Specific Regulatory Pathways | -0.6% | Europe (MDR) and fragmented Asia-Pacific frameworks | Long term (≥ 4 years) |
| Lack of Standardized Preparation Protocols | -0.4% | Global, especially affecting autologous therapies | Medium term (2-4 years) |
| Physician Skepticism & Limited Long-Term Data | -0.3% | North America, Europe, developed Asia-Pacific markets | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
High Therapy Cost & Patchy Reimbursement Coverage
Medicare pays for bone graft substitutes but excludes PRP under most CPT codes, a policy mirrored by major commercial insurers, which label autologous blood-derived products as investigational. Consequently, patients often self-fund USD 1,500-2,500 per PRP series, restricting uptake to affluent demographics. Viscosupplementation enjoys broader coverage at about USD 200 per dose, though prior-authorization hurdles tighten annually. A 2024 cost-utility study found that PRP delayed knee arthroplasty by 2.1 years and saved USD 12,000 per patient, yet payers continue to deny coverage due to the absence of 10-year results. The reimbursement stalemate slows diffusion and moderates the growth curve of the orthobiologics market.
Stringent, Region-Specific Regulatory Pathways Slowing Approvals
Europe’s Medical Device Regulation upgraded many allografts to Class III in 2021, cutting notified-body capacity almost in half and stretching approval timelines beyond 18 months. Faced with bottlenecks, firms prioritize U.S. launches, where 510(k) predicates enable clearance in roughly 6 months. Japan introduced a conditional pathway in 2024 that halves review time but adds costly five-year registry mandates. China’s fast-track office approved 12 regenerative products in 2025, yet reimbursement remains provincial, creating revenue gaps between urban and rural markets. Divergent rules force sequential rather than global rollouts, delaying returns on R&D within the orthobiologics market.
Segment Analysis
By Product Type: Viscosupplementation Dominates, PRP Gains Momentum
Viscosupplementation held a commanding 42.92% share in 2025, supported by two decades of uninterrupted Medicare reimbursement. Platelet-rich plasma is forecast to grow at a 7.09% CAGR, benefiting from point-of-care systems that deliver uniform concentrates in minutes, reducing the variability that bedeviled early centrifuge sets. Bone graft substitutes and DBM together account for about 30% of revenue, driven by more than 550,000 spinal fusion procedures performed in the United States in 2024. Stem cell therapies remain confined to trials and cash-pay clinics because of FDA enforcement against non-homologous use under Section 361.
Bone morphogenetic proteins plateaued near 8% share following black-box safety warnings, yet low-dose BMP-2 formulations that cut adverse events 60% could reignite demand. Viscosupplementation faces payer scrutiny; Anthem now requires genetic testing to confirm responders before approving multi-injection series, signaling future volume pressure. The orthobiologics market size for PRP-based products is likely to expand fastest within this category as standardized devices and elite-athlete endorsements mitigate earlier concerns.

By Application: Spinal Fusion Leads, Osteoarthritis Accelerates
Spinal fusion accounted for 52.64% of application revenue in 2025, as the procedure relies on bone graft substitutes, such as DBM and allografts, to achieve arthrodesis. Transforaminal lumbar interbody fusion, accounting for 38% of lumbar fusions, often integrates PRP to hasten osseointegration, a practice linked to 15% faster radiographic union at six months. Osteoarthritis and joint degeneration applications are projected to grow at a 9.63% CAGR as patients pursue disease-modifying interventions earlier in their care journey.
Trauma and fracture repair represent lower revenue; surgeons favor DBM and cancellous allografts to stimulate healing in nonunions. Reconstructive surgery is a steady niche where structural allografts bridge large defects in oncology and revision arthroplasty cases. As early-stage osteoarthritis patients adopt biologics to defer arthroplasty, the orthobiologics market share of degenerative-joint applications is set to widen, especially if future formulations demonstrate cartilage preservation.
By End User: Hospitals Anchor Revenue, ASCs Surge
Hospitals controlled 62.92% of 2025 revenue due to their role in complex spine and trauma cases and their capacity for in-house biologics processing. However, ASCs are forecast to grow at an 8.18% CAGR with CMS payment parity across more than 3,800 orthopedic procedures. Orthopedic and sports medicine clinics contribute about 12% of revenue, mainly through cash-pay PRP and viscosupplementation that sidestep insurance hurdles.
UnitedHealthcare's site-of-care policies now direct many single-level lumbar decompressions to ASCs, shifting tens of thousands of cases annually. Nonetheless, many ASCs lack cold-chain infrastructure for fresh-frozen allografts, an obstacle addressed by new freeze-dried DBM launches with an 18-month shelf life. Hospitals respond by scaling biologics labs for point-of-care PRP, preserving their relevance within an orthobiologics market increasingly influenced by outpatient migration.

Geography Analysis
North America generated 43.17% of global revenue in 2025, buoyed by 550,000 annual spinal fusions and the world’s largest network of in-hospital biologics labs. Section 232 tariffs accelerated reshoring, prompting processors to expand to counter 10-25% import duties on orthopedic hardware. Canada’s single-payer system restricts PRP and caps viscosupplementation to 3 injections per year, sending some patients cross-border for care. Mexico’s orthopedics sector grew 14% in 2024 as medical tourism surged, but reimbursement remains fragmented across public and private insurers.
Asia-Pacific is projected to grow at an 11.27% CAGR through 2031, propelled by China’s 12 regenerative-medicine fast-track approvals in 2025 and India’s 22% jump in orthopedic-device imports. China’s Tier 1 cities now reimburse stem cell therapies, while rural provinces lag, creating a two-speed market. India’s clinical endorsement of PRP in early 2025 legitimized private-sector offerings in a predominantly cash-pay environment. Japan’s aging population is putting pressure on capacity, extending wait times for knee replacements, and prompting the use of biologic bridge therapies. Australia harmonized its biologics rules with the FDA in 2024, slashing approval timelines to 10 months. South Korea’s National Health Insurance now covers DBM in spinal fusion, expected to lift utilization 30% over two years.
Europe represented significant revenue but faces MDR-related delays that extend product approvals beyond 18 months. Germany’s mandatory hospital admission for most spine surgery limits ASC penetration to under 8%, though pilots in Munich and Frankfurt test outpatient models. The United Kingdom’s NICE backed PRP for lateral epicondylitis in 2024, opening prospects for wider NHS adoption. France’s delisting of viscosupplementation cut its utilization 60%, spurring a shift toward PRP in private practice. GCC countries and Brazil account for the bulk of revenue in the Middle East & Africa and South America, driven by private payers and selective reimbursement for bone graft substitutes.
Competitive Landscape
The orthobiologics market is moderately concentrated; the five largest manufacturers account for a significant share of global revenue. Leading players pursue three tactics: acquiring point-of-care PRP platforms, expanding allograft catalogs, and investing in synthetic biomaterials that avoid donor variability. Stryker’s USD 340 million acquisition of a peptide-amphiphile scaffold developer in January 2025 highlights the pivot toward regenerative nanofiber platforms.
Smaller entrants emphasize data to stand out; Bioventus launched a 5,000-patient PRP outcomes registry in March 2024, giving surgeons comparative analytics that address variability concerns. Patent filings cluster around controlled-release BMP-2 and closed-loop PRP preparation; Medtronic filed seven BMP-2 delivery patents in 2024, while Arthrex automated centrifugation based on hematocrit feedback. Regulatory compliance under ISO 13485 and MDR strains small tissue banks, yet vertical integration into processing and distribution allows groups such as MTF Biologics and AlloSource to secure niche segments.
White-space opportunities include pediatric orthopedics, biologics tailored to osteoporotic bone, and scaffold-cell combination products. Organogenesis, for example, received FDA Breakthrough Device designation for a 3D-printed collagen-hydroxyapatite implant for tibial plateau fractures, with commercialization planned for late 2026. As the orthobiologics market advances, suppliers able to couple proprietary processing platforms with robust clinical data are positioned to consolidate share.
Orthobiologics Industry Leaders
BoneSupport AB
Stryker Corporation
Zimmer Biomet Holdings Inc.
Medtronic
DePuy Synthes (Johnson & Johnson)
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- May 2025: OssDsign reported that 10,000 patients had been treated with OssDsign Catalyst, its synthetic bone graft that enhances natural healing, demonstrating the product’s growing clinical adoption in the United States orthobiologics market.
- January 2025: Ventris Medical received FDA 510(k) clearance for its Backpack bone graft containment system, which includes prefilled osteoinductive and surface-activated versions for optimized bone formation.
- December 2024: The FDA approved Mesoblast’s Ryoncil (remestemcel-L-rknd), an allogeneic bone marrow mesenchymal stromal cell therapy, marking a milestone for MSC products in the United States.
- June 2024: Octane Medical Group acquired B. Braun’s global orthobiologics business, including TETEC AG and Aesculap Biologics, to strengthen its regenerative medicine platform.
Global Orthobiologics Market Report Scope
As per the report's scope, orthobiologics are used by orthopedic surgeons to help injuries heal more quickly. They accelerate the healing of broken bones and injuries to muscles, tendons, and ligaments. These products are made from substances that are naturally found in our bodies.
The Orthobiologics Market Report is Segmented by Product Type (Bone Graft Substitutes, Demineralized Bone Matrix, Allografts, Bone Morphogenetic Proteins, Viscosupplementation, Stem Cell Therapy, Platelet-Rich Plasma, Other Product Types), Application (Spinal Fusion, Trauma & Fracture Repair, Reconstructive Surgery, Osteoarthritis & Joint Degeneration, Other Applications), End User (Hospitals, Ambulatory Surgical Centers, Orthopedic & Sports Medicine Clinics, Research & Academic Institutes), and Geography (North America, Europe, Asia-Pacific, Middle East & Africa, South America). The Market Forecasts are Provided in Terms of Value (USD).
| Bone Graft Substitutes |
| Demineralized Bone Matrix |
| Allografts |
| Bone Morphogenetic Proteins |
| Viscosupplementation |
| Stem Cell Therapy |
| Platelet-Rich Plasma |
| Other Product Types |
| Spinal Fusion |
| Trauma & Fracture Repair |
| Reconstructive Surgery |
| Osteoarthritis & Joint Degeneration |
| Other Applications |
| Hospitals |
| Ambulatory Surgical Centers |
| Orthopedic & Sports Medicine Clinics |
| Research & Academic Institutes |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East & Africa | GCC |
| South Africa | |
| Rest of Middle East & Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product Type | Bone Graft Substitutes | |
| Demineralized Bone Matrix | ||
| Allografts | ||
| Bone Morphogenetic Proteins | ||
| Viscosupplementation | ||
| Stem Cell Therapy | ||
| Platelet-Rich Plasma | ||
| Other Product Types | ||
| By Application | Spinal Fusion | |
| Trauma & Fracture Repair | ||
| Reconstructive Surgery | ||
| Osteoarthritis & Joint Degeneration | ||
| Other Applications | ||
| By End User | Hospitals | |
| Ambulatory Surgical Centers | ||
| Orthopedic & Sports Medicine Clinics | ||
| Research & Academic Institutes | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East & Africa | GCC | |
| South Africa | ||
| Rest of Middle East & Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the current value of the orthobiologics market?
The orthobiologics market size reached USD 7.06 billion in 2026 and is on track to attain USD 9.52 billion by 2031.
Which product category holds the largest share of the orthobiologic market?
Viscosupplementation dominated 2025 revenue with 42.92%, driven by long-standing Medicare coverage for hyaluronic acid injections.
Which application segment is expanding fastest?
Applications for osteoarthritis and joint degeneration are advancing at a 9.63% CAGR through 2031, as patients seek biologics earlier in disease management.
How quickly are ambulatory surgical centers adopting biologics?
ASCs are projected to post an 8.18% CAGR through 2031, supported by CMS payment parity and workflow-friendly single-use biologic kits.
What geographic region is anticipated to grow most rapidly?
Asia-Pacific is forecast to register an 11.27% CAGR through 2031, reflecting accelerated regulatory approvals in China and expanding private demand in India.
Page last updated on:









