Molecular Quality Controls Market Size and Share
Molecular Quality Controls Market Analysis by Mordor Intelligence
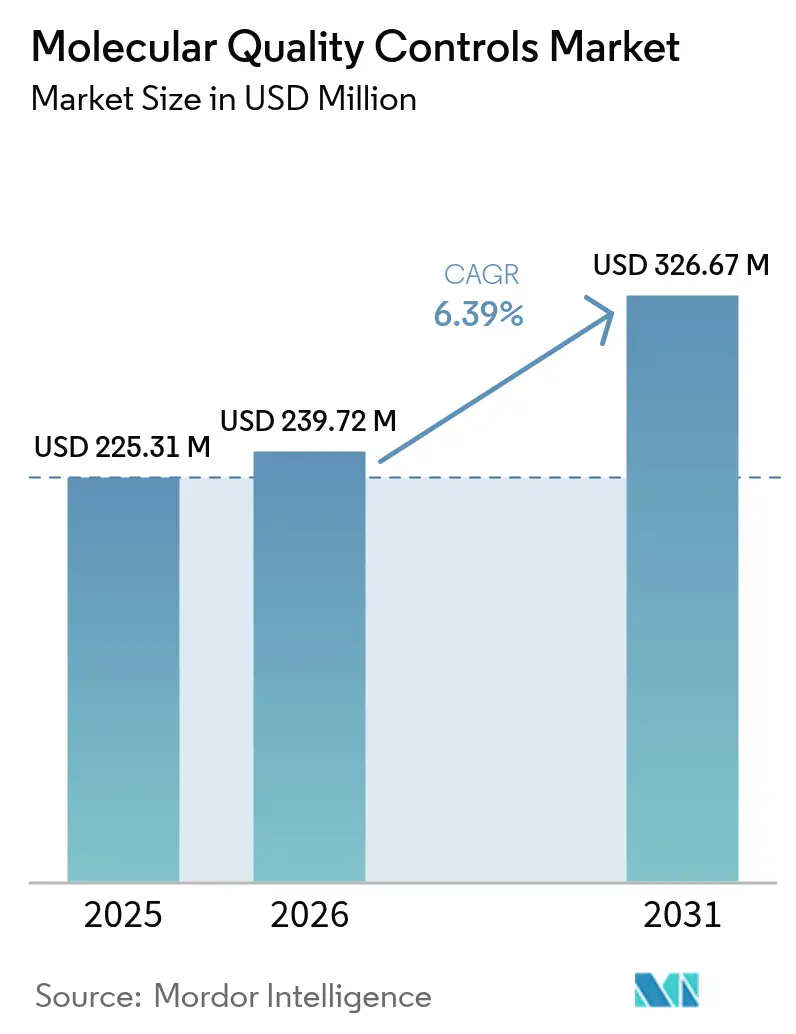
Molecular Quality Controls Market size in 2026 is estimated at USD 239.72 million, growing from 2025 value of USD 225.31 million with 2031 projections showing USD 326.67 million, growing at 6.39% CAGR over 2026-2031. Robust growth rests on three forces: the United States Food and Drug Administration’s (FDA) Laboratory Developed Tests (LDT) Final Rule, the global push for ISO 15189:2022 accreditation, and laboratories’ rapid shift from single-analyte to multiplex and next-generation sequencing (NGS) testing. Independent, third-party controls remain the default tool for demonstrating analytic accuracy, while instrument-specific controls gain momentum as laboratories integrate automation and middleware.[1]Source: U.S. Food and Drug Administration, “FDA Takes Action Aimed at Helping to Ensure the Safety and Effectiveness of Laboratory Developed Tests,” fda.gov Demand is reinforced by oncology’s expanding need for comprehensive genomic profiling, rising external-quality assessment (EQA) mandates, and the clinical move toward point-of-care molecular platforms that must still meet centralized quality standards. Conversely, high per-run control costs, supply bottlenecks for rare pathogen reference materials, and overlapping regulatory pathways temper near-term spending.
Key Report Takeaways
- By product type, independent controls led with 57.52% of molecular quality controls market share in 2025; instrument-specific controls are projected to post the fastest 7.12% CAGR through 2031.
- By technology, PCR-based controls retained 69.05% revenue share in 2025, while NGS-based controls expand at a 6.89% CAGR to 2031.
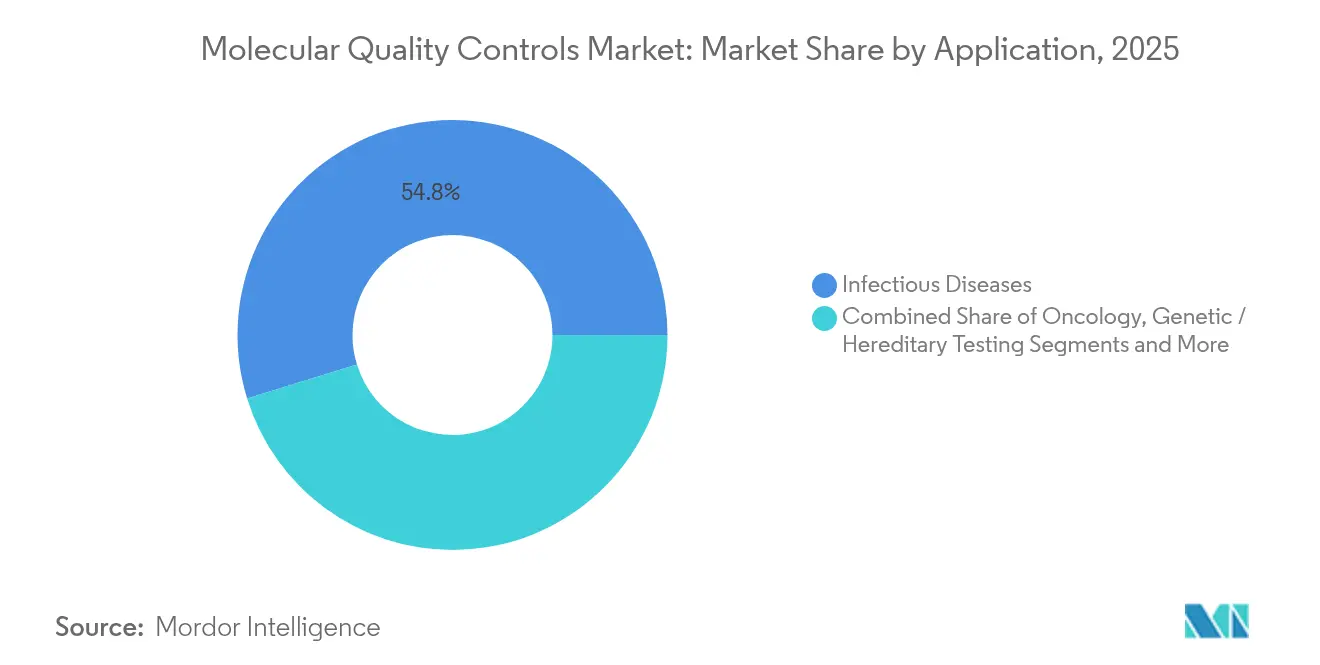
- By application, infectious diseases accounted for 54.78% of the molecular quality controls market size in 2025; oncology testing is advancing at an 7.56% CAGR through 2031.
- By end user, clinical laboratories held 45.86% of demand in 2025; IVD manufacturers and CROs record the highest 7.31% CAGR to 2031.
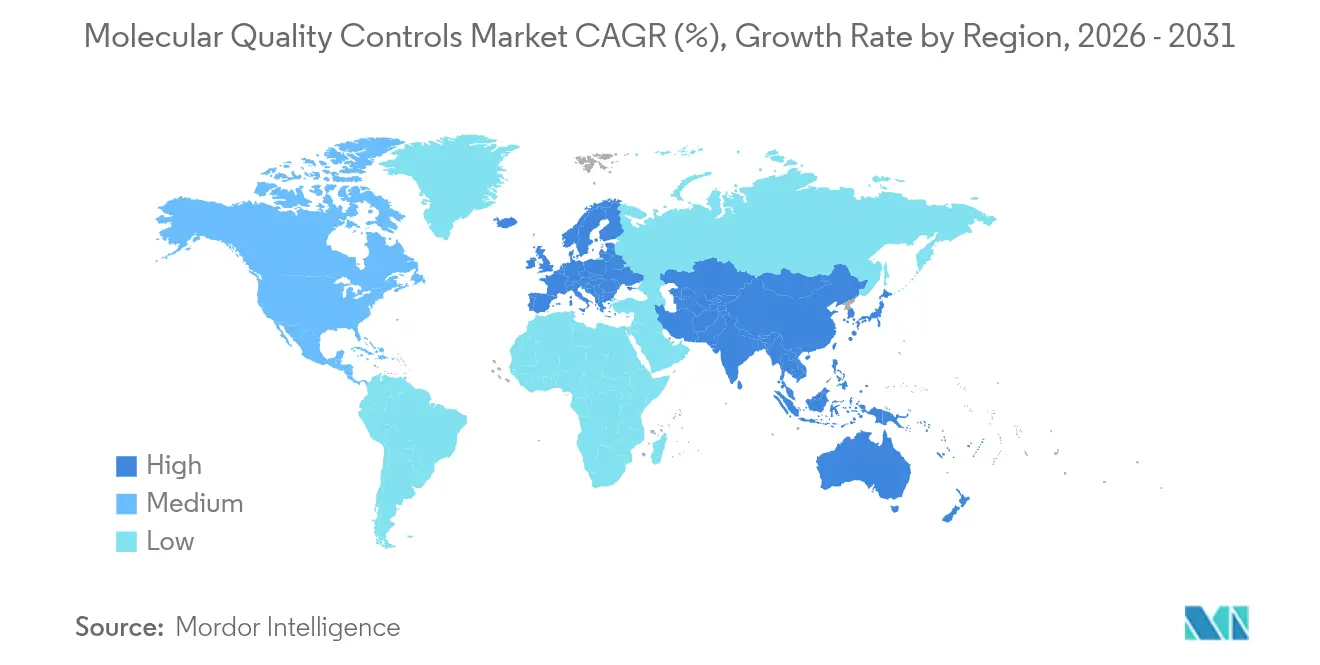
- By geography, North America captured 37.82% revenue in 2025; Asia-Pacific is the fastest-growing region at an 7.74% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Molecular Quality Controls Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising Test-Volume in Molecular Diagnostics | +1.8% | Global, with concentration in North America & Europe | Medium term (2-4 years) |
| Increasing Adoption of 3rd-Party QC For ISO 15189 Accreditation | +1.5% | Global, particularly emerging markets in APAC | Long term (≥ 4 years) |
| Growing Incidence of Cancer and Genetic Disorders | +1.2% | Global, with higher impact in developed regions | Long term (≥ 4 years) |
| Stricter External-Quality Assessment (EQA) Mandates | +1.0% | North America & EU, expanding to APAC | Short term (≤ 2 years) |
| Shift To Digital Multiplex QC Panels | +0.8% | North America & Europe, early adoption in APAC | Medium term (2-4 years) |
| Consolidation of Labs Driving Enterprise QC Data-Integration | +0.5% | North America & Europe | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Rising Test-Volume in Molecular Diagnostics
Diagnostic laboratories processed unprecedented volumes during the COVID-19 public-health emergency, with the FDA authorizing 291 molecular assays, demonstrating the scalability of high-throughput platforms.[2]Source: U.S. Department of Health and Human Services, “Testing & Diagnostics,” hhs.gov Post-pandemic, volume remains elevated as labs extend molecular testing to pharmacogenomics, antimicrobial resistance surveillance, and hereditary cancer panels. Each multiplex target adds validation layers, compelling laboratories to rely on robust third-party controls to avoid analytical drift. Digital QC dashboards integrated with laboratory information systems have trimmed manual verification steps by 62.5%, underscoring technology’s role in managing rising workloads.
Increasing Adoption of Third-Party QC for ISO 15189 Accreditation
ISO 15189:2022 raises the bar for risk management and for point-of-care integration, pushing laboratories toward externally sourced controls that demonstrate traceability and independence. The first U.S. accreditation under the new version signaled an early inflection toward global compliance momentum. Laboratories have three years to transition, anchoring sustained demand for molecular quality controls market products.
Growing Incidence of Cancer and Genetic Disorders
Cancer incidence rates continue rising globally, with molecular testing becoming essential for precision oncology treatment decisions. NGS oncology panels interrogate hundreds of genes, copy-number events, and fusions, each demanding stringent controls for variant detection accuracy. Liquid-biopsy assays amplify complexity by requiring ultra-low-frequency variant detection, escalating the need for high-sensitivity synthetic controls. Similarly, hereditary disease panels covering 100 plus actionable genes require variant-specific controls to support correct clinical reporting.
Stricter External-Quality Assessment Mandates
The 2024 CLIA update introduced 23 new molecular proficiency tests and narrowed allowable error margins by 33.3%, compelling U.S. laboratories to upgrade QC procedures. The longitudinal analysis of 20 years of EQA schemes for PCR/NAAT-based bacterial detection reveals declining in-house assay usage and increased adoption of commercial quality controls. Parallel schemes in Europe under IVDR and ISO 15189 require regular proficiency participation, cementing a compliance-driven spending channel within the molecular quality controls market.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High Per-Run Cost of Molecular QC Materials | -1.2% | Global, particularly cost-sensitive emerging markets | Short term (≤ 2 years) |
| Complex Multi-Agency Regulatory Pathway | -0.8% | North America & Europe, expanding globally | Medium term (2-4 years) |
| Unfavourable Reimbursement for Confirmatory Molecular Tests | -0.6% | North America & Europe | Long term (≥ 4 years) |
| Supply-Chain Fragility for Rare Pathogen Reference Materials | -0.4% | Global, with acute impact during outbreaks | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
High Per-Run Cost of Molecular QC Materials
The economics of molecular quality controls present significant challenges for laboratories operating under constrained budgets, particularly as test complexity increases. NGS assays cut the overall cost of patient care compared with sequential PCR yet still demand expensive multi-analyte controls that can account for 4–7% of per-sample cost, a margin non-trivial to small laboratories. Fixed control costs scale poorly when test volumes are modest, prompting labs to stretch replacement intervals and potentially compromise analytical robustness.
Complex Multi-Agency Regulatory Pathway
The regulatory landscape for molecular quality controls involves multiple agencies with overlapping jurisdictions, creating compliance complexity that extends product development timelines and increases costs. The FDA’s four-year phase-in of the LDT Final Rule requires device-like quality systems, medical-device reporting, and in many cases premarket review.[3]Source: U.S. Food and Drug Administration, “Medical Devices; Laboratory Developed Tests,” fda.gov Parallel IVDR conformity assessments in Europe oblige manufacturers to address multiple audit bodies, lengthening product launches and raising compliance costs, especially for small suppliers.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Product Type: Independent Controls Drive Market Leadership
Independent controls dominated with a 57.52% molecular quality controls market share in 2025, reflecting laboratories’ preference for vendor-neutral verification tools that satisfy ISO 15189 documentation requirements and mitigate platform bias. Bio-Rad’s Unity Data Management network, active in 38,000 labs, illustrates how third-party controls aggregate peer comparisons to detect systemic deviations quickly. Independent products span multi-analyte panels for respiratory pathogens to custom oncology variants, allowing labs to standardize across diverse instruments.
Instrument-specific controls, though smaller, are projected to post a 7.12% CAGR through 2031 as automation and integrated sample-to-answer platforms expand. Manufacturer-tuned stability and lot-to-lot consistency save validation time, a decisive advantage in high-throughput environments. Yet vendor-lock fears linger, keeping independent controls the reference option for proficiency schemes. The molecular quality controls market therefore gravitates toward a dual-sourcing model in which labs deploy independent controls for accreditation while relying on instrument-specific materials for daily workflow continuity.


By Technology: PCR Dominance Faces NGS Disruption
PCR-based products retained 69.05% revenue in 2025, anchored by high-volume infectious disease testing where turnaround time and cost trump breadth. These controls typically contain stabilized viral or bacterial nucleic acids encapsulated in non-infectious particles for biosafety.
NGS-based controls, however, are advancing at a 6.89% CAGR, reflecting oncology’s shift toward multi-gene panels and the rising use of comprehensive genomic profiling in hereditary disorders. Sample-preparation QC kits assess library complexity, fragment size, and adapter ligation efficiency before sequencing, reducing costly reruns. The molecular quality controls market size for NGS panels is poised to expand as reimbursement improves and sequencing costs continue to decline. Isothermal amplification controls occupy niche use cases such as point-of-care STI testing, where rapid qualitative answers suffice.
By Application: Infectious Diseases Leadership Under Oncology Pressure
Infectious-disease testing held 54.78% of the molecular quality controls market size in 2025, supported by continuous respiratory virus surveillance and mandated EQA participation for notifiable pathogens. Multi-target panels covering influenza, RSV, and SARS-CoV-2 remain baseline requirements for clinical labs.
Oncology, forecast to grow at an 7.56% CAGR, is narrowing the gap. Liquid-biopsy innovation and adaptive cancer trials necessitate quantitative controls capable of detecting variants at allele frequencies below 0.5%. Laboratories also bundle QC data into electronic submission packages for U.S. FDA companion-diagnostic approvals, a procedural reality lifting control volumes per assay. Genetic and hereditary disease testing gains momentum through expanded carrier screens and pharmacogenomic guidance, propelling broader adoption of multiplex controls with calibrated variant distributions.

By End-User: IVD Manufacturers Accelerate Enterprise Integration
Clinical laboratories commanded 45.86% revenue in 2025, yet purchasing dynamics increasingly shift upstream. IVD manufacturers and CROs, growing at 7.31% CAGR, bundle controls into assay development pipelines to streamline FDA or CE marking dossiers. Control consumption spikes during validation, bridging analytical sensitivity, specificity, and limit-of-detection studies.
Enterprise consolidation also moves QC selection to central corporate teams, prioritizing platforms offering data-integration APIs and automated lot-tracking. For hospital networks, middleware linking QC metrics with inventory data curtails wastage, giving suppliers that provide cloud dashboards a competitive edge. Academic medical centers remain important adopters, particularly where translational research creates novel assay designs demanding bespoke QC materials.
Geography Analysis
North America led with 37.82% revenue in 2025, buoyed by strong reimbursement, high test volume, and the FDA’s framework that elevates third-party controls from best practice to regulatory necessity. CLIA’s stricter performance thresholds compound demand as laboratories widen QC frequency to retain accreditation. Canada’s modernization of medical-device regulations supports accelerated pathways for innovative quality controls, sustaining steady regional growth.
Asia-Pacific is the fastest-growing geography with an 7.74% CAGR, propelled by government genomics programs and expanding private diagnostic chains. Japan’s reimbursement of NGS oncology panels and South Korea’s investment in cell-and-gene therapy manufacturing both translate to higher QC consumption for NGS workflows. China’s domestic instrument makers increasingly embed QC lot-tracking software, amplifying local demand. Despite fragmented regulations, the molecular quality controls market benefits from APAC’s push to harmonize quality standards with ISO 15189 and IVDR principles, fostering cross-border product adoption.
Europe exhibits consistent mid-single-digit growth as IVDR implementation compels laboratories and manufacturers to upgrade quality documentation. The United Kingdom’s National Health Service awards central laboratory contracts that require ISO 15189:2022 compliance, embedding QC use in procurement templates. Middle East & Africa and South America remain nascent but show double-digit incremental gains where new reference laboratories open. In these regions, infectious-disease surveillance projects funded by multilateral agencies often stipulate third-party controls, giving suppliers an early foothold.
Competitive Landscape
The molecular quality controls market is moderately concentrated. Bio-Rad, Roche, and Thermo Fisher anchor the top tier, leveraging broad assay portfolios and informatics layers to create high switching costs. Bio-Rad’s pending purchase of Stilla Technologies will blend digital PCR expertise with its independent control catalogue, expanding quantitative options for gene-therapy vector assays. Roche embeds sample-quality metrics into its NGS sample-prep kits, aligning control materials with workflow automation.
Mid-sized specialists such as ZeptoMetrix and Microbiologics focus on niche pathogen lines, often launching reference materials within weeks of emerging threats, a capability valued during outbreaks. Barriers to entry sit chiefly in regulatory proficiency and bio-manufacturing scale; newcomers typically partner with contract manufacturers to bridge gaps. Competitive differentiation is increasingly data-centric: platforms that stream QC results to cloud dashboards offer near-real-time performance analytics, a feature laboratories use to shorten troubleshooting windows from days to hours.
Opportunities abound in point-of-care and decentralised testing, where lateral-flow and microfluidic systems still lack robust third-party controls. Suppliers able to miniaturize controls for cartridge formats stand to capture incremental volume. Likewise, the rise of AI-powered quality-management software generates pull-through for control providers offering machine-readable certificates and barcode integration, reinforcing ecosystem lock-in beyond mere reagent sales.
Molecular Quality Controls Industry Leaders
Bio-Rad Laboratories Inc
F. Hoffmann-La Roche AG
ZeptoMetrix Corporation
bioMérieux SA
Thermo Fisher Scientific Inc.
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- June 2025: ZeptoMetrix launched NATtrol Influenza A H5N1 Quantitative Stock to strengthen avian-influenza assay validation workflows.
- November 2024: Microbiologics acquired SensID, expanding its oncology and precision-medicine control portfolio.
Global Molecular Quality Controls Market Report Scope
Molecular quality controls are used to evaluate the performance of molecular tests such as in vitro nucleic acid testing procedures for viral load assays, pathogen detection, leukocyte reduced apheresis platelet (LRAP) units, healthcare-associated infections (HAIs) like pneumonia, urinary tract infections and more. These products majorly focus on the detection of defects, quality control, and validation panels to support in the implementation and monitoring assay-kit lot performance for clinical and research laboratories, blood diagnostic centers, and IVD manufacturers.
| Independent Controls |
| Instrument-specific Controls |
| PCR-based |
| NGS-based |
| Isothermal/Other NAAT |
| Infectious Diseases |
| Oncology |
| Genetic / Hereditary Testing |
| Reproductive and Prenatal Health |
| Others |
| Clinical Laboratories |
| Hospitals and Academic Medical Centres |
| IVD Manufacturers and CROs |
| Others |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America | |
| South Africa |
| By Product Type | Independent Controls | |
| Instrument-specific Controls | ||
| By Technology | PCR-based | |
| NGS-based | ||
| Isothermal/Other NAAT | ||
| By Application | Infectious Diseases | |
| Oncology | ||
| Genetic / Hereditary Testing | ||
| Reproductive and Prenatal Health | ||
| Others | ||
| By End-user | Clinical Laboratories | |
| Hospitals and Academic Medical Centres | ||
| IVD Manufacturers and CROs | ||
| Others | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||
| South Africa | ||


Key Questions Answered in the Report
Why is the molecular quality controls market growing faster after 2026?
Demand accelerates as the FDA’s LDT Final Rule tightens quality-system requirements, compelling laboratories worldwide to adopt third-party controls and driving a 6.39% CAGR through 2031.
Which product category holds the largest molecular quality controls market share today?
Independent, vendor-neutral controls dominate with 57.52% share because they satisfy ISO 15189 documentation and cross-platform comparability needs.
How quickly are NGS-based quality controls expanding?
NGS-based controls are projected to rise at a 6.89% CAGR, reflecting oncology’s shift toward comprehensive genomic profiling and multi-gene hereditary panels.
Which region will contribute the most incremental revenue by 2031?
Asia-Pacific, poised for an 7.74% CAGR, will add the largest new revenue due to genomic-medicine initiatives and rising laboratory infrastructure investments.
What are the main cost pressures facing laboratories?
High per-run QC material costs and supply constraints for rare pathogen references can raise operational expenses by up to 7% of assay cost, especially in low-volume settings.
Page last updated on:









