Microplate Systems Market Size and Share
Microplate Systems Market Analysis by Mordor Intelligence
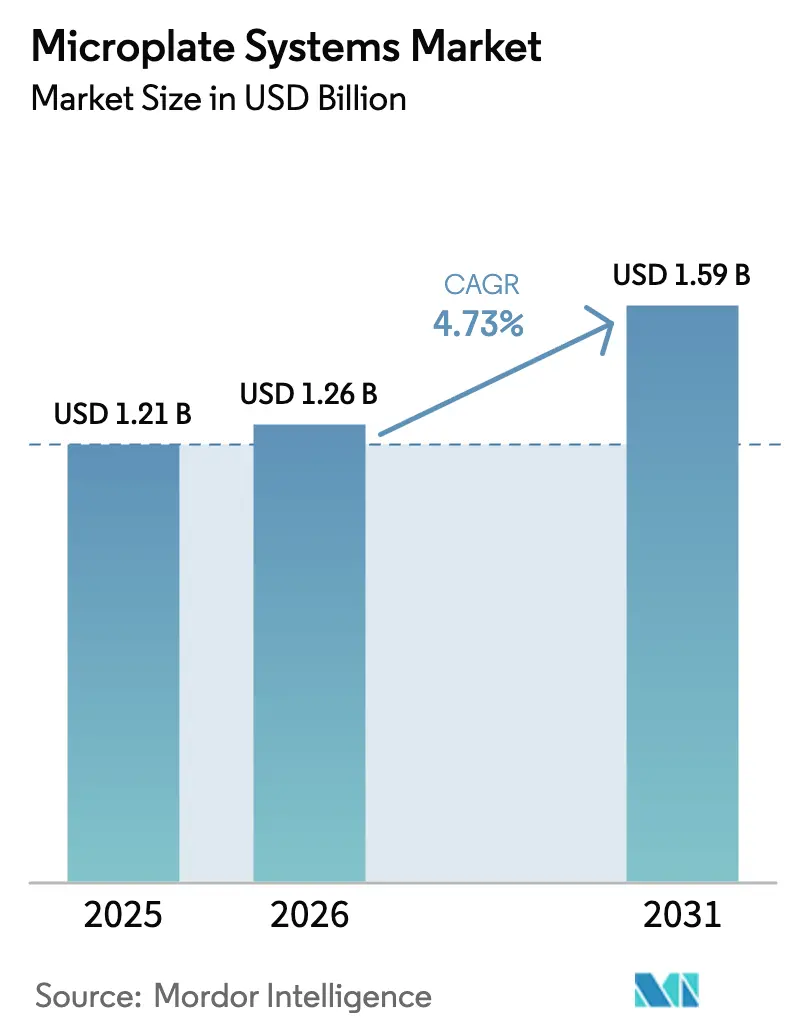
The Microplate Systems Market size is projected to expand from USD 1.21 billion in 2025 and USD 1.26 billion in 2026 to USD 1.59 billion by 2031, registering a CAGR of 4.73% between 2026 to 2031.
Sustained investment in high-throughput screening, demand for hybrid detection optics, and stronger public-sector funding underpin this steady climb. Laboratories are shifting from single-mode absorbance readers toward multi-mode platforms that integrate fluorescence, luminescence, and imaging, a pivot that compresses capital budgets while widening assay menus. Pharmaceutical companies favor these hybrid systems because they enable legacy ELISA workflows and next-generation phenotypic screens without hardware swaps, reducing validation overhead. Sovereign initiatives in precision medicine and the broader adoption of automated liquid handling are amplifying growth in Asia-Pacific, where sovereign funding is accelerating the installation of multi-mode readers. At the same time, sustainability mandates in the European Union and California push vendors to disclose energy-per-read metrics and offer take-back schemes for consumables, tilting procurement toward greener models.
Key Report Takeaways
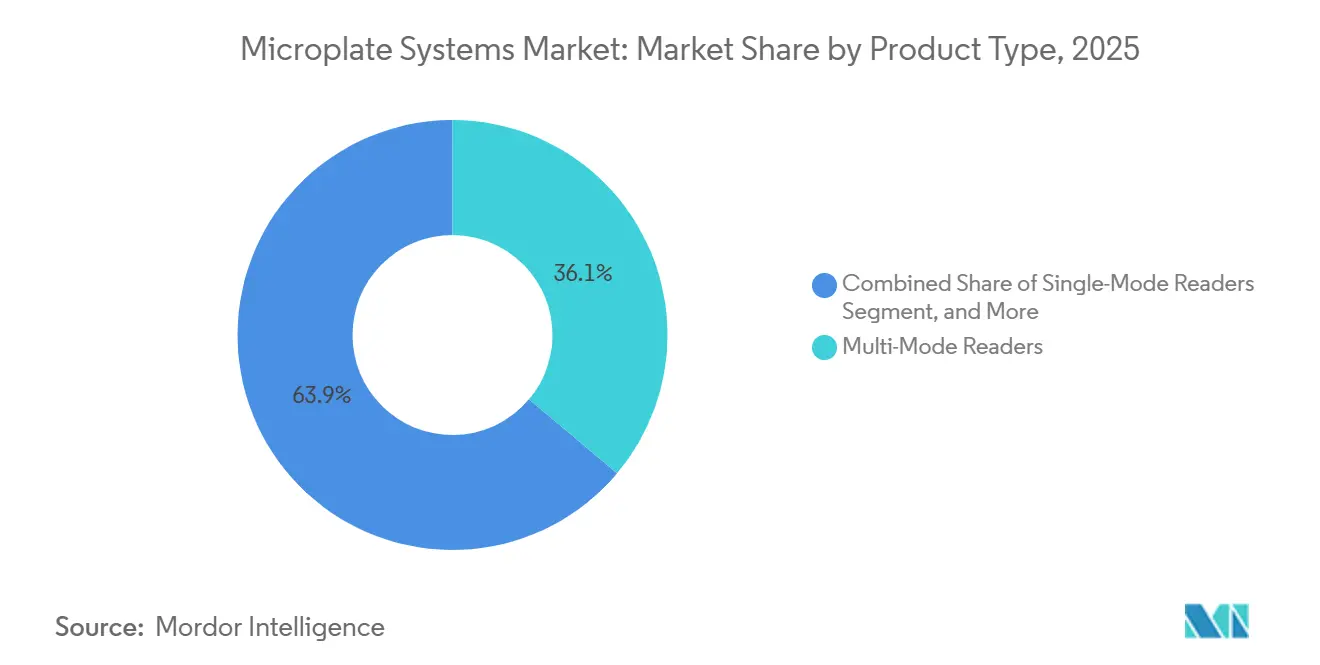
- By product type, multi-mode readers led with 36.12% of the microplate systems market share in 2025 and are advancing at a 6.06% CAGR through 2031.
- By application, drug discovery and high-throughput screening captured 47.09% of the microplate systems market size in 2025, while genomics and proteomics research is projected to expand at a 7.63% CAGR during 2026-2031.
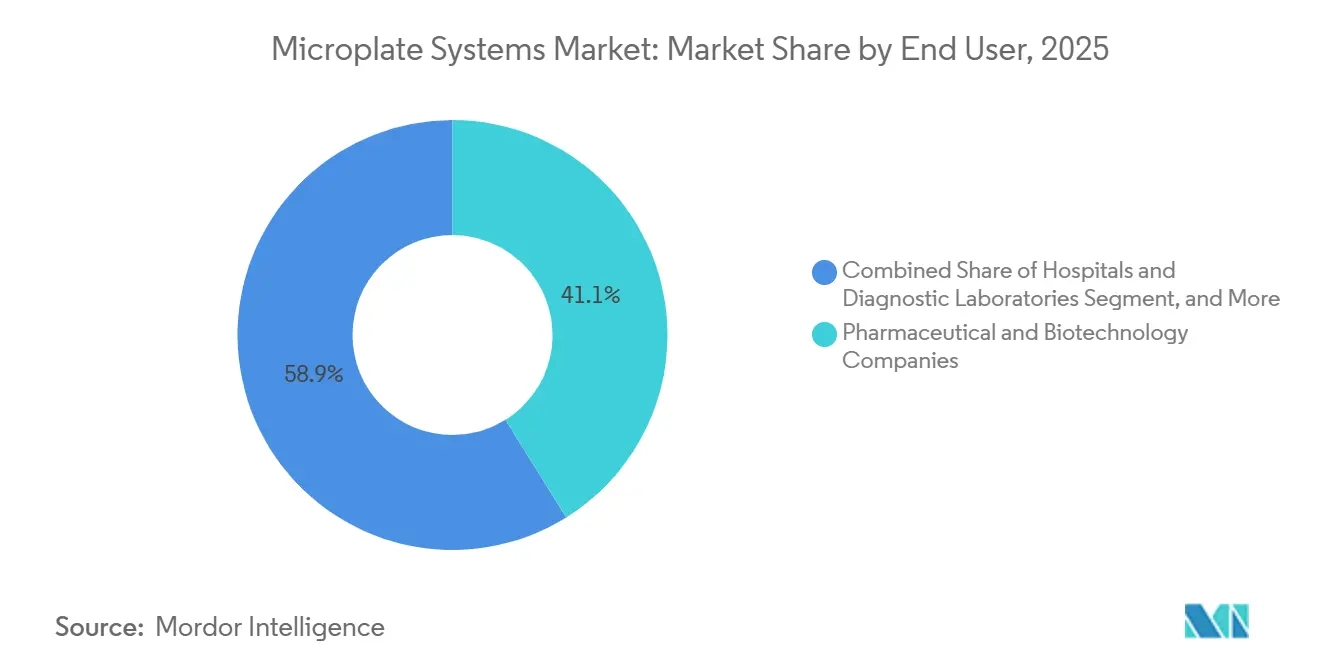
- By end user, pharmaceutical and biotechnology companies held 41.13% share of end-user spending in 2025; contract research and manufacturing organizations posted the fastest growth at 8.18% CAGR through 2031.
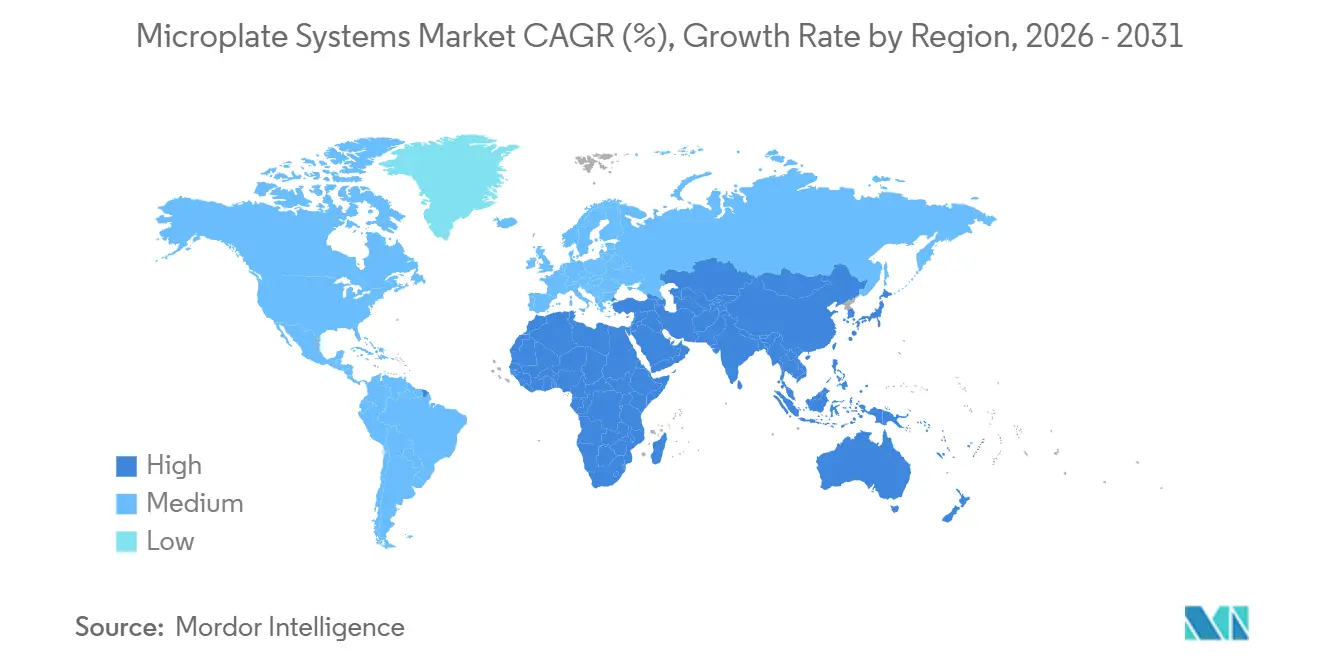
- By geography, North America commanded 38.29% 2025 sales, whereas Asia-Pacific is projected to post the strongest regional CAGR of 10.13% through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Microplate Systems Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising Adoption of High-Throughput Screening | +1.2% | North America, Europe, Asia-Pacific | Medium term (2-4 years) |
| Growing Demand for Automation & Multi-Mode Detection | +1.0% | Global, strongest in Asia-Pacific | Short term (≤2 years) |
| Expansion of Life-Sciences Research Funding | +0.8% | North America, Europe, China, India | Long term (≥4 years) |
| Technological Advances in Sensitivity & Throughput | +0.6% | Global | Medium term (2-4 years) |
| Emergence of AI-Integrated Real-Time Analytics | +0.7% | North America, Europe, early Asia-Pacific | Short term (≤2 years) |
| Sustainability Push for Energy-Efficient Instruments | +0.3% | European Union, California, Massachusetts | Long term (≥4 years) |
| Source: Mordor Intelligence | |||
Rising Adoption of High-Throughput Screening in Drug Discovery
Pharmaceutical pipelines continue to migrate toward complex modalities such as antibody-drug conjugates and cell therapies that demand phenotypic assays screening tens of thousands of compounds per campaign.[1]Nature Methods, “Advances in High-Throughput Screening,” nature.com Modern high-throughput platforms now process more than 100,000 data points per day by integrating robotic plate handlers, tight environmental control, and hybrid optics that switch between fluorescence polarization and time-resolved luminescence without manual changeover. This consolidation lets a single reader displace multiple single-mode devices, freeing bench space and reducing validation overhead. Biopharma R&D spending rose above USD 145 billion in 2025, intensifying pressure to improve productivity. Automation answers that call by allowing chemists to interrogate larger libraries with fewer full-time employees. Oncology and immunology programs show the greatest uptake because they rely on multiplexed cytokine profiling and live-cell imaging across 384- and 1536-well formats.
Growing Demand for Automation & Multi-Mode Detection in Diagnostics
Clinical laboratories now link sample reception, centrifugation, aliquoting, and microplate immunoassays into a single conveyor workflow, cutting manual touchpoints by more than 70% and reducing turnaround time for routine panels from 4 hours to under 90 minutes.[2]Academic OUP, “Total Laboratory Automation,” academic.oup.com Multi-mode readers fit seamlessly because they switch among ELISA, chemiluminescence, and fluorescence assays without operator intervention. Molecular Devices embedded AI-driven quality-control algorithms in its SpectraMax iD5e and iD3s models, released in July 2025, lowering repeat runs by an estimated 15%. Hospitals that reach 98% instrument availability can defer capital replacement by up to two years, a vital cash-flow advantage for regional networks. Demand is strongest in Asia-Pacific, where universal health coverage programs and electronic health record mandates require machine-readable assay data. Compliance frameworks such as ISO 15189 now stress audit trails, prompting vendors to add HL7 and FHIR connectivity.
Expansion of Life-Sciences Research Funding
Multi-year grants for precision-medicine programs funnel capital into proteomics core facilities that depend on microplate readers for ELISA, Luminex bead arrays, and AlphaLISA proximity assays. Genome Canada committed CAD 52 million (USD 38 million) in 2024 to shared-equipment purchases.[3]Genome Canada, “Research Funding Announcements,” genomecanada.ca India’s biotechnology roadmap targets sector expansion to USD 300 billion by 2030, with 30% directed to research infrastructure. Public funding often mandates open-access data, favoring cloud-connected readers that auto-upload to FAIR-compliant repositories. China’s biotechnology investment exceeded USD 20 billion in 2024, with procurement concentrated in the biotech parks in Shenzhen, Shanghai, and Beijing. These inflows smooth demand cycles because grant-funded equipment purchases are insulated from quarterly budget swings in private industry.
Technological Advances in Sensitivity & Throughput
Hybrid filter-monochromator optics, next-generation photomultiplier tubes, and improved thermal management tighten detection limits and shorten read times. Tecan’s Spark Cyto integrates confocal imaging that quantifies 3D spheroid growth in real time, reducing assay development time for oncology drug screening. Agilent’s BioTek Cytation combines environmental control with brightfield imaging, shrinking the workstation footprint from 6 m² to under 3 m². Piezoelectric and acoustic dispensing reach sub-100 nL precision, enabling 1536- and 3456-well formats that save reagents. Laboratories investing in these advances report up to 40% faster run times and 20% lower per-sample consumable costs, enhancing the appeal of microplate systems to lean operations.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High Capital & Maintenance Costs of Advanced Systems | -0.9% | Global, pronounced in emerging markets | Short term (≤2 years) |
| Shortage of Skilled Technicians | -0.6% | North America, Europe, Asia-Pacific | Medium term (2-4 years) |
| Semiconductor & Optical Component Volatility | -0.4% | Global | Short term (≤2 years) |
| Competition From Lab-On-Chip Microfluidics | -0.3% | North America, Europe | Long term (≥4 years) |
| Source: Mordor Intelligence | |||
High Capital & Maintenance Costs of Advanced Systems
Multi-mode readers with integrated incubators, robotic stackers, and AI modules range from USD 80,000 to USD 200,000, stretching budgets for academic cores and early-stage biotech firms. Annual service contracts add an additional 8-12% to the purchase price, covering photomultiplier replacement and software updates. Import duties in several middle-income nations inflate landed costs by up to 35%, delaying adoption. Leasing and reagent-rental models from PerkinElmer and Tecan convert capital outlay into operating expense, but multi-year minimums expose laboratories to penalty clauses if projects stall. Cost pressure sometimes steers buyers toward lower-spec single-mode devices that can become obsolete once research shifts to multiplexed assays.
Shortage of Skilled Technicians
Operating advanced microplate systems requires expertise in liquid-handling robotics and data-integrity protocols, skills that 41% of employers report as critically scarce. University curricula lag, so new hires often need six months of training on automation software such as Tecan FluentControl. Under-staffed labs leave USD 150,000 instruments idle up to 40% of the time, hurting return on investment. CROs now run internal academies to cross-train staff, but attrition remains high as pharma firms offer 30% salary premiums after two years’ experience. Vendors respond by embedding wizards that auto-suggest gain settings and flag protocol deviations, lowering the learning curve.
Segment Analysis
By Product Type: Multi-Mode Readers Drive Consolidation
Multi-mode readers accounted for 36.12% of 2025 revenue and are projected to grow at 6.06% through 2031, the fastest rate among product categories, as laboratories replace single-mode absorbance units with hybrid platforms that integrate filter wheels and monochromators in a single chassis. This segment’s expansion lifts the microplate systems market size because each unit commands premium pricing and pulls through ancillary software and service contracts. Single-mode readers survive in high-volume ELISA labs where cost per test rules, yet their share erodes as automated conveyor architectures need instruments that run chemiluminescence and fluorescence without downtime. Pipetting and dispensing systems gain relevance as 1536-well and 3456-well formats spread, but their uptake depends on precision at sub-microliter volumes.
Tecan’s Spark and Agilent’s Cytation illustrate the trend toward convergence, folding detection, environment control, and imaging into footprints under 3 m². BMG LABTECH’s VANTAstar and Molecular Devices’ iD5e embed AI quality control, cutting repeat runs by 15%. Pipettor vendors focus on acoustic or piezoelectric dispensing to achieve ≤1 nL precision, a must for ultra-high-density plates. Microplate washers remain essential for ELISA workflows but often sell bundled with readers, reflecting buyer preference for single-vendor support contracts. Handlers and incubators round out the ecosystem; open-source designs such as G-Bot lower entry costs but challenge incumbent profit pools.

By Application: Genomics and Proteomics Outpace Legacy Screening
Drug discovery and high-throughput screening accounted for 47.09% of revenue in 2025, underscoring demand for phenotypic assays in oncology programs. However, genomics and proteomics research is the fastest-growing application at 7.63% CAGR, boosted by national precision-medicine grants that require multiplexed biomarker quantification. This dynamic diversifies the microplate systems market; even if pharma R&D stalls, public-funded omics projects sustain unit orders. Clinical diagnostics provides a steady baseline, with hospital labs adopting total automation to absorb seasonal surges in sample volume.
Cell-based assays and toxicity testing are gaining momentum as regulatory encouragement of in vitro alternatives drives the need for kinetic reads over 72 hours in incubated multi-mode readers. Environmental and food-safety applications, though smaller, are moving toward stricter residue limits; ELISA screening on plates remains the cost-effective first filter before confirmatory mass spectrometry. Vendors tailor firmware to handle long kinetic runs and integrate with LIMS for traceability. Feature roadmaps now include flexible scheduling that interleaves fast endpoint assays with multi-day cell viability studies, maximizing reader utilization.
By End User: CROs and CMOs Accelerate Faster Than Pharma
Pharmaceutical and biotechnology firms accounted for 41.13% of spending in 2025, yet contract research and manufacturing organizations grew faster at 8.18% through 2031 as outsourcing intensified. CROs buy automation-ready stacks with SiLA 2 interfaces that meet audit and data-integrity mandates, driving the microplate systems market share across service providers. Hospitals and diagnostic laboratories install multi-mode readers to shrink assay menus onto fewer devices amid reimbursement pressure.
Academic and research institutes rely on multi-year capital cycles funded by national science agencies, smoothing demand volatility. They emphasize open-access scheduling and cloud repositories, rewarding vendors that bundle no-code assay builders. Regional suppliers like Shenzhen Mindray and Rayto Life Sciences penetrate price-sensitive hospital segments with systems priced 30-40% below Western peers and backed by 24-hour service networks. Sales strategies must therefore segment by value driver: uptime for CROs, flexibility for pharma, cost for hospitals, and openness for academia.

Geography Analysis
North America retained a 38.29% share in 2025, fueled by biotech clusters in Boston and San Francisco, which together raised more than USD 30 billion in venture funding in 2024. Large CROs such as Charles River and IQVIA operate fleets of more than 500 readers each, ensuring consistent replacement cycles. FDA’s predictable 510(k) and Computer Software Assurance frameworks lower regulatory risk, speeding adoption of AI-enabled models. Canada contributes via Genome Canada grants that direct funds to precision-medicine cores, while Mexico’s demand is nascent but growing as private labs in Mexico City shift from manual ELISA to semi-automatic platforms.
Asia-Pacific is the fastest-growing region, with a 10.13% CAGR to 2031, double the global pace. China’s state funding of above USD 20 billion in 2024 and India’s roadmap to a USD 300 billion biotechnology value by 2030 supply robust capital for laboratory infrastructure. NMPA’s accelerated approvals enable domestic firms like Mindray to capture hospital share through localized service. Japan’s aging demographics drive laboratory automation, though lengthy consensus procurement slows cycles. Australia and South Korea maintain steady demand tied to genomics and biosecurity projects at leading universities.
Europe’s market reflects the revised in vitro diagnostic regulation, which increases data and environmental reporting requirements. Germany, the United Kingdom, and France dominate demand, blending pharmaceutical giants with academic heavyweights such as Max Planck Institutes and the Francis Crick Institute. Sustainability rules push buyers toward ACT-labeled instruments and recyclable consumables. The Middle East and Africa, though smaller, grow as Gulf states build biotech hubs; King Abdullah University and Dubai’s Mohammed bin Rashid University both installed multi-mode readers in 2025. South America sees Brazil leading, but high tariffs and currency swings temper growth. Collectively, these dynamics shape a geographically diversified microplate systems market with region-specific catalysts.
Competitive Landscape
The top five vendors, Danaher (Molecular Devices), Agilent Technologies, Revvity, Tecan Group, and Bio-Rad Laboratories, command significant global revenue, pointing to moderate concentration. These incumbents lock in customers through installed-base ecosystems that bundle readers, washers, and software, thereby raising switching costs associated with assay revalidation and staff retraining. Competition pivots on software and cloud services; vendors offering FAIR-compliant auto-upload and no-code assay builders lower adoption hurdles for resource-constrained labs.
Disruptors emerge from open-source automation consortia like Advanced Cell Culture System, which publish hardware blueprints that labs assemble for under USD 10,000, eroding ancillary revenue streams. Regional champions such as Shenzhen Mindray, Rayto Life Sciences, and Shimadzu win share in Asia-Pacific by pairing CE-mark or NMPA-cleared devices with local-language interfaces and rapid on-site service. Technology roadmaps now highlight AI algorithms for real-time QC, confocal imaging for 3D spheroids, and SiLA 2 compatibility, features that command 15-20% premiums because they compress assay development cycles.
Strategic moves continue to reshape the field. Ingersoll Rand purchased Scinomix in January 2026, adding plate handlers that integrate via SiLA 2 and broadening its precision portfolio. Thermo Fisher’s USD 4.1 billion acquisition of Solventum’s purification unit in February 2025 further tightens its grip on upstream workflows that feed microplate assays. Agilent’s 2024 takeover of BIOVECTRA creates a captive customer base for its BioTek readers. Such vertical integrations intensify service bundling and could raise entry barriers for smaller hardware-only entrants.
Microplate Systems Industry Leaders
Bio-Rad Laboratories, Inc.
Agilent Technologies, Inc.
Danaher Corporation
Shenzhen Mindray Bio-Medical Electronics Co., Ltd
Thermo Fisher Scientific Inc.
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- May 2025: Agilent Technologies reported Q2 2025 revenue of USD 1.67 billion, representing 6% year-over-year growth, with the Life Sciences and Diagnostics Markets Group generating USD 654 million and 8% growth, demonstrating strong market demand for analytical instrumentation and laboratory automation solutions.
- March 2025: Thermo Fisher Scientific launched the Vulcan Automated Lab, an integrated system combining robotic handling with AI-enhanced instruments to streamline transmission electron microscopy workflows for semiconductor manufacturing, addressing the demand for atomic-scale analysis capabilities.
- January 2025: ABB Robotics and Agilent Technologies announced a collaboration to develop automated laboratory solutions combining Agilent's analytical instrumentation with ABB's robotics technology, aimed at improving workflows and efficiency in pharmaceutical and biotechnology sectors.
- January 2025: ABB Robotics and Agilent Technologies announced a collaboration to develop automated laboratory solutions combining Agilent's analytical instrumentation with ABB's robotics technology, aimed at improving workflows and efficiency in pharmaceutical and biotechnology sectors.
Research Methodology Framework and Report Scope
Market Definitions and Key Coverage
Our study defines the global microplate systems market as the total revenue generated by single-mode and multi-mode plate readers, washers, dispensers, stackers, and the bundled control software that process 96- to 1536-well plates for biochemical, genomic, and cell-based assays.
Scope Exclusion: Reagents, disposable plates, and liquid handlers that lack an integrated plate deck sit outside this assessment.
Segmentation Overview
- By Product Type
- Single-Mode Readers
- Multi-Mode Readers
- Microplate Washers
- Pipetting & Dispensing Systems
- Handlers & Incubators
- By Application
- Drug Discovery & HTS
- Genomics & Proteomics Research
- Clinical Diagnostics
- Cell-based Assays & Toxicity Testing
- Environmental & Food Safety Testing
- By End User
- Pharmaceutical & Biotechnology Companies
- CROs & CMOs
- Hospitals & Diagnostic Laboratories
- Academic & Research Institutes
- By Geography
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific
- China
- Japan
- India
- Australia
- South Korea
- Rest of Asia-Pacific
- Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
- South America
- Brazil
- Argentina
- Rest of South America
- North America
Detailed Research Methodology and Data Validation
Primary Research
Mordor analysts interviewed lab-automation engineers, CRO buyers, and regional distributors across North America, Europe, and Asia. These talks validated installed-base counts, flagged price gaps in emerging labs, and explained service-contract attach rates that rarely show up online.
Desk Research
We began by pulling instrument flow and funding signals from NIH RePORTER, Eurostat biotech outlays, UN Comtrade HS 9027.50 records, and China grant dashboards. These public feeds drew the first boundary of our model. Guidelines from the Clinical and Laboratory Standards Institute, citation bursts in the Journal of Biomolecular Screening, and patent sets accessed through Questel fixed typical throughput and life-cycle norms.
Next, our team read company 10-K filings, investor decks, and revenue bands on D & B Hoovers and Dow Jones Factiva, which clarified average selling prices and channel splits. The titles above illustrate the evidence base; many smaller statistics and trade notes helped verify niche assumptions.
Market-Sizing & Forecasting
We anchor a top-down build that multiplies installed reader stocks by five-year replacement cycles and global shipment data. We then cross-check results with sampled price-times-volume roll-ups for the ten largest makers. Drivers such as pharma R & D budgets, high-throughput project counts, plate-density shifts, regulatory clearances, and public automation grants feed a multivariate regression projecting 2025-2030 values. Share proxies from distributor insight bridge the missing data for smaller suppliers.
Data Validation & Update Cycle
Automatic variance flags, peer audits, and senior sign-off give three layers of review. We refresh the model each year and trigger interim updates when recalls, tariff moves, or blockbuster assay launches alter demand.
Why Mordor's Microplate Systems Baseline Earns Trust
Published values often differ because every firm chooses its own scope, currency base, and update rhythm. By spelling out inclusions and leaning on transparent drivers, Mordor gives users figures they can trace.
Key gap drivers include some sources folding consumables into hardware, others freezing price curves, and a few stretching uptake multipliers without mid-cycle checks.
Benchmark comparison
| Market Size | Anonymized source | Primary gap driver |
|---|---|---|
| USD 1.21 B (2025) | Mordor Intelligence | N/A |
| USD 0.81 B (2023) | Global Consultancy A | Leaves out washers and clips retail prices |
| USD 1.98 B (2025) | Trade Journal B | Bundles accessories and adds OEM mark-ups |
| USD 4.73 B (2024) | Specialist Research C | Adds reagents and applies PPP currency |
These contrasts show how disciplined variable choice and timely refresh give clients a balanced, reproducible baseline they can trust.


Key Questions Answered in the Report
What CAGR is projected for microplate systems between 2026 and 2031?
The microplate systems market is forecast to grow at a 4.73% CAGR over 2026-2031.
Which product segment grows fastest?
Multi-mode readers are set to advance at 6.06% CAGR, leading product-level growth.
How large is the share of drug discovery applications?
Drug discovery and high-throughput screening accounted for 47.09% of 2025 revenue.
Why are CROs buying more readers?
CROs and CMOs expand at 8.18% CAGR as pharma outsources routine assays, valuing automation-ready readers with GxP compliance.
Which region posts the highest growth rate?
Asia-Pacific leads with 10.13% CAGR through 2031, powered by Chinese and Indian biotech investment.
Page last updated on:









