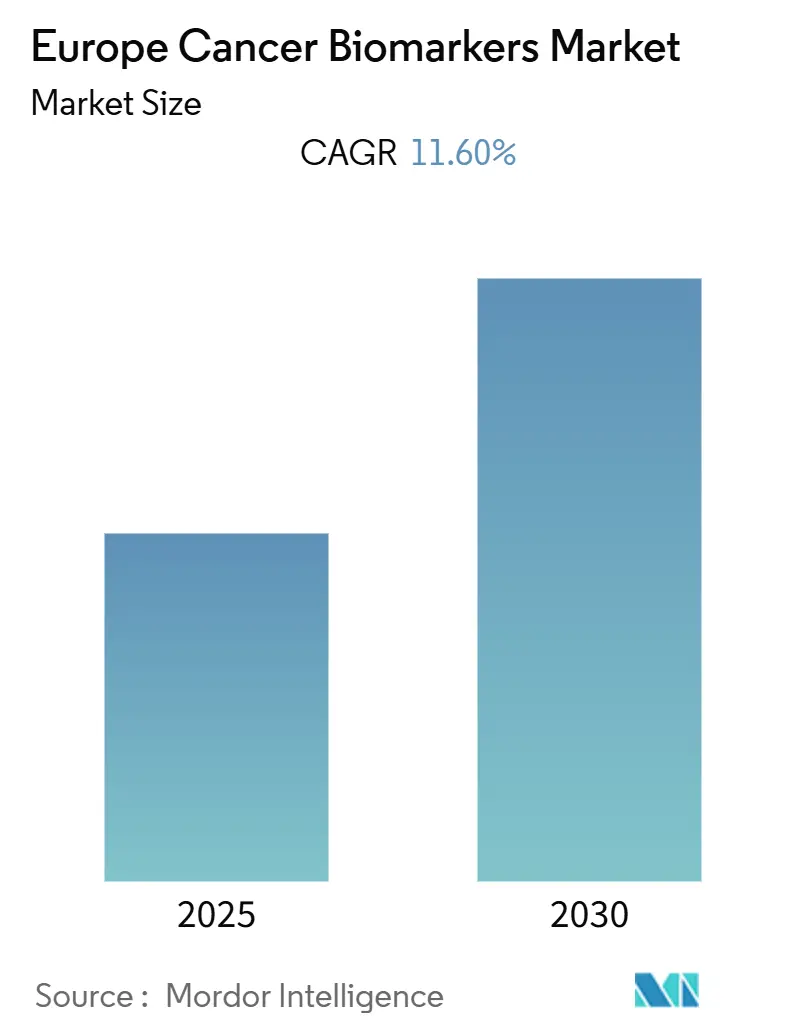
Europe Cancer Biomarkers Market Analysis by Mordor Intelligence
The Europe Cancer Biomarkers Market is expected to register a CAGR of 11.60% during the forecast period.
With the rising of the COVID-19, several companies increased their focus on the development of drugs or vaccines for COVID-19, showing a negative impact on interventional and other cancer biomarker-related clinical trials, which have been stopped as a result of the COVID-19 virus situation across the world. It is observed that it will have a negative impact for a short period of time. The companies such as Pfizer, Eli Lilly, and Bristol-Myers Squibb have also announced that they had halted the new clinical trials as a result of COVID-19. As per Pfizer's announcement, in late March 2020, the company paused the recruitment portion of certain global interventional clinical trial studies to avoid adding to the demands on the healthcare system during the peak of the crisis.
As per the estimates of the Globocan 2020, there were 467,965 new cases of cancer in France, and there had been 185,621 deaths due to it. According to the Fondation de France, cancer affects over 350,000 people every year and kills close to 150,000, representing roughly 1/3 of total deaths. Due to this high prevalence of cancer, increasing efforts are being made in R&D for the development of novel cancer therapeutics and diagnostics.
Thus, cancer biomarker is found playing a pivotal role in risk assessment and early diagnosis. Biomarkers, particularly those associated with genetic factors, offer a quantitative way to determine when individuals are predisposed to some form of cancer. These biomarkers have been developed for almost all cancers with a high prevalence rate. Some of the examples for such biomarkers are mutation on KRAS, p53, EGFR, erbB2 for colorectal, esophageal, liver and pancreatic cancer, and BRCA1 and BRCA2 for breast and ovarian cancer.
Biomarker-based early detection enables sensitive and cost-effective testing, which is expected to reduce cancer mortality. The biomarker's potential to assess the risk precisely has helped in improving the adoption of the technology, which has led to market expansion.
Furthermore, in November 2019, Hitachi and Centre Léon Bérard cancer center in France promoted R&D aimed at improving the efficiency of diagnosis and treatment of cancer and established a new research laboratory called Hitachi Lyon Lab. The research also includes the identification of biomarkers exploiting genomic data in the context of resistance, which may allow predicting responses and prognoses upon radiation treatment. Hence, the high burden of cancer and increasing R&D activities are expected to boost the overall growth of the market.
Europe Cancer Biomarkers Market Trends and Insights
The Lung Cancer Segment is Expected to Hold a Major Market Share in the European Cancer Biomarkers Market
Lung cancer is caused by the uncontrolled growth of abnormal cells that multiply in one lung or both lungs. Although it can affect people who never smoked, people who smoke have the highest risk of lung cancer. The risk of lung cancer increases with the duration and number of cigarettes that the patient smoked. Other risk factors include occupational or environmental exposure to secondhand smoke, asbestos, certain metals, some organic chemicals, radiation, air pollution, and diesel exhaust.
With the rising cancer cases, there are millions of people that are being infected with COVID-19 around the globe. Since the focus of healthcare facilities was shifted to COVID-19, cancer patients' treatment was restricted for a certain period, followed by cancer biomarker-related clinical trials were being halted for a less period of time that showed a slight negative impact on the overall market.
According to the estimates of Globocan 2020, there were 457,960 new cancer cases in the United Kingdom, while prostate, breast, lung, colon, and skin cancer are the most common types of cancer detected in the United Kingdom. Based on the same estimates, there were 415,269 new cases of cancer in Italy and about 174,759 deaths. Therefore, owing to the rising number of cases of cancer, the cancer biomarkers market in Europe is expected to record substantial growth over the forecast period.
Some of the leading biomarkers for lung cancer include CYFRA 21-1, Plasma Kallikrein (KLKB1), Serum Amyloid A (SAA), Haptoglobin β Chain (Hp β), Complement Component 9 (C9), Endoglin (CD105), Peroxiredoxin 1 (PRX1), and Pro-gastrin-releasing Peptide (proGRP). As most of the aforementioned biomarkers are commercially available, the intensity of competition is already high in this segment. Therefore, the early diagnosis, coupled with the rising level of awareness among people, may lead to the healthy growth of the market studied.


Competitive Landscape
The European cancer biomarkers market is highly competitive and consists of a few major players. Some companies currently dominating the market include Abbott Laboratories Inc., Agilent Technologies, Ambry Genetics, Biomerieux, Celera Corporation (Quest Diagnostics), F. Hoffmann-La Roche Ltd, Hologic Inc., Illumina Inc., and Thermo Fisher Scientific. Furthermore, some of the major market players, such as Novartis and Gilead in cancer therapy, are from Europe, one of the factors boosting the R&D activities in the region.
Europe Cancer Biomarkers Industry Leaders
Abbott Laboratories Inc
F. Hoffmann-La Roche Ltd
Illumina Inc
Thermo Fisher Scientific
Biomerieux
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- November 2020: Agilent Technologies announced the launch of the Biomarker Pathologist Training Program, a global initiative created to empower pathologists to score biomarkers accurately and confidently.
- March 2020: Proteomedix announced that Proclarix, its blood-based test for prostate cancer diagnosis, is commercially available in Europe. Proclarix combines proprietary biomarkers and a risk score that accurately identifies an individual patient's risk for clinically significant prostate cancer.


Europe Cancer Biomarkers Market Report Scope
As per the scope of the report, biomarkers are chemical, physical, or biological parameters, which can be used to indicate disease states. Oncology biomarkers offer high-speed, non-invasive cancer diagnoses, and enhance cancer detection and screening. The European cancer biomarkers market is segmented by disease (prostate cancer, breast cancer, lung cancer, colorectal cancer, cervical cancer, and other diseases), type (protein biomarkers, genetic biomarkers, and other types), and profiling technology (omics technology, imaging technology, immunoassays, and cytogenetics). The report offers the value (in USD million) for the above segments.
| Prostate Cancer |
| Breast Cancer |
| Lung Cancer |
| Colorectal Cancer |
| Cervical Cancer |
| Other Diseases |
| Protein Biomarkers |
| Genetic Biomarkers |
| Other Types |
| OMICS Technology |
| Imaging Technology |
| Immunoassays |
| Cytogenetics |
| Europe | Germany (By Disease, Type, and Profiling Technology) |
| United Kingdom (By Disease, Type, and Profiling Technology) | |
| France (By Disease, Type, and Profiling Technology) | |
| Italy (By Disease, Type, and Profiling Technology) | |
| Spain (By Disease, Type, and Profiling Technology) | |
| Rest of Europe (By Disease, Type, and Profiling Technology) |
| By Disease | Prostate Cancer | |
| Breast Cancer | ||
| Lung Cancer | ||
| Colorectal Cancer | ||
| Cervical Cancer | ||
| Other Diseases | ||
| By Type | Protein Biomarkers | |
| Genetic Biomarkers | ||
| Other Types | ||
| By Profiling Technology | OMICS Technology | |
| Imaging Technology | ||
| Immunoassays | ||
| Cytogenetics | ||
| By Geography | Europe | Germany (By Disease, Type, and Profiling Technology) |
| United Kingdom (By Disease, Type, and Profiling Technology) | ||
| France (By Disease, Type, and Profiling Technology) | ||
| Italy (By Disease, Type, and Profiling Technology) | ||
| Spain (By Disease, Type, and Profiling Technology) | ||
| Rest of Europe (By Disease, Type, and Profiling Technology) | ||


Key Questions Answered in the Report
What is the current Europe Cancer Biomarkers Market size?
The Europe Cancer Biomarkers Market is projected to register a CAGR of 11.60% during the forecast period (2025-2030)
Who are the key players in Europe Cancer Biomarkers Market?
Abbott Laboratories Inc, F. Hoffmann-La Roche Ltd, Illumina Inc, Thermo Fisher Scientific and Biomerieux are the major companies operating in the Europe Cancer Biomarkers Market.
What years does this Europe Cancer Biomarkers Market cover?
The report covers the Europe Cancer Biomarkers Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Europe Cancer Biomarkers Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
Europe Cancer Biomarkers Market Report
Statistics for the 2025 Europe Cancer Biomarkers market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Europe Cancer Biomarkers analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.





