Thrombosis Drugs Market Size
Thrombosis Drugs Market Analysis
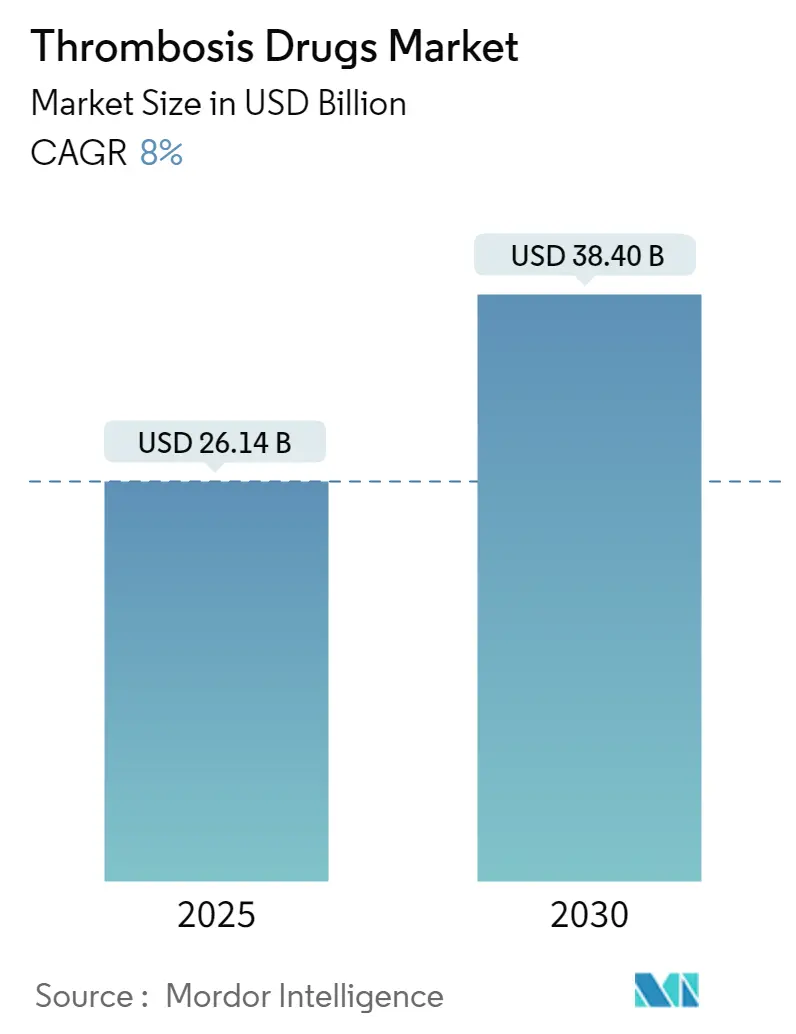
The Thrombosis Drugs Market size is estimated at USD 26.14 billion in 2025, and is expected to reach USD 38.40 billion by 2030, at a CAGR of 8% during the forecast period (2025-2030).
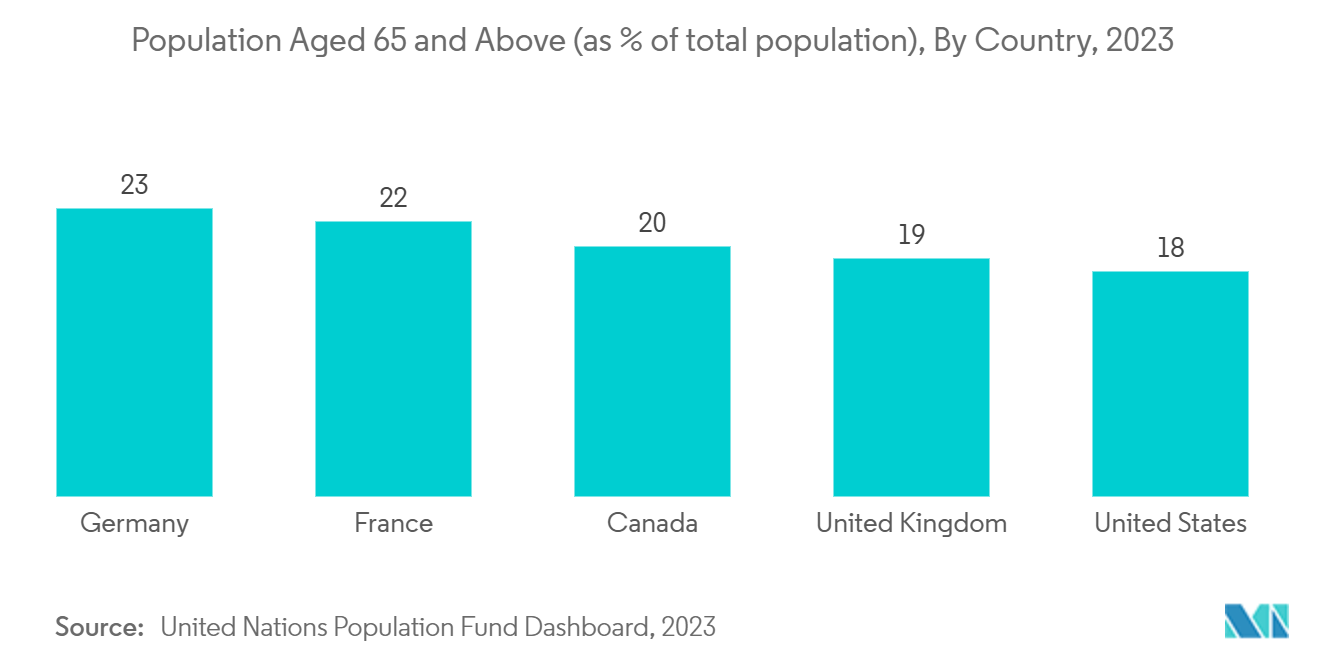
The market is primarily driven by the increase in the geriatric and obese population, the rising burden of cardiovascular diseases and trauma cases, and the technological advancement in hematology. For instance, according to the World Population Aging Report 2023, developed countries are expected to move to a more advanced stage of population aging, with the proportion of older persons rising from 20% in 2023 to 28% in 2050. For other developing countries, this proportion is expected to rise from 9% to 17%. Least developing countries, while still at a lower level, are expected to experience a more rapid increase of about 180% from nearly 4% to over 6% during this period. Since the older population is prone to the disease, it is expected to generate the need for these drugs for treatment, thereby driving the market's growth.
The increasing burden of obesity has also changed significantly in the last decade globally. Developed and emerging markets are having significant issues controlling the associated diseases. For instance, according to the September 2023 published CDC data, in 2022, 22 states in the US reported an adult obesity prevalence of 35% or higher, compared to 19 states in the previous year. Thus, with the increase in the obese population, the demand for thrombotic drugs is expected to increase over the forecast period, as obesity is a significant cause of vascular diseases.
The strategic activities of the market players, such as research and development activities and product launches, are expected to contribute to market growth over the forecast period. For instance, in February 2023, Bayer announced that first patients had been enrolled in the initial trials for its OCEANIC clinical trial program, designed to explore the use of asundexian (BAY2433334), an investigational oral Factor XIa (FXIa) inhibitor, in patients with atrial fibrillation (AF) and in patients with a non-cardioembolic ischemic stroke or high-risk transient ischemic attack (TIA), a temporary period of symptoms similar to those of a stroke. In July 2022, the Food and Drug Administration (FDA) granted abelacimab, a new drug from Anthos Therapeutics, a fast-track designation to treat cancer-related thrombosis.
However, a lack of awareness about venous thromboembolism conditions and the side effects associated with thrombosis drugs may restrain the market's growth over the forecast period.
Thrombosis Drugs Market Trends
Deep Vein Thrombosis (DVT) Segment is Expected to Register a Significant Growth during the Forecast Period
Deep vein thrombosis (DVT) is a blood clot in one of the large, deep veins that returns blood from the leg and rarely in the arm or heart. The segment's growth is driven by the increasing prevalence of chronic deep vein thrombosis and product launches by key market players. Increasing focus on developing low-cost drugs and a large patient pool further contribute to the growth of the thrombosis drugs market.
According to a study published in Nature Journal in October 2023, approximately 1,220,000 total cases of VTE occur in the United States annually. The incidence of VTE in Europe and the United States is estimated to be ~1–2 per 1,000 person-years but varies widely by age, sex, race, and medical conditions. In Asia, the rates of VTE are thought to be lower than in Europe and the USA. For instance, the incidence of VTE in South Korea was estimated to be 0.2 per 1,000 person-years. Thus, the growing prevalence of deep vein thrombosis is expected to drive the segment's growth over the forecast period.
Market players are focusing on introducing innovative products, further adding to the market growth. For instance, in February 2024, Bayer initiated proof-of-concept trials for a novel therapy aimed at patients suffering from deep vein thrombosis (DVT). Differing from existing medications, Bayer's German team has launched the SIRIUS phase 2 trial. This trial focuses on evaluating the efficacy of their anti-alpha2 antiplasmin (anti-α2ap) antibody, BAY3018250, in DVT patients. DVT manifests when blood clots develop in the deep veins, predominantly in the legs. DVT is often associated with old age, which is also expected to enhance the segment growth.
Thus, all factors above, such as the high prevalence of DVT and increasing product launches, are expected to boost the market's growth over the forecast period.
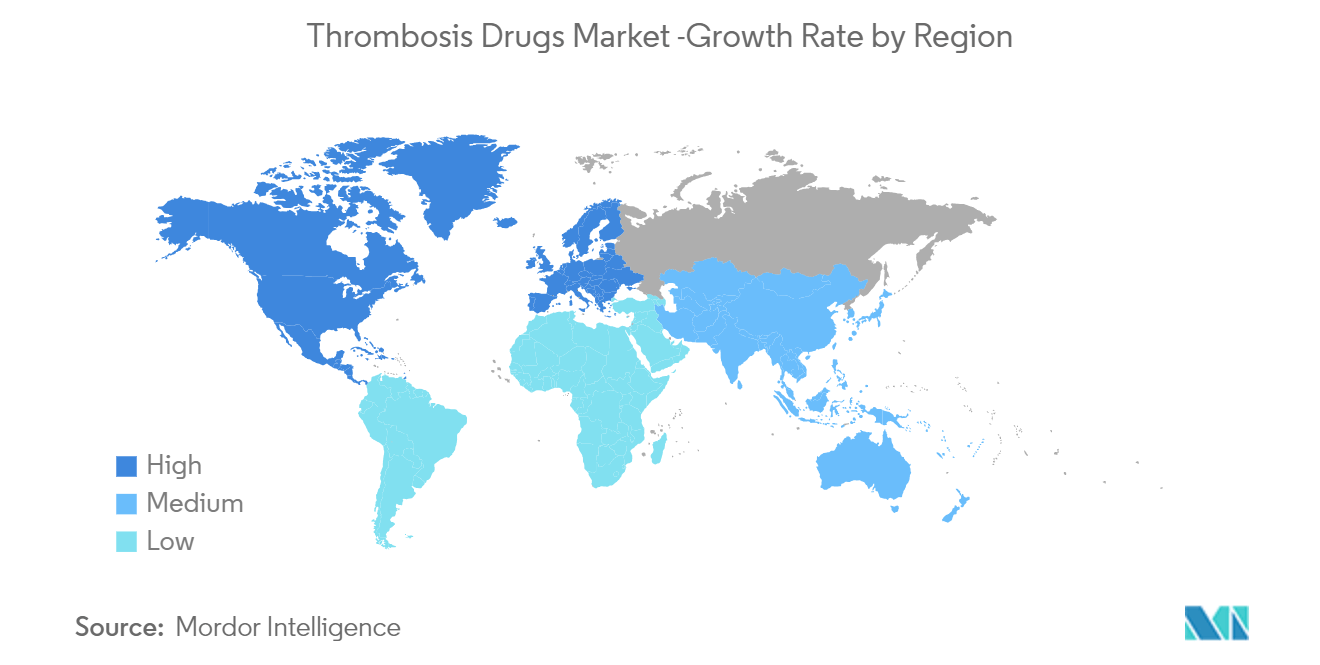
North America is Expected to Hold a Significant Share in the Market Over the Forecast Period
North America is expected to hold a significant share of the market owing to the prevalence of cardiovascular diseases and obesity, the upsurge in demand for progressive treatment options, the early adoption of new technologies, and an increase in the elderly population. Besides, the presence of a diverse patient pool and the increase in participation of domestic and foreign companies in the campaign in these regions is also projected to contribute to the market growth.
The growing burden of thrombotic diseases is expected to contribute in the growth of the market over the forecast period. For instance, as per the data updated by CDC in May 2024, up to 900,000 people in the United States are affected by venous thromboembolism (VTE, a blood clot), each year. Thus, a high burden is anticipated to increase demand for thrombosis drugs, thereby boosting the market growth.
Moreover, according to the data updated by the Canadian Institute for Health Information in July 2022, it was estimated that about 2.4 million Canadians had heart disease in the current year. Thus, with the increasing burden of cardiovascular diseases across the country, the studied market is expected to witness strong growth, as it would lead to increased usage of thrombosis drugs for treatment.
Thus, all aforementioned factors such as the high prevalence of cardiovascular diseases and the growing geriatric population are expected to boost the market growth over the forecast period.
Thrombosis Drugs Industry Overview
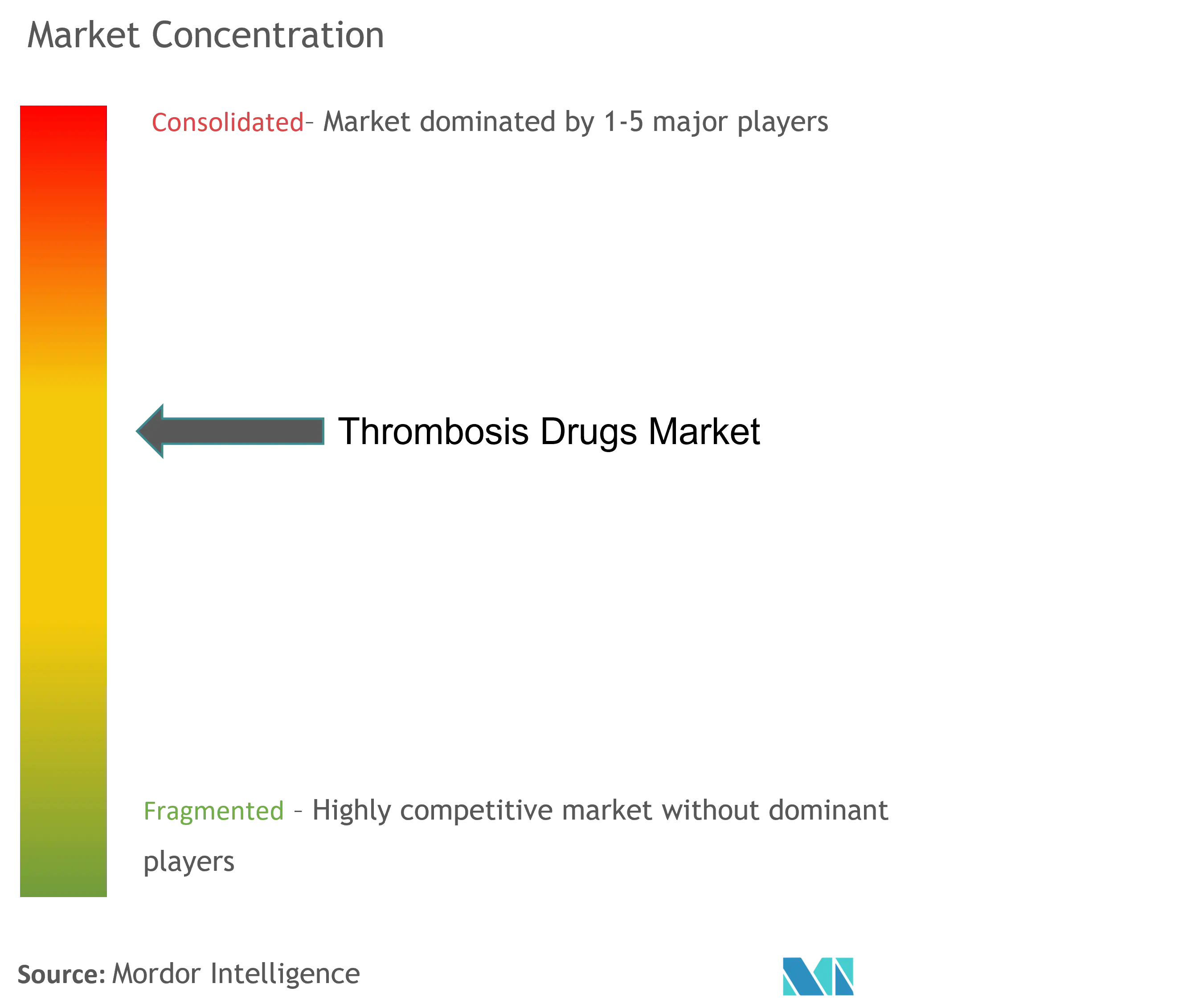
The thrombosis drugs market is semi-consolidated, with the presence of a few global players in the market. For instance, Bayer AG, Boehringer Ingelheim GmbH, Bristol-Myers Squibb, Johnson & Johnson, Sanofi, Pfizer, Inc., etc., are providing these products across the world. These key players are evolving through various strategies, such as new product launches and acquisitions, to secure their positions in the global competitive market.
Thrombosis Drugs Market Leaders
-
Boehringer Ingelheim GmbH
-
Johnson & Johnson
-
Sanofi SA
-
Pfizer Inc.
-
Laurus Lab (Aspen Pharmacare Holdings Limited)
- *Disclaimer: Major Players sorted in no particular order
Thrombosis Drugs Market News
- April 2024: Cadrenal Therapeutics Inc. revealed that the FDA had bestowed Orphan Drug Designation (ODD) upon tecarfarin in the United States. This designation was specifically for averting thromboembolism and thrombosis in patients with various implanted mechanical circulatory support devices, including the left ventricular assist device (LVAD), right ventricular assist device (RVAD), collectively referred to as ventricular assist devices (VADs), as well as the biventricular assist device and total artificial heart.
- :
Thrombosis Drugs Industry Segmentation
As per the scope of the report, a thrombus is a blood clot that develops on the inside of the heart or on the walls of blood vessels as a result of the adhesion of blood platelets, proteins, and cells. Thrombosis is considered to be the major source of morbidity and mortality among elderly patients. It has been established that thrombosis is caused due to certain cardiovascular disorders as a result of old age or obesity.
The thrombosis drugs market is segmented by drug class, disease type, distribution channel, and geography. By drug class, the market is segmented as factor Xa Inhibitor, Heparin, P2Y12 Platelet Inhibitor, and other drug classes. By disease type, the market is segmented as pulmonary embolism, atrial fibrillation, deep vein thrombosis, and other disease types. By distribution channel, the market is segmented into hospital pharmacies, retail pharmacies, and online pharmacies. By geography, the market is segmented as North America, Europe, Asia-Pacific, Middle East and Africa, and South America. The market report also covers the estimated market sizes and trends for 17 different countries across major regions globally. The report offers the value (in USD) for the above segments.
| By Drug Class | Factor Xa Inhibitor | ||
| Heparin | |||
| P2Y12 Platelet Inhibitor | |||
| Other Drug Classes | |||
| By Disease Type | Pulmonary Embolism | ||
| Atrial Fibrillation | |||
| Deep Vein Thrombosis | |||
| Other Disease Types | |||
| By Distribution Channel | Hospital Pharmacies | ||
| Retail Pharmacies | |||
| Online Pharmacies | |||
| Geography | North America | United States | |
| Canada | |||
| Mexico | |||
| Europe | Germany | ||
| United Kingdom | |||
| France | |||
| Italy | |||
| Spain | |||
| Rest of Europe | |||
| Asia-Pacific | China | ||
| Japan | |||
| India | |||
| Australia | |||
| South Korea | |||
| Rest of Asia-Pacific | |||
| Middle East and Africa | GCC | ||
| South Africa | |||
| Rest of Middle East and Africa | |||
| South America | Brazil | ||
| Argentina | |||
| Rest of South America | |||
| Factor Xa Inhibitor |
| Heparin |
| P2Y12 Platelet Inhibitor |
| Other Drug Classes |
| Pulmonary Embolism |
| Atrial Fibrillation |
| Deep Vein Thrombosis |
| Other Disease Types |
| Hospital Pharmacies |
| Retail Pharmacies |
| Online Pharmacies |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
Thrombosis Drugs Market Research FAQs
How big is the Thrombosis Drugs Market?
The Thrombosis Drugs Market size is expected to reach USD 26.14 billion in 2025 and grow at a CAGR of 8% to reach USD 38.40 billion by 2030.
What is the current Thrombosis Drugs Market size?
In 2025, the Thrombosis Drugs Market size is expected to reach USD 26.14 billion.
Who are the key players in Thrombosis Drugs Market?
Boehringer Ingelheim GmbH, Johnson & Johnson, Sanofi SA, Pfizer Inc. and Laurus Lab (Aspen Pharmacare Holdings Limited) are the major companies operating in the Thrombosis Drugs Market.
Which is the fastest growing region in Thrombosis Drugs Market?
Asia-Pacific is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Thrombosis Drugs Market?
In 2025, the North America accounts for the largest market share in Thrombosis Drugs Market.
What years does this Thrombosis Drugs Market cover, and what was the market size in 2024?
In 2024, the Thrombosis Drugs Market size was estimated at USD 24.05 billion. The report covers the Thrombosis Drugs Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Thrombosis Drugs Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on: November 22, 2024
Our Best Selling Reports
Thrombosis Drugs Industry Report
Statistics for the 2025 Thrombosis Drugs market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Thrombosis Drugs analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.