Clinical Trials Market Analysis by Mordor Intelligence
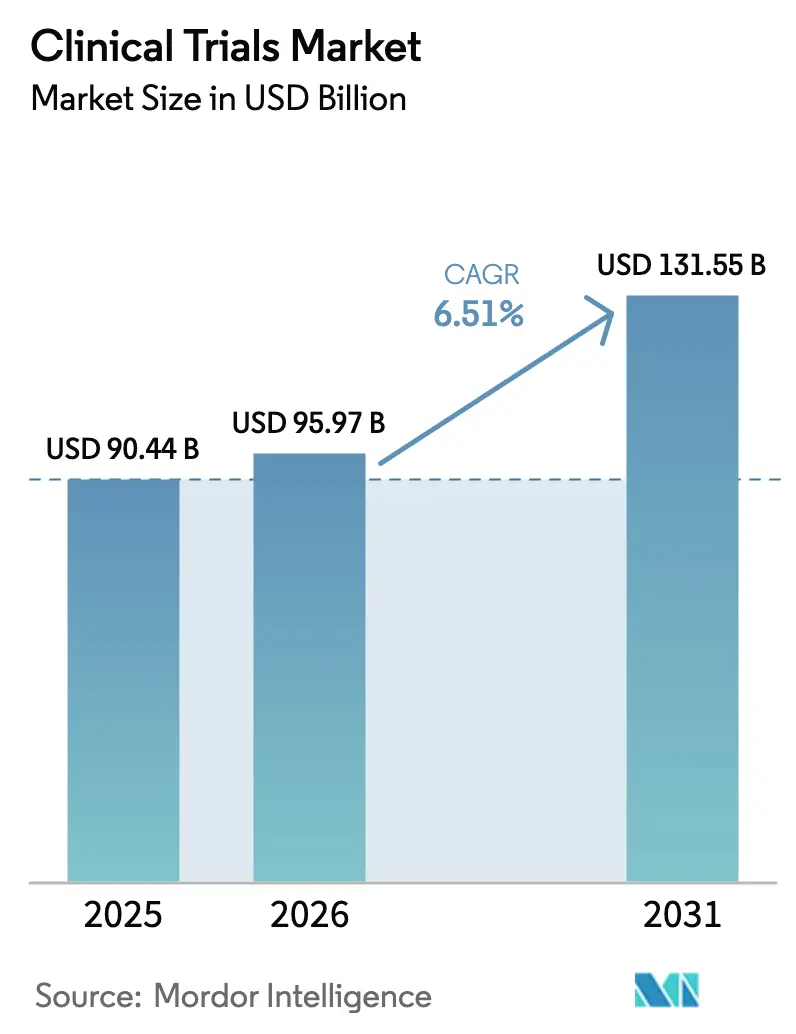
The Clinical Trials Market size was valued at USD 90.44 billion in 2025 and is estimated to grow from USD 95.97 billion in 2026 to reach USD 131.55 billion by 2031, at a CAGR of 6.51% during the forecast period (2026-2031).
Hybrid execution models that blend on-site visits with virtual workflows are becoming increasingly common in the post-pandemic era, prompting sponsors to adopt decentralized components that meet regulatory requirements while also appealing to participants. Phase II studies are attracting larger budgets as precision-medicine candidates demand biomarker-driven proof-of-concept validation before scaling to Phase III. Adaptive designs are shortening timelines for rare-disease programs, while artificial intelligence screening tools are trimming enrollment bottlenecks in oncology and neurology. Competitive dynamics center on technology integration, with leading contract research organizations (CROs) vying to embed predictive analytics that reduce monitoring costs and enhance site selection. Sponsors are also diversifying geographically, shifting enrollment to Asia-Pacific sites that now clear approvals in 30 days, a practice that relieves pressure on saturated U.S. centers.
Key Report Takeaways
- By phase, Phase III studies led with 55.00% of the clinical trials market share in 2025; Phase II is projected to grow at a 6.80% CAGR through 2031.
- By study design, interventional trials captured 72.30% share of the clinical trials market size in 2025, while adaptive formats are advancing at an 8.20% CAGR to 2031.
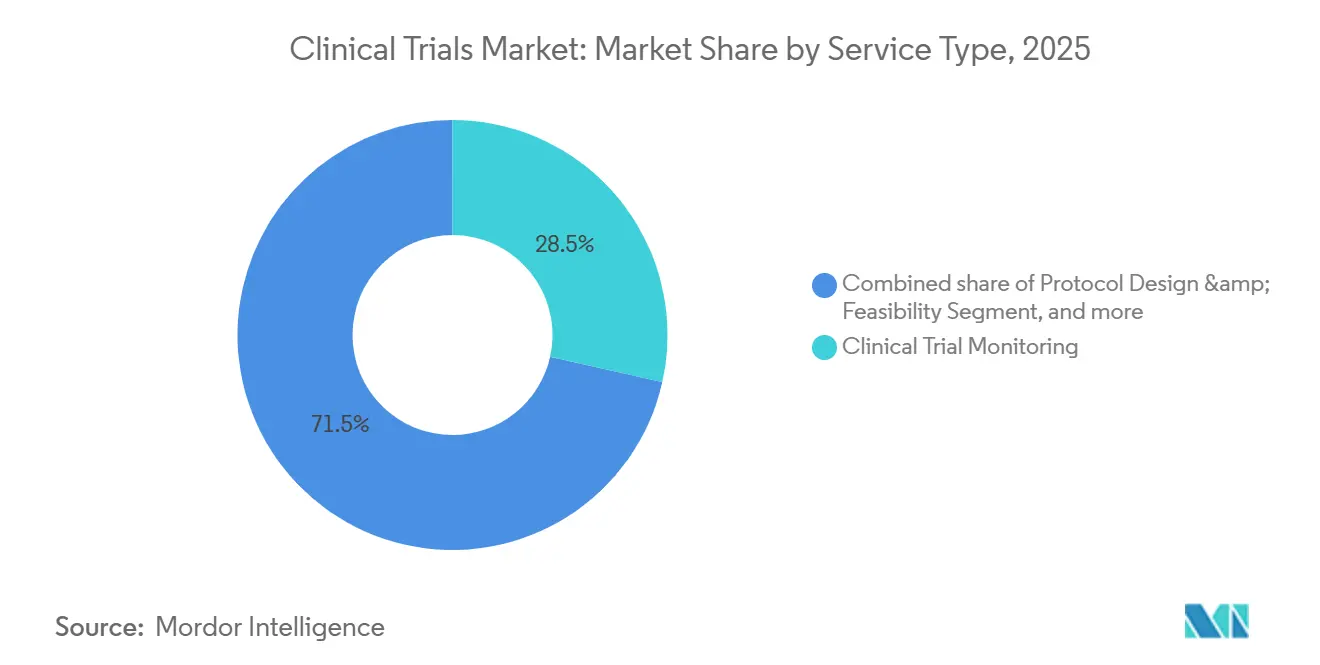
- By service type, clinical trial monitoring accounted for 28.50% of spend in 2025, and decentralized services are forecast to expand at a 14.60% CAGR over 2026-2031.
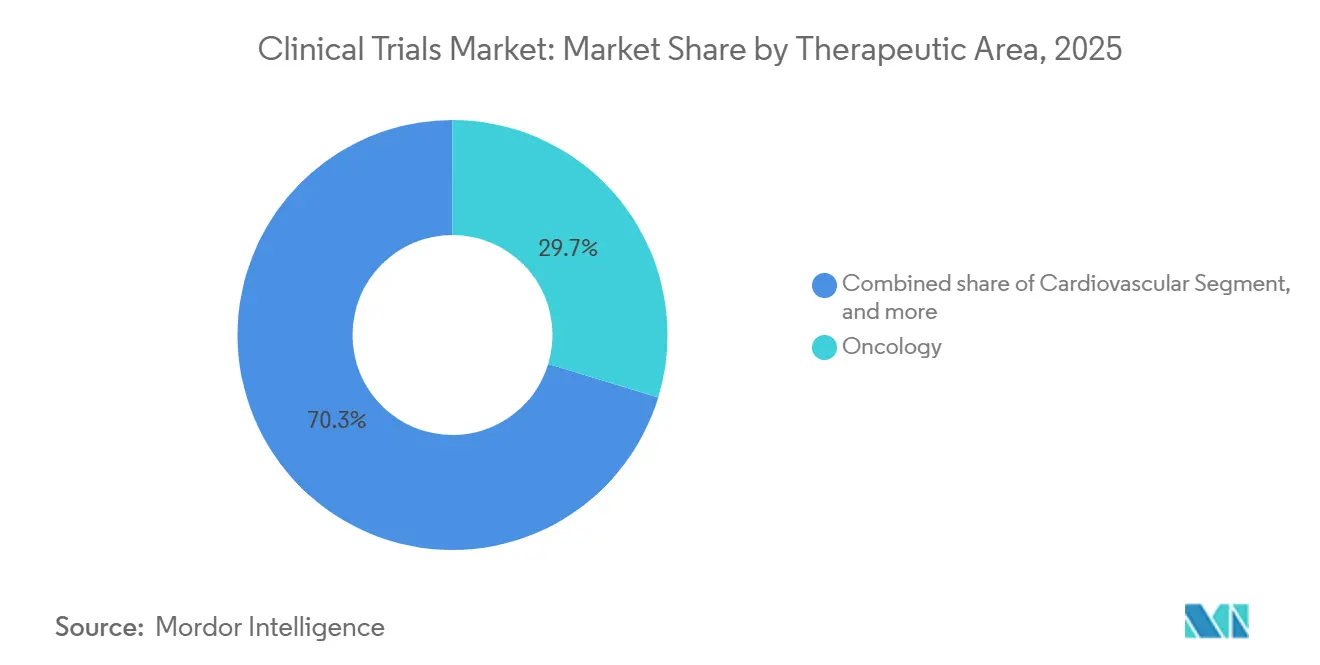
- By therapeutic area, oncology dominated with a 29.70% revenue share in 2025; neurology is poised for the fastest growth, with a 9.10% CAGR to 2031.
- By sponsor, pharmaceutical and biopharmaceutical companies retained 68.00% of the clinical trials industry share in 2025; government and non-profit funding is rising at a 7.50% CAGR.
- By geography, North America accounted for 49.20% of the revenue in 2025; however, Asia is expected to deliver a 7.90% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Clinical Trials Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Growing Volume of Complex Biologics & Precision Medicines Requiring Extensive Trials | +1.8% | North America, EU | Long term (≥ 4 years) |
| Expanded Outsourcing to Full-Service CROs for Cost & Speed Advantages | +1.5% | North America, Asia-Pacific | Medium term (2-4 years) |
| Regulatory Reforms Accelerating Approval Pathways (FDA Fast Track, EMA PRIME) | +1.2% | North America, EU | Medium term (2-4 years) |
| Pandemic-Induced Digital Transformation Driving eClinical Adoption | +1.4% | North America, UK, Australia | Short term (≤ 2 years) |
| Rise of Patient-Owned Health Data Wallets Facilitating Remote Monitoring | +1.1% | United States, EU | Short term (≤ 2 years) |
| Blockchain-Based Consent Management Enhancing Auditability & Compliance | +0.8% | EU, North America | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Growing Volume of Complex Biologics and Precision Medicines Requiring Extensive Trials
Biologics and precision medicines represented 38% of all investigational assets in 2025, up from 29% five years earlier, driving increased demand for longer and data-intensive studies[1]. Cell-and-gene therapy programs needed 1,847 active INDs in the United States during 2025, yet viral-vector capacity constraints meant just 64% of planned protocols actually initiated on schedule. CROs have started acquiring manufacturing assets to mitigate these delays; Thermo Fisher invested USD 420 million to double vector capacity by 2027. Monoclonal-antibody Phase III studies averaged 52 months in 2025, compared to 38 months for small molecules, thereby elevating demand for platforms that manage four to six terabytes of genomic and imaging data per trial.
Expanded Outsourcing to Full-Service CROs for Cost and Speed Advantages
Pharmaceutical sponsors outsourced 73% of their clinical budgets to CROs in 2025 and secured Phase III enrollment 5.3 months faster than in-house programs. IQVIA posted USD 29.4 billion in backlog, equal to 2.1 years of revenue visibility, while Medpace hit a 94% on-time database-lock rate by embedding oncology nurses at sites. Asian CROs still offer a three-to-one cost edge, although 14 Indian sites received FDA warning letters in 2025 for GCP lapses, signaling that quality oversight remains critical.
Regulatory Reforms Accelerating Approval Pathways
FDA granted 87 Fast Track designations in 2025 and EMA accepted 41 PRIME submissions, cutting review cycles by several months and allowing sponsors to file rolling applications that monetize products four to seven months sooner. Japan followed suit with conditional approvals for regenerative therapies, shaving almost one year off local launches. Harmonized monitoring requirements under ICH E6(R3) now permit a single plan across multiple regions, trimming compliance outlays by 18%.
Pandemic-Induced Digital Transformation Driving eClinical Adoption
Decentralized studies accounted for 29% of all new protocols in 2025, a leap from 11% in 2019. Medidata processed 4.2 million ePRO entries in 2025, flagging adverse events 3.4 days faster than paper diaries. Wearable sensors featured in 22% of cardiovascular and neurology trials, reducing on-site visits by 40%. Pfizer enrolled 1,840 participants across 38 U.S. states without a single brick-and-mortar site and achieved 91% retention. FDA draft guidance released in March 2025 formally recognized home-based assessments as primary endpoints if validated, although GDPR continues to mandate data residency within the EU.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High Attrition Rates in Oncology and CNS Trials Increasing Cost Risk | -0.9% | North America, EU | Long term (≥ 4 years) |
| Stringent Data-Privacy Legislations Limiting Cross-Border Data Flows | -0.6% | EU, UK, Asia-Pacific | Medium term (2-4 years) |
| Scarcity of GMP-Compliant Viral Vector Manufacturing Capacity for Gene Therapy Trials | -0.7% | North America, EU, Asia | Long term (≥ 4 years) |
| Rising Carbon-Footprint Accountability Pressures on Multisite Trials | -0.4% | Global | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
High Attrition Rates in Oncology and CNS Trials Increasing Cost Risk
Oncology protocols posted a 52% Phase III failure rate in 2025, driven by tumor heterogeneity and stringent payer demands for overall survival data. Alzheimer’s programs fared worse, with a 68% failure rate, causing each unsuccessful study to absorb USD 180-240 million in sunk costs. Adaptive designs help mitigate risk; Roche’s 2025 basket trial trimmed enrollment by 34% and saved USD 62 million by dropping weak cohorts mid-stream. Germany’s IQWiG rejected 41% of oncology submissions in 2025, prompting sponsors to focus on more rigorous endpoints[2].
Stringent Data-Privacy Legislations Limiting Cross-Border Data Flows
GDPR added 8-10 weeks to multinational start-up timelines in 2025 as sponsors set up EU data lakes. China’s Personal Information Protection Law lengthened interim analyses by up to 18 weeks, while India’s Data Protection Act forced CROs to build domestic data centers costing an extra USD 47 million in 2025. Pfizer’s blockchain consent pilot cut compliance costs 18% yet adoption sits below 5% due to integration burdens.
Segment Analysis
By Phase: Adaptive Phase II Designs Accelerate Go/No-Go Decisions
Phase II studies are projected to grow at a 6.80% CAGR through 2031 and captured rising interest as adaptive methodologies optimize dose-finding and efficacy confirmation inside a single protocol. Novartis used Bayesian randomization in 2025 to shift enrollment toward high-performing arms, trimming total exposure by 28%. Despite Phase III’s 55% clinical trials market share in 2025, its duration and attrition rates spur sponsors to invest earlier in seamless Phase II/III pathways that may condense timelines by 9-12 months. Phase I programs benefit from micro-dosing and rapid mass-spectrometry assays that deliver PK data within 48 hours, while Phase IV studies piggyback on pivotal trials to amortize site costs.
By Study Design: Interventional Models Dominate but Observational Evidence Gains Traction
Interventional trials accounted for 72.30% of 2025 revenue, yet pragmatic and observational approaches are also essential for generating real-world evidence. EMA guidance allows label expansions supported by observational datasets of at least 1,200 subjects adjusted by propensity scores[3]. Expanded-access programs accounted for 4% of total activity and helped build physician familiarity ahead of launches. The NIH funded 11 pragmatic megatrials in 2025 that utilized electronic health-record randomization to reduce costs by two-thirds.
Note: Segment shares of all individual segments available upon report purchase
By Service Type: Monitoring Leads as Decentralized Services Surge
Monitoring represented 28.50% of 2025 revenue, reflecting regulatory preference for data verification, but centralized analytics now trigger risk-based visits and lower costs five-fold. Medidata AI flagged 14% more anomalies than manual review, reducing monitoring labor 31% in 2025. Decentralized trial services, advancing at 14.60% CAGR, bundle telehealth, wearables, and direct-to-patient logistics, enabling the clinical trials market size for virtual elements to climb rapidly. IQVIA’s feasibility algorithm cut site-selection cycles to three weeks, boosting enrollment accuracy by 19 points.
By Therapeutic Area: Oncology Still Dominates, Neurology Accelerates
Oncology kept a 29.70% hold on revenue, supported by 1,340 active mid- to late-stage programs. High failure rates push adoption of basket and umbrella schemes aimed at biomarker-defined cohorts. Neurology trials, aided by amyloid-PET and plasma-tau assays, grow at 9.10% CAGR, improving patient selection and trimming sample sizes 35%. Cardiovascular studies gained momentum with nine FDA approvals in 2025, while metabolic, infectious-disease, and immunology programs remain steady contributors.
Note: Segment shares of all individual segments available upon report purchase
By Sponsor Type: Pharma Leads, Public Funding Spurs Precision Oncology
Pharma and biopharma sponsors retained 68% share in 2025, directing late-stage budgets that often top USD 500 million per Phase III protocol. Government and non-profit bodies logged a 7.50% CAGR as NIH’s Cancer Moonshot steered USD 2.8 billion to 14 multicenter trials. Academic institutes covered 18% of activity, championing head-to-head comparisons the private sector avoids. Device makers and foundations further diversified the clinical trials market, especially in cardiovascular and infectious-disease fields.
Geography Analysis
North America controlled 49.20% of global revenue in 2025, stoked by FDA incentives, a high site density, and enrollment rates of 1.2 patients per site each month, 40% above Europe. Canada added 6% of regional studies, benefiting from protocol alignment with the United States, while Mexico gained prominence as ICON’s 2025 operations hub targeted 30% cost savings.
Asia-Pacific outpaced all regions at 7.90% CAGR through 2031, powered by China’s 62 IND clearances in 2025 and India’s 30-40% per-patient cost advantages. Conditional approvals for regenerative medicines in Japan and a 43.5% R&D tax refund in Australia further widened the region’s appeal. South Korea leveraged its national insurance database to supply real-world comparators for single-arm oncology designs.
Europe captured 28% market share in 2025 after the Clinical Trials Regulation slashed average approval time to 10 months and enabled simultaneous multi-country site activation. The UK’s rolling-review model enticed 14 rare-disease protocols in 2025. GCC nations funded landmark diabetes and oncology studies to diversify health economies, while Brazil and Argentina together made up 4% of global volume despite currency and import bottlenecks.
Competitive Landscape
The top five CROs captured about 42% of 2025 revenue, leaving ample room for mid-tier and niche providers. IQVIA’s Orchestrated Clinical Trials suite won eight long-term contracts worth USD 3.2 billion, demonstrating sponsor appetite for unified platforms. Thermo Fisher’s 2024 acquisition of CorEvitas integrated registry data with trial workflows, signaling that real-world data capabilities are becoming essential. Novotech and Caidya outperformed in early-phase oncology by leveraging Asia-Pacific cost advantages and AI-based pharmacokinetic modeling that surfaced safety signals six days earlier than legacy monitoring.
AI-driven patient recruitment, blockchain-secured data integrity, and direct-to-patient delivery comprise emerging white-space opportunities as decentralized protocols head toward 40% of total trials by 2028. Technology-forward CROs secure awards 1.4 times faster than peers, while FDA warning letters—14 in 2025—continue to penalize low-compliance regions and reinforce the value of robust quality systems.
Clinical Trials Industry Leaders
IQVIA Holdings Inc.
Laboratory Corporation of America (Labcorp)
ICON plc
Parexel International Corp.
Syneos Health
- *Disclaimer: Major Players sorted in no particular order
Recent Industry Developments
- July 2025: Hoth Therapeutics, Inc., a clinical-stage biopharmaceutical company, announced its collaboration with ICON Clinical Research Limited to expand its Phase II clinical trial. The trial focuses on cancer patients experiencing skin toxicities caused by Epidermal Growth Factor Receptor Inhibitors (EGFRi). This collaboration aims to advance treatment options for managing EGFRi-related skin side effects.
- March 2025: Mural Health Technologies, Inc. and ICON plc have formed a partnership to use the Mural Link platform for clinical trial participant management. ICON will leverage Mural Link’s features for participant payments, tax handling, travel support, communication, and analytics. This collaboration aims to enhance efficiency and support for clinical trial participants.
- March 2025: ICON plc has become the first major Clinical Research Organization to adopt Medidata Clinical Data Studio fully. This integration aims to enhance data management efficiency and facilitate streamlined review processes. The move positions ICON as a leader in innovative clinical data solutions.
Research Methodology Framework and Report Scope
Market Definitions and Key Coverage
Our study defines the clinical trials market as all revenue generated from planning, execution, and close-out services provided for Phase I-IV interventional, observational, and expanded-access studies sponsored by pharmaceutical, biotechnology, and medical-device organizations. Activities span protocol design, site and patient recruitment, regulatory submissions, monitoring, data management, biostatistics, medical writing, and associated decentralized or hybrid trial support.
Scope exclusion: animal and pre-clinical in-vivo research expenditures are outside the present estimate.
Segmentation Overview
- By Phase
- Phase I
- Phase II
- Phase III
- Phase IV
- By Study Design
- Interventional / Treatment Studies
- Observational Studies
- Expanded Access Studies
- By Service Type
- Protocol Design & Feasibility
- Site Identification & Start-Up
- Regulatory Submission & Approval
- Clinical Trial Monitoring
- Data Management & Biostatistics
- Medical Writing
- Other Service Types
- By Therapeutic Area
- Oncology
- Cardiovascular
- Neurology
- Infectious Diseases
- Metabolic Disorders (Diabetes, Obesity)
- Immunology / Autoimmune
- Other Therapeutic Areas
- By Sponsor Type
- Pharmaceutical & Biopharmaceutical Companies
- Medical Device Companies
- Academic & Research Institutes
- Government & Non-Profit Organizations
- Geography
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Rest Of Europe
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest Of Asia-Pacific
- Middle-East And Africa
- GCC
- South Africa
- Rest Of Middle East And Africa
- South America
- Brazil
- Argentina
- Rest Of South America
- North America
Detailed Research Methodology and Data Validation
Desk Research
Analysts first mapped the universe of ongoing and completed studies using freely accessible registries such as ClinicalTrials.gov, WHO-ICTRP, EMA's EudraCT, and Japan's jRCT. We then overlaid cost norms and wage indices from sources such as the U.S. Bureau of Labor Statistics, Eurostat, and the World Bank to approximate phase-specific spend by region. Industry position papers from the Association of Clinical Research Organizations, peer-reviewed journals in Trials, and company filings enriched benchmark assumptions. Paid repositories, including D&B Hoovers for CRO financials and Dow Jones Factiva for deal tracking, supplied granular validation points. The sources cited illustrate our approach; many additional records informed final judgments.
Primary Research
Mordor analysts conducted structured interviews with clinical operations leaders at global CROs, principal investigators across North America, Europe, and Asia-Pacific, and procurement heads within mid-sized biopharma firms. These conversations clarified real-world ranges for monitoring visit frequency, decentralized tool uptake, typical margin structures, and regional price uplifts, allowing us to fine-tune desk-derived ratios.
Market-Sizing & Forecasting
A top-down construct begins with the global count of active and newly initiated trials, which is multiplied by phase-weighted cost curves and an outsourcing penetration factor to yield the total addressable service pool. Select bottom-up checks, sampled CRO revenue roll-ups and investigator grant surveys, are layered in to reconcile gaps. Key model drivers include annual R&D outlays, trial start velocity, protocol complexity scores, decentralized trial adoption rates, inflation-adjusted wage trends, and disease-burden shifts. Multivariate regression links these variables to historical spend and projects through 2030, while scenario analysis stress-tests upside and downside cases. Where bottom-up samples under-represent emerging regions, regional CPI-adjusted cost proxies bridge the gap.
Data Validation & Update Cycle
Outputs pass three rounds of variance checks against independent indices, after which senior analysts review assumptions and contact respondents to resolve anomalies. The dataset refreshes each year, with mid-cycle updates triggered by material regulatory or macro-economic events, ensuring clients see the latest vetted view.
Building Confidence in Our Clinical Trials Baseline
Why Mordor's Clinical Trials Benchmark Earns Decision-Maker Trust
Published estimates often diverge because firms choose different service baskets, inflate or deflate decentralization uptake, and lock models on varying refresh dates.
Key gap drivers include: some publishers omit in-house sponsor spend, others cap forecasts at CRO revenues alone, while a few apply static average study costs that ignore rising protocol complexity and regional wage drift.
Mordor's disciplined inclusion of both outsourced and internal spend, yearly recalibration of cost curves, and explicit treatment of hybrid trial services close these gaps.
Benchmark comparison
| Market Size | Anonymized source | Primary gap driver |
|---|---|---|
| USD 90.10 B (2025) | Mordor Intelligence | - |
| USD 64.94 B (2025) | Global Consultancy A | Excludes decentralized platforms and applies conservative cost inflation |
| USD 84.70 B (2024) | Industry Journal B | Uses earlier base year and partial ancillary service coverage |
The comparison shows that when scope, cost escalators, and refresh cadence are harmonized, Mordor's figure offers a balanced, transparent baseline grounded in clearly traceable variables and repeatable steps, giving stakeholders dependable numbers for strategic choices.
Key Questions Answered in the Report
How large is the clinical trials market today?
The clinical trials market size reached USD 95.97 billion in 2026 and is forecast to climb to USD 131.55 billion by 2031.
Which phase is growing the fastest?
Phase II protocols are expanding at a 6.80% CAGR through 2031 owing to adaptive designs that streamline dose selection and efficacy confirmation.
Why are decentralized trials gaining traction?
Decentralized models lift patient-retention rates to 85% and lower monitoring costs by replacing many on-site visits with telehealth and wearables.
Which region is projected to post the highest growth?
Asia-Pacific leads with a 7.90% CAGR, supported by regulatory reforms in China and cost advantages in India.
Who are the leading service providers?
IQVIA, Labcorp, ICON, Parexel, and Thermo Fisher's PPD division together account for about 42% of global CRO revenue.
What is the biggest operational hurdle for multinational trials?
Divergent data-privacy laws such as GDPR and China's PIPL add 8-18 weeks to timelines by requiring localized data storage and extra approvals.



