Vaccine Adjuvants Market Size and Share
Vaccine Adjuvants Market Analysis by Mordor Intelligence
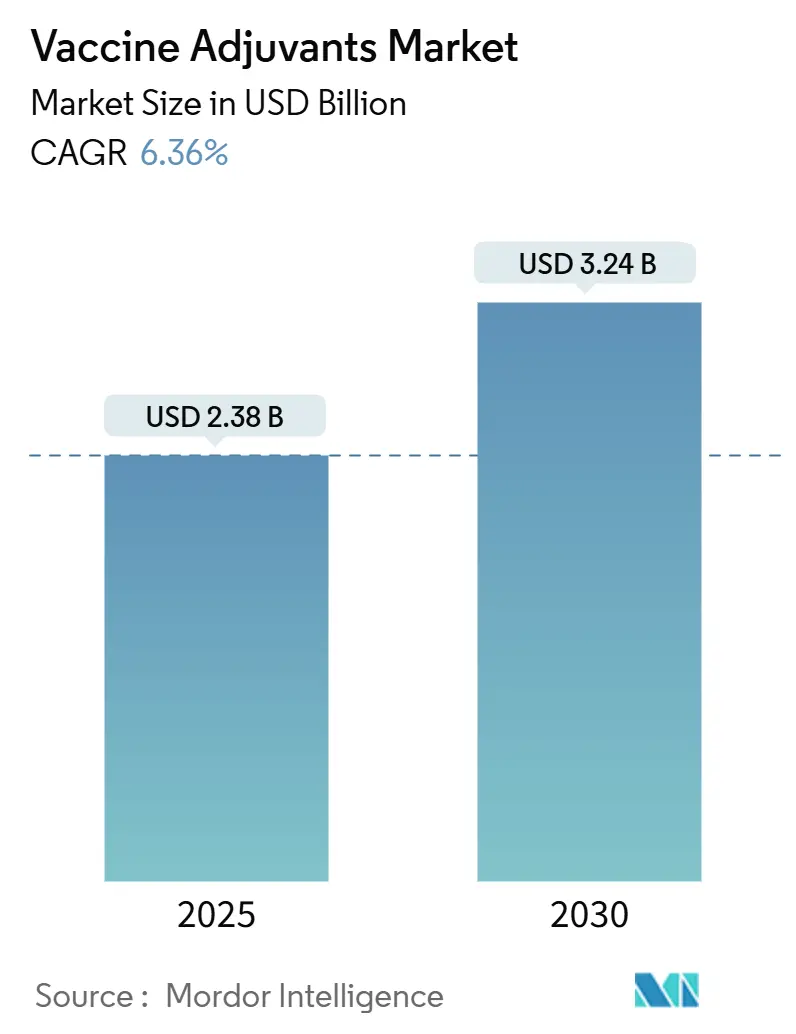
The vaccine adjuvants market stands at USD 2.38 billion in 2025 and is forecast to reach USD 3.24 billion by 2030, advancing at a 6.36% CAGR. This sustained expansion reflects the pharmaceutical sector’s pivot toward next-generation immunization platforms that need sophisticated adjuvant technologies to amplify immune responses, enable novel antigen formats, and support thermostable formulations. Government commitments to pandemic preparedness add predictable purchase volumes, while AI-guided design shortens formulation cycles and reduces cold-chain dependence, lowering distribution costs and widening global access. Intensifying research into mRNA, self-amplifying RNA, and virus-like particle (VLP) vaccines further lifts demand, as these platforms rely on potent adjuvants to offset the low intrinsic immunogenicity of purified or synthetic antigens [1]Nature, "Vaccine innovation moves beyond COVID-19," nature.com. Supply security for saponin and triterpenoid inputs and regulatory clarity for novel pathways such as STING agonism remain watchpoints, yet continued capital inflows into biotech innovation signal confidence in the long-term attractiveness of the vaccine adjuvants market.
Key Report Takeaways
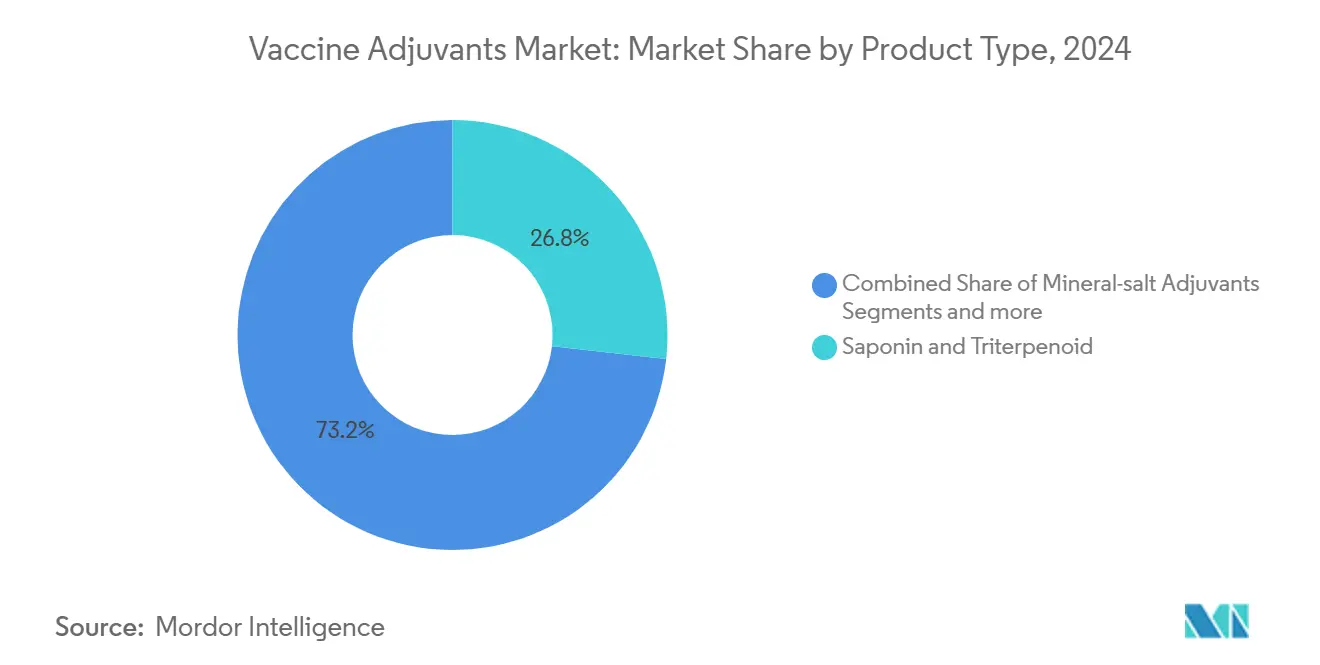
- By product type, saponin and triterpenoid systems led with 26.78% revenue share in 2024, while virus-like particles are projected to grow at a 7.12% CAGR through 2030.
- By usage type, active immunostimulants held 47.89% of the vaccine adjuvants market share in 2024; vehicle adjuvants record the highest forecast CAGR at 7.04% to 2030.
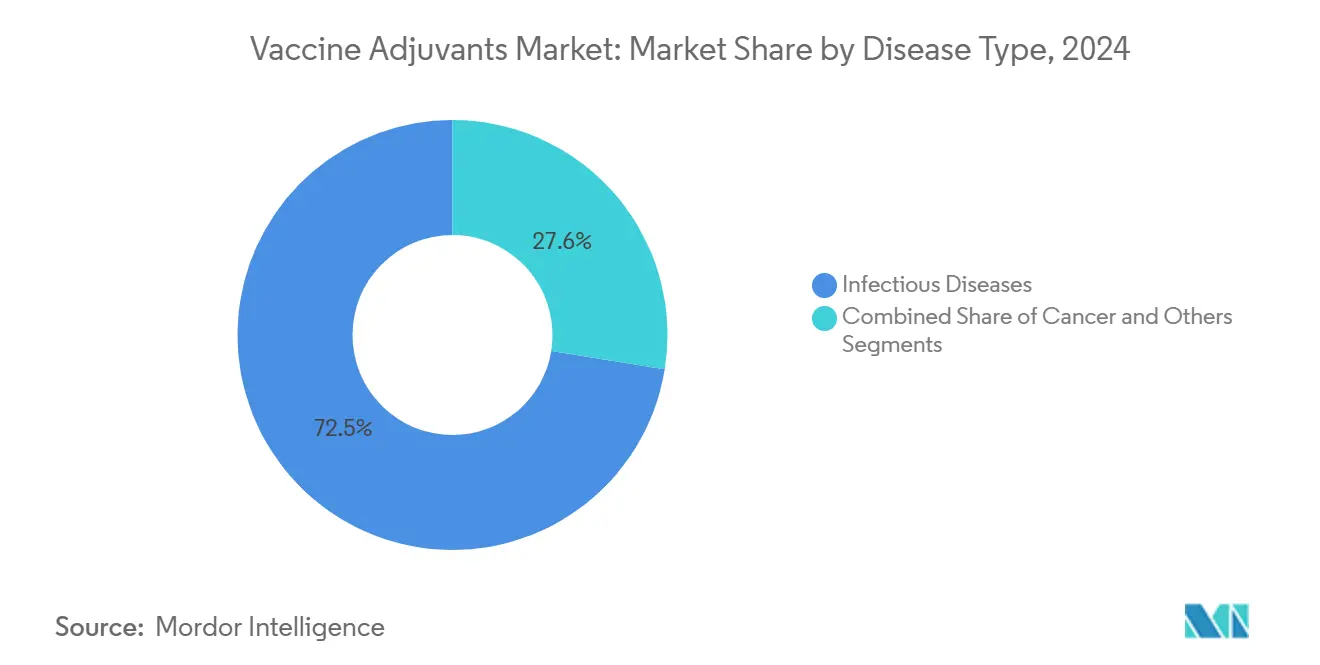
- By disease type, infectious-disease vaccines accounted for 72.45% of the vaccine adjuvants market size in 2024, whereas oncology applications are set to expand at 7.06% CAGR.
- By application, commercial vaccines captured 68.31% share of the vaccine adjuvants market size in 2024; research applications are advancing at a 7.34% CAGR through 2030.
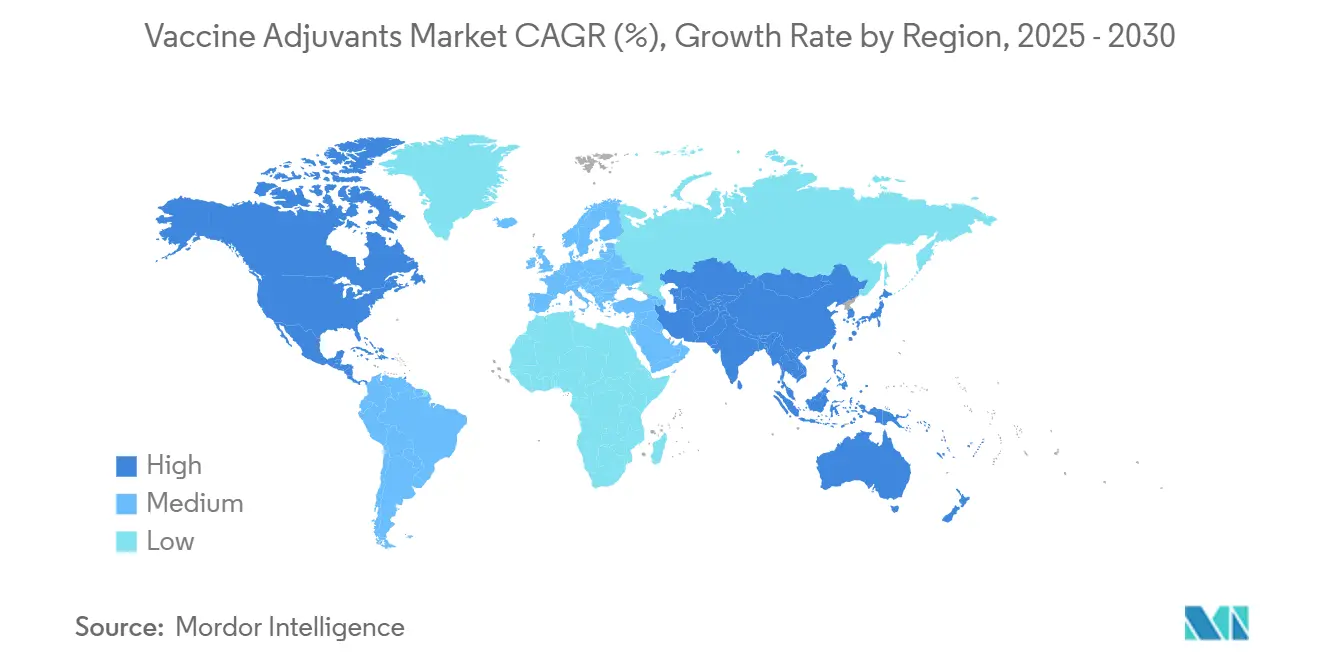
- By geography, North America commanded 41.12% share in 2024, while Asia-Pacific is forecast to accelerate at a 7.45% CAGR to 2030.
Global Vaccine Adjuvants Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Expanding government immunization recommendations | +1.0% | Global, strongest in Asia-Pacific and emerging markets | Medium term (2-4 years) |
| Unmet vaccine needs for emerging zoonoses | +0.8% | Tropical and subtropical regions | Short term (≤2 years) |
| Rising adoption of recombinant & synthetic antigens | +1.1% | North America and EU leading, Asia-Pacific following | Medium term (2-4 years) |
| Accelerating mRNA-platform demand for novel adjuvants | +1.4% | North America and EU as early adopters | Long term (≥4 years) |
| Expansion of microbial-derived TLR agonist pipelines | +0.5% | Global, concentrated in biotechnology hubs | Long term (≥4 years) |
| AI-designed nano-alum formulations enabling cold-chain-free distribution | +0.9% | Global, highest impact in resource-limited settings | Long term (≥4 years) |
| Source: Mordor Intelligence | |||
Expanding Government Immunization Recommendations
Broader national vaccine schedules now target adolescents, adults, and the elderly, steadily enlarging the eligible base for adjuvanted products. The 2024 FDA approval of an adjuvanted H5N1 vaccine for pandemic stockpiling and the EMA’s recommendation of MF59-enhanced influenza formulations for adults over 65 illustrate policy momentum that rewards manufacturers with reliable volume offtake [2]Peter Malfertheiner, "Safety, Tolerability, and Immunogenicity of aH5N1 Vaccine in Adults with and Without Underlying Immunosuppressive Conditions," MDPI, mdpi.com. Public-health authorities also highlight cost-avoidance benefits tied to reduced hospitalization rates, reinforcing budget allocations for adjuvant-rich products. This alignment between health economics and procurement creates a stable demand floor for the vaccine adjuvants market.
Unmet Vaccine Needs for Emerging Zoonoses
Climate-linked habitat shifts, intensified urban–wildlife interfaces, and global trade facilitate spillover events, heightening demand for fast-acting vaccines that rely on potent adjuvants for rapid immunogenicity. Self-amplifying RNA candidates show antigen-dose reductions of up to 40-fold when paired with optimized adjuvants, enabling emergency surge manufacturing [3]Thomas Vallet, "Self-Amplifying RNA: Advantages and Challenges of a Versatile Platform for Vaccine Development," MDPI, mdpi.com. The WHO’s Disease X framework explicitly lists broad-spectrum adjuvant platforms as priority technologies, signaling multi-lateral funding support that lifts near-term purchase certainty.
Rising Adoption of Recombinant & Synthetic Antigens
Synthetic biology provides precise antigen constructs but often weak innate stimulus, making adjuvants indispensable for protective titers. The Matrix-M-enhanced recombinant spike vaccine demonstrated 90% efficacy in pivotal trials, confirming how tailored adjuvants can transform purified proteins into high-performance vaccines. Scalable cell-culture production and absence of pathogen handling bolster gross-margin potential, stimulating further recombinant pipeline expansion and reinforcing structural growth for the vaccine adjuvants market.
Accelerating mRNA-Platform Demand for Novel Adjuvants
Beyond COVID-19, mRNA pipelines now span influenza, RSV, and personalized cancer vaccination. New lipid-nanoparticle chemistries such as poly(carboxybetaine) improve endosomal escape and mitigate anti-PEG responses, while trans-amplifying mRNA architectures cut payload needs forty-fold, decreasing batch-capacity constraints. Each breakthrough requires adjuvant–delivery co-optimization, underpinning long-range growth of the vaccine adjuvants market.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Local & systemic toxicity concerns | -0.5% | Global, heightened scrutiny in EU and North America | Short term (≤2 years) |
| High discovery & pre-clinical screening costs | -0.8% | Global, highest impact on biotechnology companies | Medium term (2-4 years) |
| Scale-up challenges for squalene & QS-21 supply chains | -0.4% | Global, concentrated in established manufacturing regions | Medium term (2-4 years) |
| Regulatory uncertainty around novel STING agonists | -0.6% | North America and EU, with spillover effects globally | Long term (≥4 years) |
| Source: Mordor Intelligence | |||
Local & Systemic Toxicity Concerns
Post-marketing surveillance increasingly detects rare inflammatory events, compelling regulators to tighten data requirements. Class B CpG constructs, for instance, destabilize protein antigens and may heighten off-target reactivity, prompting extended toxicology panels and pharmacovigilance. Heightened safety thresholds elongate timelines and raise capital demands, tempering the vaccine adjuvants market’s near-term growth.
High Discovery & Pre-Clinical Screening Costs
Mechanistic studies across multiple species, formulation stress testing, and complex analytics can consume USD 50–100 million before a first-in-human trial. Such outlays exceed the financing bandwidth of many early-stage developers, concentrating innovation within large pharmas that can amortize risk over broad portfolios. The resulting capital hurdle inhibits pipeline diversity and stalls some high-potential candidates.
Segment Analysis
By Product Type: Saponin Dominance Faces VLP Disruption
Saponin and triterpenoid systems controlled 26.78% of the vaccine adjuvants market size in 2024, anchored by QS-21 and AS01 deployments in shingles, malaria, and tuberculosis programs. Their dual induction of humoral and cellular immunity sustains demand, yet natural-source extraction risks and rising sustainability mandates propel investment in semi-synthetic analogs. Virus-like particles, though holding a smaller base, will rise at a 7.12% CAGR through 2030, propelled by BacFreets contamination-reduction technology that simplifies scale-up.
Manufacturers increasingly assess supply diversification to mitigate saponin harvest volatility, while synthetic biology labs refine VLP scaffolds that co-display multivalent antigens and intrinsic pattern-recognition motifs, potentially sidelining separate adjuvant components. Aluminum-salt, emulsion, and liposome formulations continue anchoring routine pediatric schedules, whereas carbohydrate and bacterial-derived TLR agonists address niche indications that require tailored immune polarization. This coexistence of legacy and disruptive technologies ensures the vaccine adjuvants market retains a heterogeneous product landscape.
Note: Segment shares of all individual segments available upon report purchase
By Usage Type: Active Immunostimulants Lead Vehicle Innovation
Active immunostimulants secured 47.89% of the vaccine adjuvants market share in 2024, underpinned by mechanistically defined agents such as Dynavax’s CpG 1018 and GSK’s MPL. Regulatory familiarity with these pathways accelerates review timelines and fosters platform approvals across multiple antigens. Vehicle adjuvants, encompassing lipid nanoparticles and polymeric carriers, are projected to grow at 7.04% CAGR through 2030 as developers demand integrated delivery–stimulation solutions.
The vaccine adjuvants market increasingly values vehicles that co-encapsulate antigens and immunopotentiators, maintaining colloidal stability across temperature excursions. Recent manganese-lipid hybrid particles demonstrated stronger CD8+ responses against varicella-zoster versus alum comparators, highlighting functional gains that drive substitution waves. Carrier adjuvants maintain relevance for mucosal or slow-release applications, ensuring each modality retains a defined opportunity space within the broader vaccine adjuvants market.
By Disease Type: Cancer Applications Accelerate Beyond Infectious-Disease Base
Established infectious-disease programs represented 72.45% of the vaccine adjuvants market size in 2024, backed by government procurement for influenza, pneumococcal, and H5N1 reserves. Despite this dominance, oncology candidates will post a 7.06% CAGR to 2030 as neoantigen and tumor-associated antigen pipelines mature. Personalized vaccines demand adjuvants that orchestrate robust CD4+ and CD8+ activation while avoiding regulatory T-cell expansion, spurring exploration of STING agonists and TLR7/8 modulators.
Clinical readouts show adjuvanted melanoma vaccines achieving durable response rates when combined with checkpoint inhibitors, suggesting cross-portfolio synergies that elevate total addressable value. Beyond cancer, autoimmune desensitization and allergy prophylaxis remain exploratory territories, holding incremental upside for the vaccine adjuvants market if safety-efficacy trade-offs can be resolved.
Note: Segment shares of all individual segments available upon report purchase
By Application: Research Momentum Catalyzes Commercial Pipelines
Commercial supply accounted for 68.31% of the vaccine adjuvants market size in 2024, driven by influenza, pediatric combination, and travel-health franchises. However, research applications will outpace with a 7.34% CAGR, reflecting post-pandemic R&D budgets that prioritize rapid-response platforms. Grants from BARDA, CEPI, and Horizon Europe specifically earmark funds for adjuvant discovery, ensuring a steady influx of preclinical data that de-risks later commercial launches.
Academic–industry consortia now employ high-content screening, AI-assisted molecular design, and systems immunology to uncover novel adjuvant–antigen synergies, shortening lead-optimization cycles. This iterative feedback loop means today’s laboratory breakthroughs seed tomorrow’s licensed formulations, embedding a virtuous innovation cycle inside the vaccine adjuvants market.
Geography Analysis
North America preserved its leadership with 41.12% share in 2024, supported by mature manufacturing capacity, public-health procurement budgets, and FDA regulatory precedents that streamline platform reviews. Under Operation Warp Speed and successor initiatives, federal funding subsidizes scale-up of mRNA-optimized adjuvant systems, further entrenching regional dominance. Clustered academic centers in Boston, San Francisco, and Toronto forge translational pipelines that feed commercial portfolios, ensuring the vaccine adjuvants market remains anchored in the region.
Asia-Pacific is projected to record a 7.45% CAGR through 2030, propelled by China’s biopharma capacity additions, India’s contract-manufacturing economies of scale, and ASEAN immunization-program expansions. Government subsidies for thermostable adjuvant R&D address tropical cold-chain constraints, while Japan’s chemical-industry strength accelerates lipid-nanoparticle innovations. Local regulatory harmonization under ASEAN’s Vaccine Regulatory Mechanism reduces approval redundancies, improving speed-to-market for regional developers and elevating the vaccine adjuvants market in Asia-Pacific.
Europe maintains steady mid-single-digit growth as the EMA’s adaptive-pathways framework supports conditional licensing for priority adjuvant platforms. Cross-border procurement mechanisms under the EU Joint Procurement Agreement aggregate demand, giving suppliers predictable volumes while enabling price negotiations that preserve margin discipline. Chemical-specialty infrastructure in Germany and the Netherlands sustains high-purity excipient supply, supporting export of adjuvant intermediates to other regions.
Competitive Landscape
The vaccine adjuvants industry exhibits moderate concentration. GSK leverages its AS-series portfolio across shingles, malaria, and RSV programs, while Dynavax’s CpG platform anchors hepatitis B and COVID-19 offerings. Novavax pairs Matrix-M with VLP and recombinant constructs, demonstrating how proprietary adjuvants elevate antigen value. Collectively, the top five companies hold around 60–65% of global revenues, leaving a competitive corridor for mid-cap biotech entrants.
Strategic acquisitions reinforce platform control: Croda’s USD 185 million purchase of Avanti Polar Lipids secured high-grade lipids essential for mRNA products, whereas SK Bioscience’s USD 244 million absorption of IDT Biologika integrated fill-finish capacity with adjuvant manufacturing. Partnerships proliferate, with large pharmas licensing TLR7/8 or STING agonists from specialty firms to diversify pipeline risk. White-space opportunities center on thermostable nano-alum, personalized adjuvant selection based on HLA haplotypes, and combination formulations that synchronize innate-immune triggers.
Barriers to entry remain significant due to regulatory complexity, toxicology costs, and supply-chain validation, yet venture investment into first-in-class mechanisms signals sustained appetite for disruptive differentiation. Companies able to prove both immunological superiority and cost-effective scalability are positioned to capture incremental share as the vaccine adjuvants market expands toward 2030.
Vaccine Adjuvants Industry Leaders
-
Adjuvatis
-
Merck KGaA
-
GlaxoSmithKline plc
-
Novavax Inc.
-
Croda International plc
- *Disclaimer: Major Players sorted in no particular order
Recent Industry Developments
- June 2025: University of Pittsburgh and Pennsylvania State University reported trans-amplifying mRNA constructs requiring 40× less mRNA than conventional formats, cutting raw-material costs and easing manufacturing bottlenecks.
- June 2025: Wageningen University introduced BacFreets processing, eliminating 99.97% baculovirus contamination in VLP production, enhancing commercial scalability.
- June 2025: ACIP endorsed phasing out thimerosal-containing influenza shots and approved clesrovimab monoclonal antibody for RSV prophylaxis, signaling evolving safety standards.
- May 2025: Cornell University scientists developed poly(carboxybetaine) lipids that substitute PEG in mRNA nanoparticles, reducing anti-PEG antibody formation and enabling repeat dosing.
Global Vaccine Adjuvants Market Report Scope
As per the scope of the report, an adjuvant is a pharmacological or immunological agent that helps synergize and direct the adaptive immune response to a vaccine agent. Adjuvants are compounds that improve the body's immunogenic reaction against antigens. Therefore, adjuvants are majorly added to vaccines to augment their capability to ensure long-term protection and safety against any infection.
The vaccine adjuvants market is segmented by product type (mineral salt-based adjuvants, tensoactive adjuvants, adjuvant emulsions, liposome adjuvants, carbohydrate adjuvants, bacteria-derived adjuvants, virus-like particles (vlp), and other product types), usage type (active immunostimulants, carriers, and vehicle adjuvants), disease type ( infectious disease and cancer), application (research applications and commercial applications), and geography (North America, Europe, Asia-Pacific, Middle East and Africa, and South America). The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD) for the above segments.
| Mineral-salt Adjuvants |
| Saponin and Triterpenoid |
| Emulsion-based |
| Liposome and Virosome |
| Carbohydrate & Polysaccharide |
| Bacteria-derived TLR agonists |
| Virus-Like Particles |
| Other Product Types |
| Active Immunostimulants |
| Carriers |
| Vehicle Adjuvants |
| Infectious Diseases |
| Cancer |
| Others |
| Research Applications |
| Commercial Applications |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product Type | Mineral-salt Adjuvants | |
| Saponin and Triterpenoid | ||
| Emulsion-based | ||
| Liposome and Virosome | ||
| Carbohydrate & Polysaccharide | ||
| Bacteria-derived TLR agonists | ||
| Virus-Like Particles | ||
| Other Product Types | ||
| By Usage Type | Active Immunostimulants | |
| Carriers | ||
| Vehicle Adjuvants | ||
| By Disease Type | Infectious Diseases | |
| Cancer | ||
| Others | ||
| By Application | Research Applications | |
| Commercial Applications | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||
Key Questions Answered in the Report
What is the current value of the vaccine adjuvants market?
The market is valued at USD 2.38 billion in 2025 and is forecast to reach USD 3.24 billion by 2030.
Which product category leads the vaccine adjuvants market?
Saponin and triterpenoid systems dominate with 26.78% share in 2024, reflecting widespread use in shingles and malaria vaccines.
What segment is growing fastest within the vaccine adjuvants market?
Virus-like particle adjuvants show the quickest rise, registering a 7.12% CAGR to 2030 due to scalable nanostructure platforms.
Which region is advancing most rapidly?
Asia-Pacific is projected to grow at a 7.45% CAGR as China, India, and Southeast Asia expand immunization programs and local manufacturing.
Why are adjuvants crucial for mRNA vaccines?
MRNA constructs require delivery particles that ensure endosomal escape and balanced innate activation; optimized adjuvants boost antigen expression while moderating inflammation.
How concentrated is the competitive landscape?
The top five companies control a bit more than 60% of revenue, producing a moderate concentration level that still leaves room for disruptive entrants.
Page last updated on:



