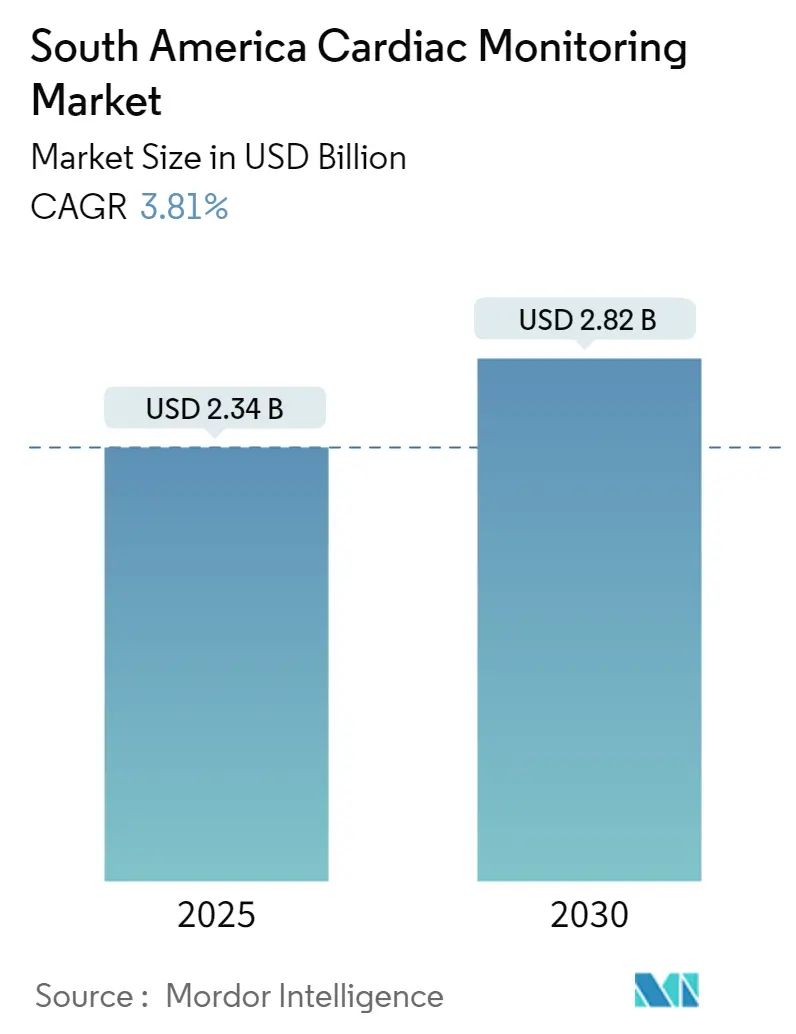
South America Cardiac Monitoring Market Analysis by Mordor Intelligence
The South America Cardiac Monitoring Market size is estimated at USD 2.34 billion in 2025, and is expected to reach USD 2.82 billion by 2030, at a CAGR of 3.81% during the forecast period (2025-2030).
The outbreak of the COVID-19 pandemic has pushed the healthcare industry into action, with a race to develop both therapeutic and preventive drugs. Exponentially increasing cases of the coronavirus worldwide are leading to the need for the development of novel treatments, with several clinical trials underway. Initially, the pandemic imposed an adverse effect on the market in Latin America. For instance, as per the study titled, "The Impact of COVID-19 on Diagnosis of Heart Disease in Latin America an INCAPS COVID Sub-analysis" published in April 2022, COVID-19 was associated with a significant decrease in cardiac diagnostic procedures in South America, which was mainly related to social distancing rather than the incidence of COVID-19. It also stated that the pandemic has disrupted the delivery of care for cardiovascular diseases in South America. Similarly, as per the study titled, "International Impact of COVID-19 on the Diagnosis of Heart Disease" published in January 2021, globally, cardiac diagnostic procedure volumes decreased by 42.0% from March 2019 to March 2020 and by 64.0% from March 2019 to April 2020 with which the greatest regional decreases was in South America. Furthermore, according to the study titled, "Physicians' perceptions on the impact of COVID-19 in coronary artery disease diagnostic imaging and treatment: A Latin America survey. For the Society of Cardiovascular Imaging of the Inter-American Society of Cardiology" published in June 2022, the pandemic severely impacted the clinical management of coronary artery diseases in South America. Therefore, such instances indicate that the market witnessed a slow growth during the pandemic. However, with the ease of restrictions and lockdown, the market is expected to gain momentum, thereby considerable growth is anticipated over the forecast period.
The rise in the incidences of cardiovascular diseases and investments in research and development for product development and demand for home based, point of care & remote monitoring cardiac systems are the major drivers for the market. For instance, according to the study titled, "Evaluation of cardiometabolic profile in Health Professionals of Latin America" published in August 2021, the prevalence of risk factors for cardiovascular diseases among the South American physicians was relatively high. It also reported that, the prevalence of high-risk scores for cardiovascular diseases and diabetes mellitus was high and healthy lifestyle habits were low. Similarly, as per the study titled, "Epidemiology of Hypertension and Diabetes Mellitus in Latin America" published in February 2021, in South America, hypertension, prediabetes, and type 2 diabetes mellitus prevalence range varies from 30.0% to 50.0%, 6.0% to 14.0%, and 8.0% to 13.0% respectively . It also reported that the proportion of awareness, treatment and control of hypertension was very low in Latin America. All these conditions are associated with increased risk of cardiovascular diseases owing to which such instances are anticipated to propel the cases of cardiovascular diseases. Since, cardiac monitoring devices are one of the essential requirements for the diagnosis of cardiovascular disease, therefore, significant market growth is expected over the forecast period.
However, risk of infection due to implantable monitoring devices is anticipated to hinder the market growth over the forecast period.
South America Cardiac Monitoring Market Trends and Insights
Hospitals and Clinics is Expected to Witness Growth Over the Forecast Period
The majority of cardiovascular diseases are treated in hospitals and clinics. The ECG Monitors, event recorders, implantable cardiac loop recorders, pacemakers, defibrillators, cardiac resynchronization therapy (CRT) devices, and smart wearables are generally used in hospitals and clinics for recovering patients suffering from cardiovascular diseases.
The outbreak of the pandemic has increased the in-hospital mortality of patients suffering from cardiac arrest, especially among patients who have suffered from COVID-19. According to the study titled, 'A Multicenter Evaluation of Survival After In-Hospital Cardiac Arrest in Coronavirus Disease 2019 Patients' published in May 2021, in-hospital cardiac arrest survival among COVID-19 patients has been reported to range from 0.0% to 12.0% in South America. Similarly, according to the study titled, 'Effects of COVID-19 on in-hospital Cardiac Arrest: Incidence, Causes, and Outcome - a Retrospective Cohort Study' published in February 2021, hospital admissions declined during the pandemic, but a higher incidence of in-hospital cardiac arrest was observed. It also reported that in-hospital cardiac arrest in patients with COVID-19 was a common phenomenon compared to patients with non-COVID-19-related respiratory failure.
Therefore, increasing in-hospital cardiac arrest cases in South America is anticipated to increase the demand for cardiac monitoring devices, as these are one of the essential requirements for recovering patients from cardiovascular diseases, thereby considerable segment growth is expected over the forecast period.
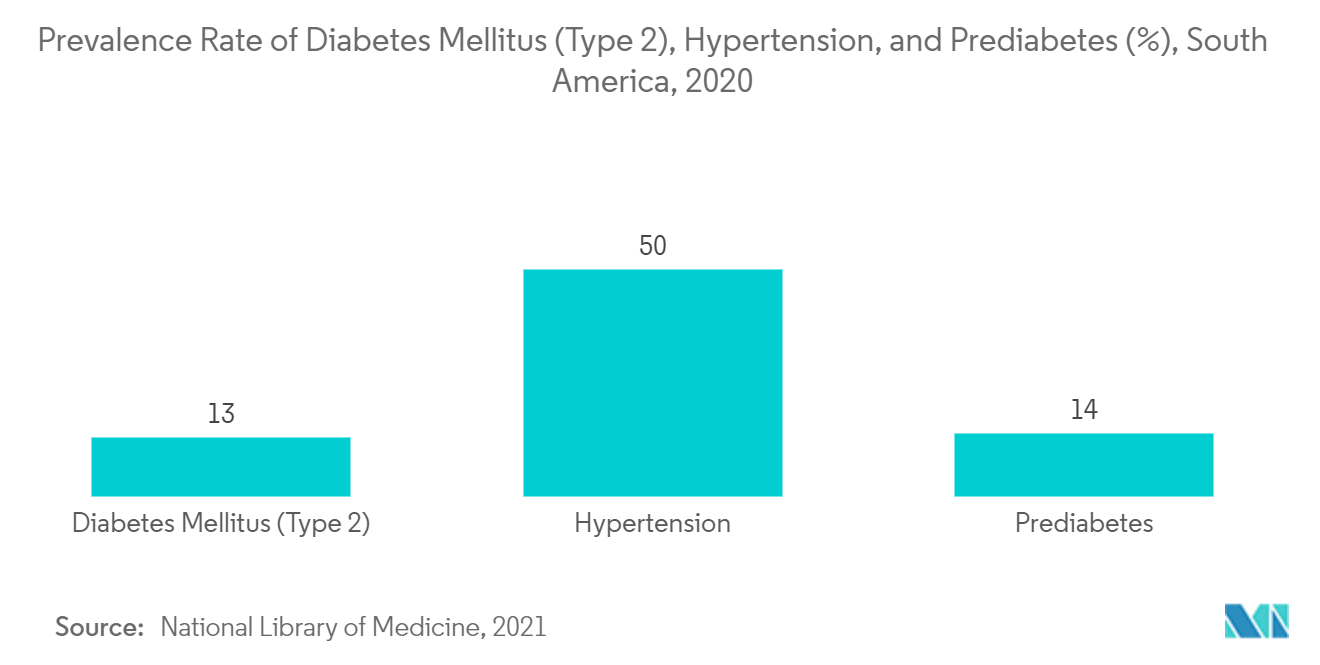


Brazil is Expected to Dominate the South America Cardiac Monitoring Market Over the Forecast Period
Brazil is expected to dominate the overall South America cardiac monitoring market, over the forecast period. The growth is due to factors such as the rising cases of cardiovascular diseases, diabetes, and hypertension. For instance, as per the study titled, "Characterization of Brazilian hospital admissions due to cardiovascular diseases: a longitudinal study" published in June 2020, it is estimated that around three-quarters of all deaths related to cardiovascular diseases occur in low and middle-income countries such as Brazil. It also stated that cardiovascular diseases were responsible for more than 10.0% of all hospital admissions in Brazil.
Furthermore, as per the PLOS journal published in September 2021, cardiovascular diseases represent a significant public health problem, as they are the leading cause of death and disability affecting adults of full working age, accounting for about 27.7% of deaths in Brazil. Similarly, as per the report published by the International Diabetes Federation in 2021, the number of people living with diabetes per 1,000 population in Brazil was around 15,733.6 in 2021.
Therefore, due to the abovementioned factors, the market is anticipated to witness considerable growth over the forecast period.


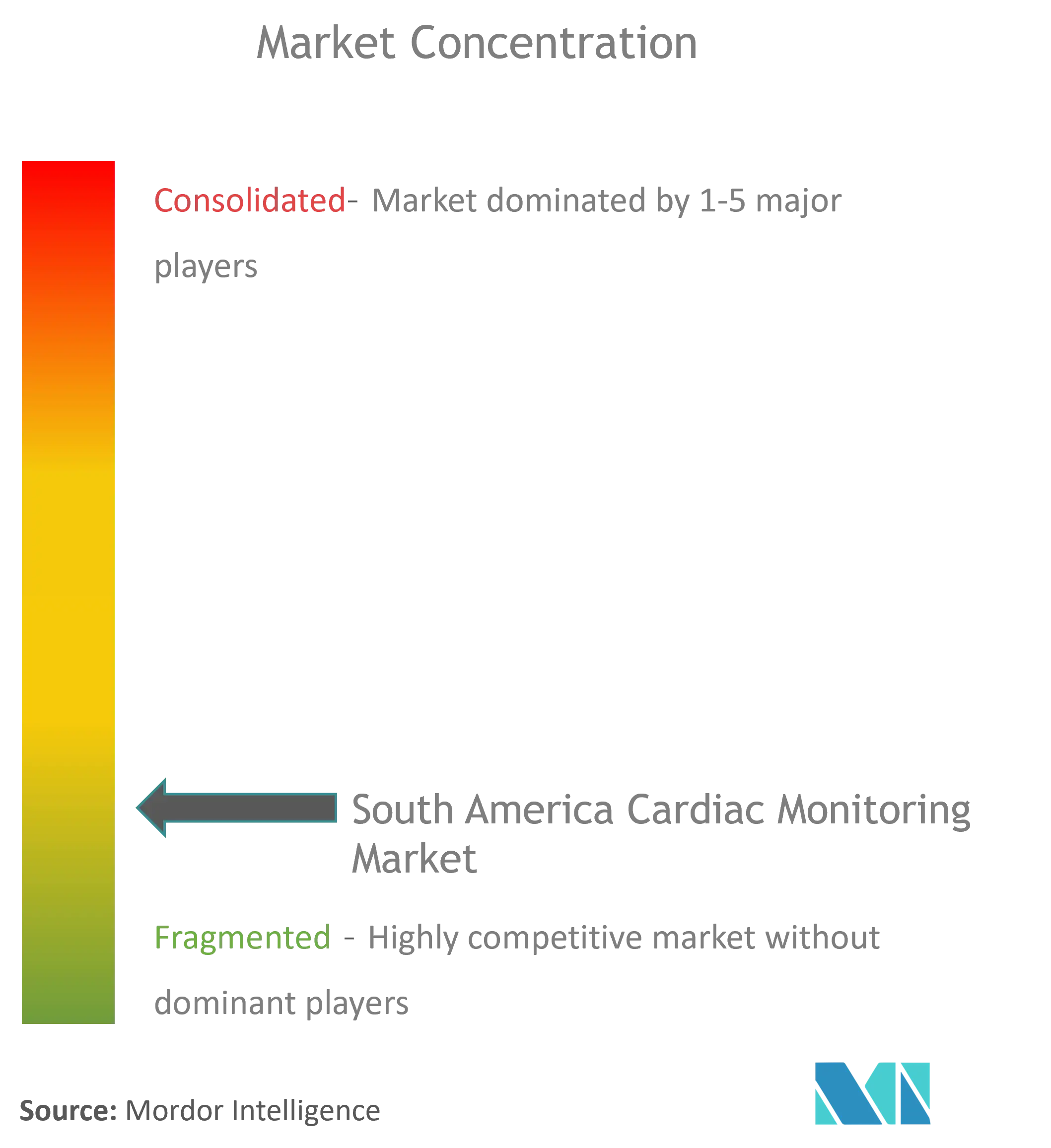
Competitive Landscape
The market is fragmented and focused because of the presence of many large, medium, and small players. The multinational players possess a substantial share in the region. The competitive landscape includes an analysis of a few international as well as local companies which hold the market shares and are well known including Medtronic PLC, GE Healthcare, Biotronik, and Boston Scientific Corporation among others.
South America Cardiac Monitoring Industry Leaders
Medtronic PLC
GE Healthcare
Boston Scientific Corporation
Phillips Healthcare
Biotronik
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- In July 2022, The researchers from the South American and Caribbean Cohort Consortium (CC-LAC) developed a risk algorithm specific to the Latin American and Caribbean region. These equations include information such as age, cholesterol level, and blood pressure, among others, in order to provide the probability that a person will have cardiovascular disease in the next 10 years.
- In July 2022, Medtronic's smallest pacemakers, Micra AV and Micra VR Pacemakers were launched in Brazil.


South America Cardiac Monitoring Market Report Scope
As per the scope of the report, cardiac monitoring devices are used for the observation of cardiac activity. Continuous monitoring of the heart is mandatory to assess the patient's condition suffering from cardiovascular diseases. Cardiac monitoring devices record and display pressure and electrical waveforms of the cardiovascular system for measurement and treatment. The South America Cardiac Monitoring Market is segmented by Device Type (ECG Monitor, Event Recorder, Implantable Cardiac Loop Recorder, Pacemaker, Defibrillator, Cardiac Resynchronization Therapy (CRT) Devices, Smart Wearable, Other Device Types), End Users (Hospitals and Clinics, Home Care Settings, and Other End Users), and Geography (Brazil, Argentina, and Rest of South America). The report offers the value (in USD million) for the above segments.
| ECG Monitor |
| Event Recorder |
| Implantable Cardiac Loop Recorder |
| Pacemaker |
| Defibrillator |
| Cardiac Resynchronization Therapy (CRT) Devices |
| Smart Wearable |
| Other Device Types |
| Hospitals and Clinics |
| Hospitals and Clinics |
| Others End-users |
| Brazil |
| Argentina |
| Rest of South America |
| By Device Type | ECG Monitor |
| Event Recorder | |
| Implantable Cardiac Loop Recorder | |
| Pacemaker | |
| Defibrillator | |
| Cardiac Resynchronization Therapy (CRT) Devices | |
| Smart Wearable | |
| Other Device Types | |
| By End Users | Hospitals and Clinics |
| Hospitals and Clinics | |
| Others End-users | |
| Geography | Brazil |
| Argentina | |
| Rest of South America |


Key Questions Answered in the Report
How big is the South America Cardiac Monitoring Market?
The South America Cardiac Monitoring Market size is expected to reach USD 2.34 billion in 2025 and grow at a CAGR of 3.81% to reach USD 2.82 billion by 2030.
What is the current South America Cardiac Monitoring Market size?
In 2025, the South America Cardiac Monitoring Market size is expected to reach USD 2.34 billion.
Who are the key players in South America Cardiac Monitoring Market?
Medtronic PLC, GE Healthcare, Boston Scientific Corporation, Phillips Healthcare and Biotronik are the major companies operating in the South America Cardiac Monitoring Market.
What years does this South America Cardiac Monitoring Market cover, and what was the market size in 2024?
In 2024, the South America Cardiac Monitoring Market size was estimated at USD 2.25 billion. The report covers the South America Cardiac Monitoring Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the South America Cardiac Monitoring Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
South America Cardiac Monitoring Market Report
Statistics for the 2025 South America Cardiac Monitoring market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. South America Cardiac Monitoring analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.





