Neurothrombectomy Devices Market Size and Share
Neurothrombectomy Devices Market Analysis by Mordor Intelligence
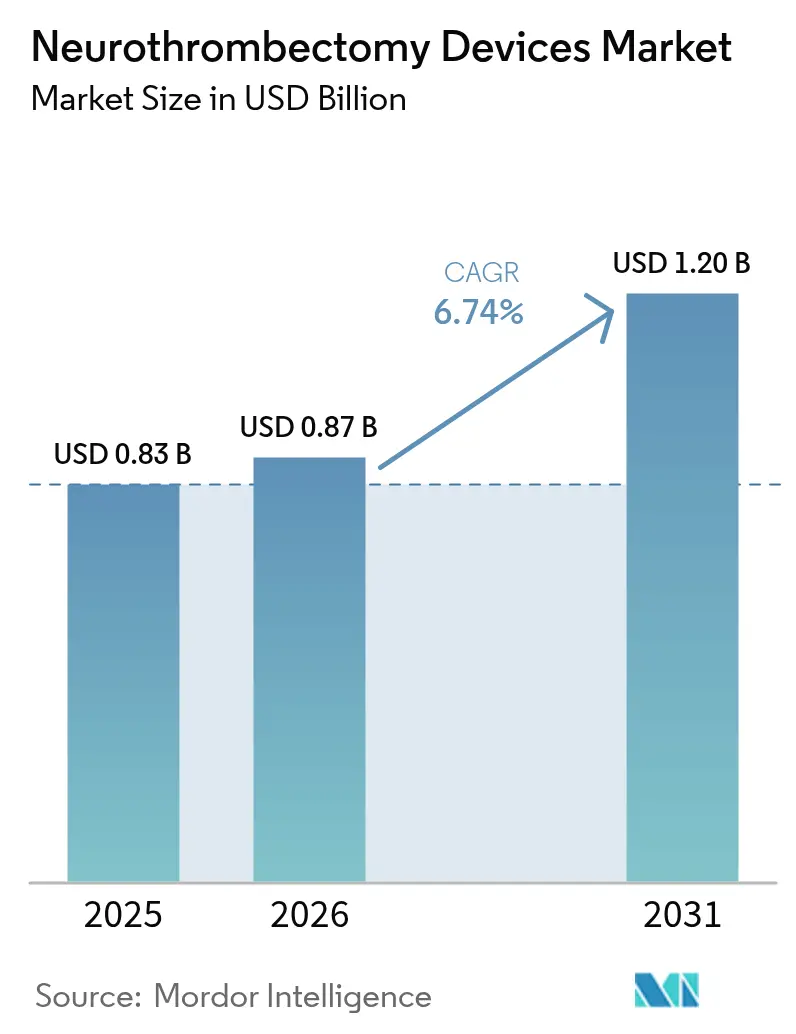
The Neurothrombectomy Devices Market size is projected to be USD 0.83 billion in 2025, USD 0.87 billion in 2026, and reach USD 1.20 billion by 2031, growing at a CAGR of 6.74% from 2026 to 2031.
This steady climb reflects a maturing technology cycle that followed the post-DAWN trial surge, with hospitals now optimizing patient selection through automated perfusion imaging and streamlined hub-and-spoke networks. Aspiration techniques that lower fluoroscopy time and reduce device exchanges are gaining traction among U.S. and European operators, while China’s mandate for tertiary stroke-center accreditation is lifting procedure volumes across more than 2,000 hospitals. AI-driven triage platforms such as Viz.ai and RapidAI compress door-to-puncture times by roughly 20 minutes, helping community hospitals meet guideline-recommended treatment windows. At the same time, reimbursement certainty under Medicare MS-DRG 023-027, category-C1 coverage in Japan, and harmonized European Stroke Organisation guidelines continue to make large-vessel occlusion treatment financially viable for care providers.[1]Centers for Disease Control and Prevention, “Stroke Facts,” cdc.gov
Key Report Takeaways
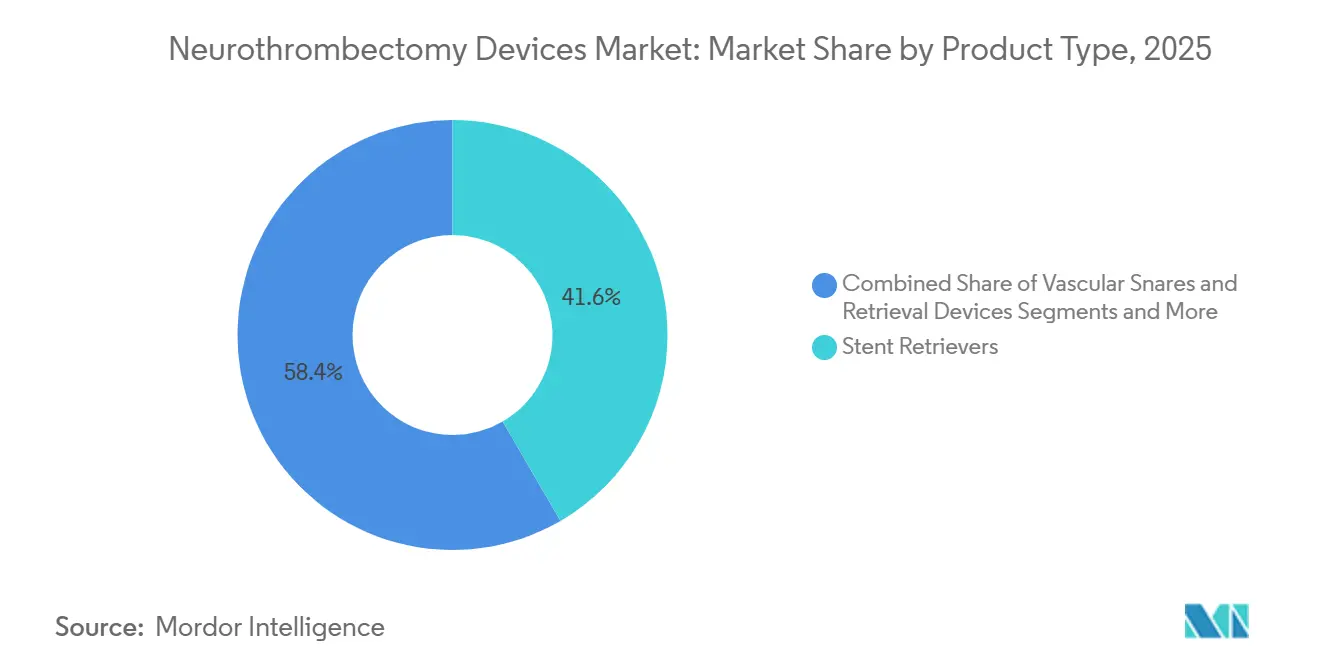
- By product type, stent retrievers led with 41.62% revenue share in 2025, while aspiration devices are forecast to expand at a 10.13% CAGR to 2031.
- By access route, trans-femoral techniques held 76.13% of the neurothrombectomy devices market share in 2025, but trans-radial and brachial approaches are pacing at a 9.34% CAGR through 2031.
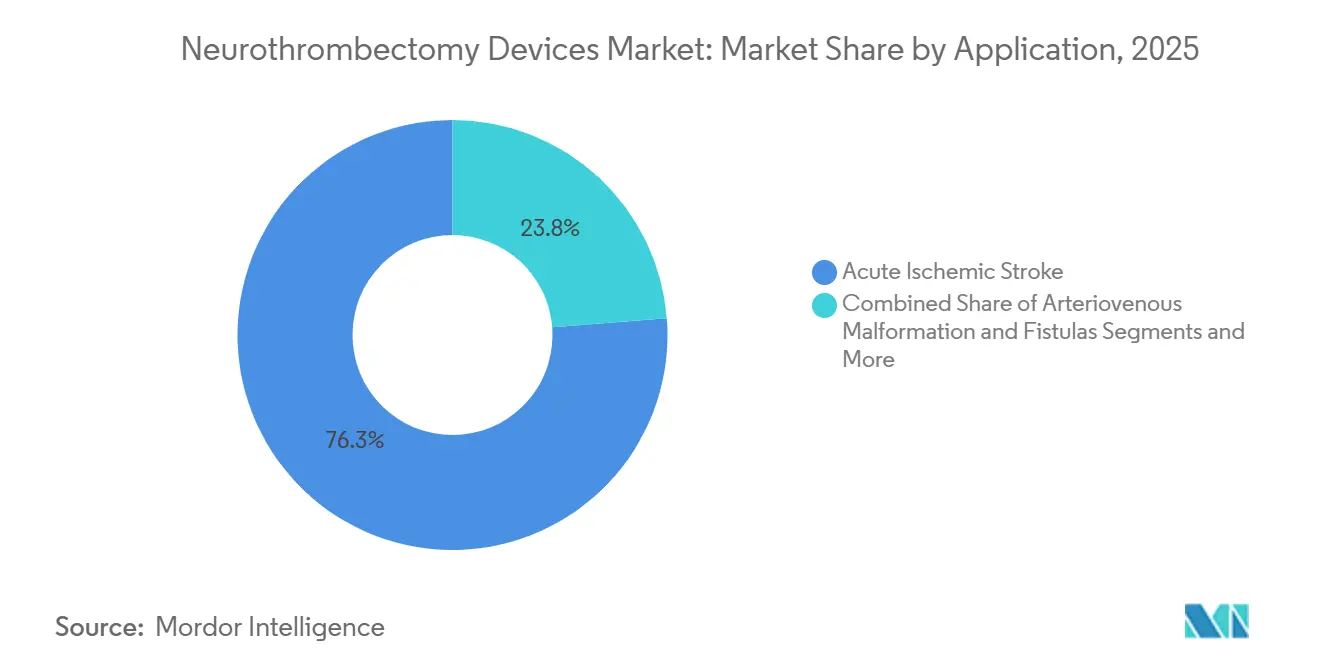
- By application, acute ischemic stroke accounted for 76.25% of the neurothrombectomy devices market size in 2025, whereas arteriovenous malformations and fistulas are advancing at an 8.64% CAGR to 2031.
- By treatment window, procedures within 6 hours held 66.91% share in 2025, yet the 6-to-24-hour cohort is growing at a 10.35% CAGR during the forecast period.
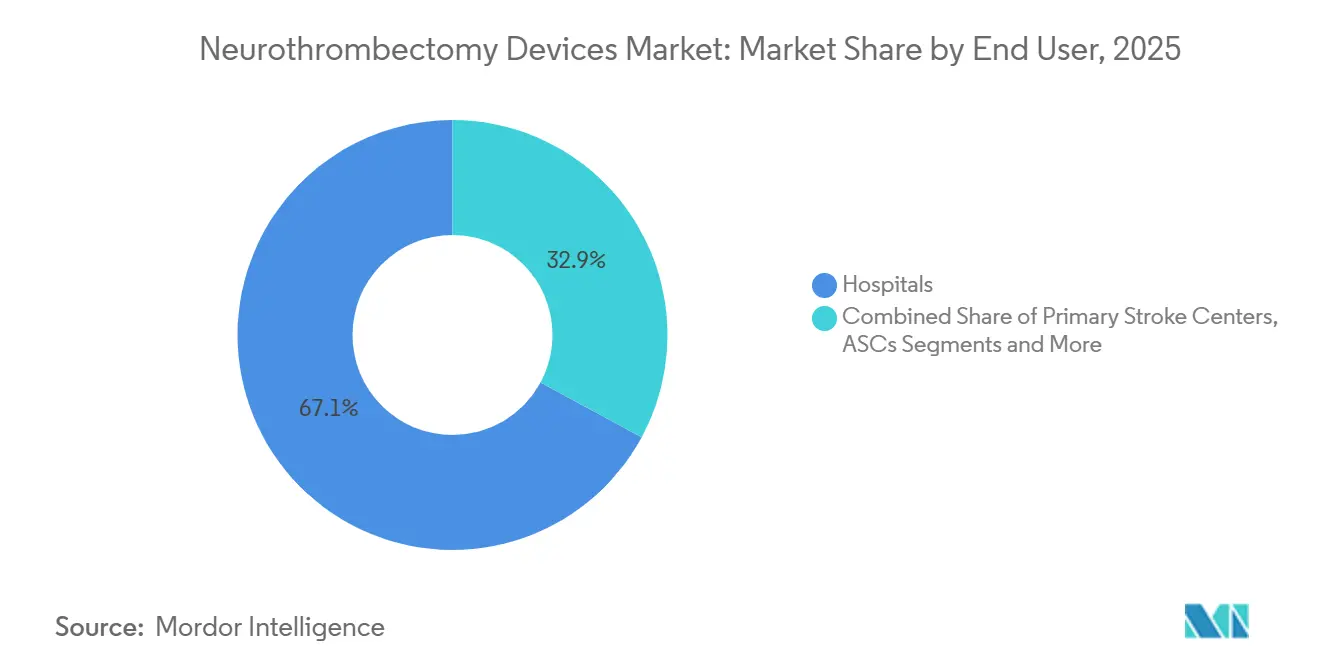
- By end user, hospitals commanded 67.12% share in 2025; ambulatory surgical centers deliver the fastest growth at an 8.13% CAGR through 2031.
- By geography, North America captured 39.13% of 2025 revenue, while Asia-Pacific is on course for the highest regional CAGR at 8.12% to 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Neurothrombectomy Devices Market Trends and Insights
Driver Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising Global Incidence of Acute Ischemic Stroke | +1.2% | APAC, Europe, North America | Long term (≥ 4 years) |
| Expansion of Treatment Guidelines and Reimbursement Coverage | +1.5% | North America, Western Europe | Medium term (2-4 years) |
| Technological Advances in Stent Retrievers and Aspiration Catheters | +1.0% | Global | Medium term (2-4 years) |
| Growing Adoption in Emerging Markets | +1.3% | China, India, Latin America | Long term (≥ 4 years) |
| AI-Enabled Stroke Triage Platforms | +0.9% | North America, Western Europe | Short term (≤ 2 years) |
| Telestroke and Remote Proctoring | +0.6% | Rural U.S., Europe, India | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Rising Global Incidence of Acute Ischemic Stroke
The World Health Organization logged 11.95 million strokes in 2021, 87% of which were ischemic, and aging populations in Japan, Germany, and the United States continue to nudge the caseload upward by 2-3% each year.[2]World Health Organization, “Stroke Statistics,” who.int China alone records nearly 3 million new events annually, while India’s incidence rises 4% per year as urban lifestyles fuel hypertension and diabetes. These epidemiological pressures stretch thrombolysis capacity, given that tissue plasminogen activator fails to recanalize 60-70% of large-vessel occlusions. Health systems are therefore prioritizing neurothrombectomy suite build-outs, expanding eligibility to late presenters, and investing in specialist training programs to keep pace with demand.
Expansion of Treatment Guidelines and Reimbursement Coverage
Updated 2026 AHA/ASA guidelines endorse mechanical thrombectomy up to 24 hours after the last known well time when perfusion imaging shows salvageable penumbra.[3]American Heart Association, “2026 Guideline for the Early Management of Patients With Acute Ischemic Stroke,” ahajournals.org Medicare reimburses these procedures under MS-DRG 023-027 at USD 7,200-8,500, and similar policy shifts in Germany, France, and Japan cut hospital payback times on biplane angiography equipment to under five years. Coverage clarity encourages capital spending and helps smaller community hospitals justify staff expansion, moving the neurothrombectomy devices market toward broader geographic penetration.
Technological Advances in Stent Retrievers and Aspiration Catheters
Penumbra’s Lightning Flash 2.0 and Medtronic’s React 71 aspiration lines introduce variable-stiffness shafts and vacuum-assist pumps that trim median procedure times below 40 minutes. Cerenovus’ Cereglide 71 intermediate catheter features a low-profile distal tip that tackles tortuous intracranial anatomy without vessel trauma. These refinements reduce distal embolization risk, cut contrast use, and lower anesthesia exposure, while the 510(k) pathway for aspiration devices lets manufacturers iterate faster than PMA-regulated stent retrievers.
Growing Adoption in Emerging Markets
China requires every tertiary hospital to run a certified stroke center, propelling device uptake across 2,000 sites, while domestic vendors price retrievers 40-50% below Western imports. India’s telestroke networks link rural district hospitals to urban specialists, and Brazil’s Unified Health System has started paying for thrombectomy in public centers. These initiatives expand the addressable neurothrombectomy devices market, although workforce shortages and fragmented supply chains temper the pace.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High Device and Procedure Costs in Low-Resource Settings | –0.8% | Sub-Saharan Africa, South Asia | Long term (≥ 4 years) |
| Stringent Regulatory and Clinical-Trial Requirements | –0.5% | North America, EU | Medium term (2-4 years) |
| Shortage of Neuro-Interventional Specialists in Rural Areas | –0.6% | Rural U.S., Eastern Europe, India | Long term (≥ 4 years) |
| Supply-Chain Fragility for Nitinol and Polymer Components | –0.4% | North America, Europe | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
High Device and Procedure Costs in Low-Resource Settings
A single thrombectomy can cost USD 15,000-40,000, exceeding the annual per-capita health spend in South Asia by up to 200 times. India’s Ayushman Bharat covers only USD 1,800 per case, forcing hospitals either to forgo treatment or absorb losses. Similar gaps exist across sub-Saharan Africa and rural Latin America, where import tariffs and currency volatility further inflate prices, limiting neurothrombectomy access to donor-funded pilots.
Restraint Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High device & procedure cost in emerging markets | -1.4% | APAC, South America, MEA | Medium term (2-4 years) |
| Shortage of trained neuro-interventionists outside urban centers | -1.1% | Global, particularly acute in rural areas | Long term (≥ 4 years) |
| Supply-chain vulnerability for nitinol & platinum-iridium alloys | -0.9% | Global, with acute impact on specialized manufacturers | Short term (≤ 2 years) |
| Reimbursement delays for posterior-circulation thrombectomies | -0.7% | North America & Europe, selective payer policies | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Stringent Regulatory and Clinical-Trial Requirements
The FDA’s 21 CFR 882.5600 classification mandates PMA filings backed by randomized trials enrolling at least 200 patients, a hurdle that inflates per-patient trial cost to USD 50,000. Europe’s Medical Device Regulation adds post-market audit layers that can stretch certification timelines to three years, disadvantages small innovators, and slows the release cadence of next-generation retrieval architectures.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Product Type: Aspiration Momentum Picks Up
Stent retrievers secured 41.62% of 2025 revenue, yet aspiration catheters are advancing at a 10.13% CAGR, nearly double the overall neurothrombectomy devices market rate. Lower exchange counts and shorter fluoroscopy times make direct aspiration USD 2,000-3,000 cheaper per case. Combined aspiration-stent systems are emerging but await long-term outcome data. Balloon guide catheters maintain steady demand because proximal flow arrest minimizes emboli, whereas vascular snares remain a niche rescue option.
Second-generation aspiration platforms such as Medtronic React and Stryker Catalyst use graduated shaft stiffness for pushability while keeping soft distal tips. The ADAPT protocol now guides about 40% of U.S. thrombectomy first passes, though stent retrievers still dominate distal M2/M3 cases where small vessels hamper catheter delivery. Faster 510(k) cycles encourage more new entrants, intensifying price competition and broadening the neurothrombectomy devices market choice set.

By Access Route: Radial Techniques Challenge Femoral Dominance
Trans-femoral access held 76.13% share in 2025, sustained by larger vessel caliber and operator familiarity, yet radial and brachial entry sites are climbing at a 9.34% CAGR. Radial access cuts groin complications and supports same-day discharge, a selling point for ambulatory centers. Direct carotid puncture serves <3% of cases but grows 8-9% where arch tortuosity blocks traditional routes.
Learning curves of 20-30 cases elevate fluoroscopy times early on, but Japanese registry data show parity with femoral success once skills mature. Slender 6-French guide systems can limit inner lumen for large aspiration catheters, occasionally forcing access crossover mid-procedure, yet patient-reported comfort scores favor radial entry.
By Application: Off-Label Uses Gain Ground
Acute ischemic stroke contributed 76.25% of 2025 procedures, driven by a persistent global caseload near 12 million events annually. Off-label uses in arteriovenous malformations and fistulas expand at an 8.64% CAGR as operators employ aspiration catheters to clear intraprocedural thrombus. Cerebral aneurysm thrombosis, while rare, leverages retrieval devices to protect distal territories when sac clotting threatens flow.
Large-core trials such as SELECT2 and ANGEL-ASPECT enrolled >2,000 patients and widened eligibility to ASPECTS 3-5, enlarging the neurothrombectomy devices market size without inflating stroke incidence. Basilar artery evidence remains mixed, so most payers reimburse under existing stroke DRGs rather than create new codes.

By Treatment Window: Imaging Extends Eligibility
Procedures inside 6 hours still account for 66.91% of volume, reflecting early-trial legacies, yet 6-to-24-hour cases are growing 10.35% per year thanks to CT perfusion and automated mismatch software. Wake-up strokes constitute a smaller but rising cohort because collateral-rich tissue may stay salvageable well past 24 hours.
RapidAI and Brainomix generate perfusion maps within five minutes, enabling community hospitals to triage late presenters without on-site neuroradiologists. Extended-window growth is faster in low- and middle-income countries where transport delays make early arrival rare, whereas the sub-6-hour segment in high-income regions edges toward saturation.
By End User: ASC Models Emerge
Hospitals controlled 67.12% of 2025 spending, justified by USD 2-3 million biplane labs and on-site neuro-ICUs. Ambulatory surgical centers are advancing 8.13% annually, treating NIHSS <10 patients under conscious sedation and discharging within 23 hours. However, CMS approval for ASC thrombectomy remains state-dependent, and liability insurers demand higher cover because intensive care beds are off-site.
Device makers now bundle guide catheters, aspiration systems, and hemostasis patches in single-use trays tailored for ASCs, shrinking setup time and inventory costs. Primary stroke centers continue to act mainly as referring hubs, stabilizing patients and organizing transport to thrombectomy-capable facilities.

Geography Analysis
North America held 39.13% share in 2025, anchored by 1,600 neurointerventionalists, 1,500+ thrombectomy-capable centers, and robust Medicare reimbursement. Canada’s provincial stroke networks lift access yet still perform about 30% fewer procedures per capita than the United States because of longer imaging queues. Mexico presents a two-tier structure: private hospitals in major metros match U.S. standards, while public facilities lack specialists, leaving up to 80% of candidates untreated.
Asia-Pacific is the fastest-growing region at an 8.12% CAGR, underpinned by China’s nationwide stroke-center mandate and India’s hub-and-spoke telestroke rollouts. Chinese device makers already own ~25% domestic share through prices that undercut imports by 40-50%, pressuring multinationals to localize production. Japan’s aging society sustains roughly 300,000 strokes yearly, with local firms Asahi Intecc and Terumo supplying anatomy-specific microcatheters, while India’s penetration lingers below 2% because only 200 specialists serve 1.4 billion people.
Europe led by Germany, France, and the United Kingdom, each treating 15-20% of large-vessel occlusions. Updated 2024 ESO guidelines narrowed cross-border practice gaps, and EU structural funds help Poland, the Czech Republic, and Hungary add new labs at 9-10% annual growth. Southern regions like Andalusia and Sicily remain underserved due to specialist shortages, although GCC states in the Middle East are investing aggressively in Western-staffed comprehensive centers. Sub-Saharan Africa represents <1% share because device costs far exceed national health budgets.
Competitive Landscape
The four key players includes Medtronic, Stryker, Penumbra, and Cerenovus. Medtronic booked USD 9.846 billion in FY2025 neuroscience revenue and bundles Solitaire X retrievers with React aspiration and Pipeline flow-diversion stents under volume contracts. Stryker generated USD 1.307 billion in 2024 neurovascular sales and added hemorrhage technology via its 2024 buyout of NICO Corporation. Penumbra still leads aspiration innovation with the Lightning platform, while Cerenovus exploits Johnson & Johnson’s supply chain to push EmboTrap and Cereglide in Europe and Asia.
Start-ups are exploiting patent expiries on early retrievers. Imperative Care raised USD 260 million to commercialize the Zoom combined system that cut procedure times 25% in its IMPERATIVE trial. Rapid Medical’s Comaneci flow-arrest device and Perflow’s ANAIS funnel catheter tackle niche clot morphologies. FDA PMA costs and MDR audits favor capital-rich incumbents, yet Asian biosimilar contenders now market retrievers at half Western prices in cost-sensitive regions, testing brand loyalty and compressing margins across the neurothrombectomy devices market.
Neurothrombectomy Devices Industry Leaders
Medtronic plc
Stryker Corporation
Johnson & Johnson
Penumbra, Inc.
Terumo Corporation
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2026: Vesalio secured CE-Mark for NeVa VS vasospasm device and the NeVa 3.0 mm Thrombectomy System, while its aspiration catheters gained expanded 510(k) clearance for distal access
- January 2026: Terumo scheduled the domestic launch of its Kanshas drug-coated balloon for lower-limb arteries after Category C1 reimbursement approval.
- December 2025: India’s CDSCO green-lit Gravity Medical’s Supernova stent retriever based solely on domestic trials at AIIMS Delhi, marking the country’s first homegrown stroke device approval.
Global Neurothrombectomy Devices Market Report Scope
Neurothrombectomy devices are medical tools designed to remove or break up blood clots in the brain's cerebral vasculature during acute ischemic strokes, restoring blood flow through mechanical, laser, or ultrasound technology.
The Neurothrombectomy Devices Market Report is segmented by Product Type, Access Route, Application, Treatment Window, End User, and Geography. By Product Type, the market is segmented into Stent Retrievers, Aspiration Devices, Combined Systems, Balloon Guide Catheters, and Vascular Snares. By Access Route, the market is segmented into Trans-femoral, Trans-radial/Brachial, and Direct Carotid approaches. By Application, the market is segmented into Acute Ischemic Stroke, Cerebral Aneurysm Thrombosis, and AVMs/Fistulas. By Treatment Window, the market is segmented into ≤6 Hours, 6–24 Hours, and >24 Hours. By End User, the market is segmented into Hospitals, Primary Stroke Centers, ASCs, and Emergency Clinics. By Geography, the market is segmented into North America, Europe, Asia-Pacific, MEA, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. Market Forecasts are Provided in Terms of Value (USD).
| Stent Retrievers |
| Aspiration Devices |
| Combined Aspiration–Stent Systems |
| Balloon Guide Catheters |
| Vascular Snares & Retrieval Devices |
| Trans-femoral |
| Trans-radial / Brachial |
| Direct Carotid / Percutaneous |
| Acute Ischemic Stroke |
| Cerebral Aneurysm Thrombosis |
| Arteriovenous Malformations & Fistulas |
| ≤ 6 Hours From Symptom Onset |
| > 6 to 24 Hours (Extended Window) |
| > 24 Hours (Late-Presenter / Wake-Up Stroke) |
| Hospitals |
| Primary Stroke Centers |
| Ambulatory Surgical Centers |
| Emergency & Specialty Clinics |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| France | |
| United Kingdom | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| South Korea | |
| Australia | |
| Rest of Asia-Pacific | |
| Middle East & Africa | GCC |
| South Africa | |
| Rest of Middle East & Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product Type | Stent Retrievers | |
| Aspiration Devices | ||
| Combined Aspiration–Stent Systems | ||
| Balloon Guide Catheters | ||
| Vascular Snares & Retrieval Devices | ||
| By Access Route | Trans-femoral | |
| Trans-radial / Brachial | ||
| Direct Carotid / Percutaneous | ||
| By Application | Acute Ischemic Stroke | |
| Cerebral Aneurysm Thrombosis | ||
| Arteriovenous Malformations & Fistulas | ||
| By Treatment Window | ≤ 6 Hours From Symptom Onset | |
| > 6 to 24 Hours (Extended Window) | ||
| > 24 Hours (Late-Presenter / Wake-Up Stroke) | ||
| By End User | Hospitals | |
| Primary Stroke Centers | ||
| Ambulatory Surgical Centers | ||
| Emergency & Specialty Clinics | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| France | ||
| United Kingdom | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| South Korea | ||
| Australia | ||
| Rest of Asia-Pacific | ||
| Middle East & Africa | GCC | |
| South Africa | ||
| Rest of Middle East & Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
How fast is the neurothrombectomy devices market expected to grow between 2026 and 2031?
The market is projected to expand from USD 0.874 billion in 2026 to USD 1.19 billion by 2031, at a 6.47% CAGR.
Which product category is advancing the quickest?
Aspiration devices are projected to grow at a 10.13% CAGR through 2031 due to lower procedure times and costs.
What region shows the highest future growth?
Asia-Pacific is forecast to post an 8.12% CAGR, driven by mandated stroke-center rollouts in China and India.
Why are ambulatory surgical centers gaining share?
ASCs handle stable, small-core infarct cases under conscious sedation, enabling 23-hour discharge and trimming facility costs.
What limits adoption in low-income countries?
Device costs of USD 15,000-40,000 per case, scarce insurance coverage, and import tariffs keep penetration below 2% of eligible patients.
Page last updated on:









