MEA Single-use Medical Device Reprocessing Market Analysis by Mordor Intelligence
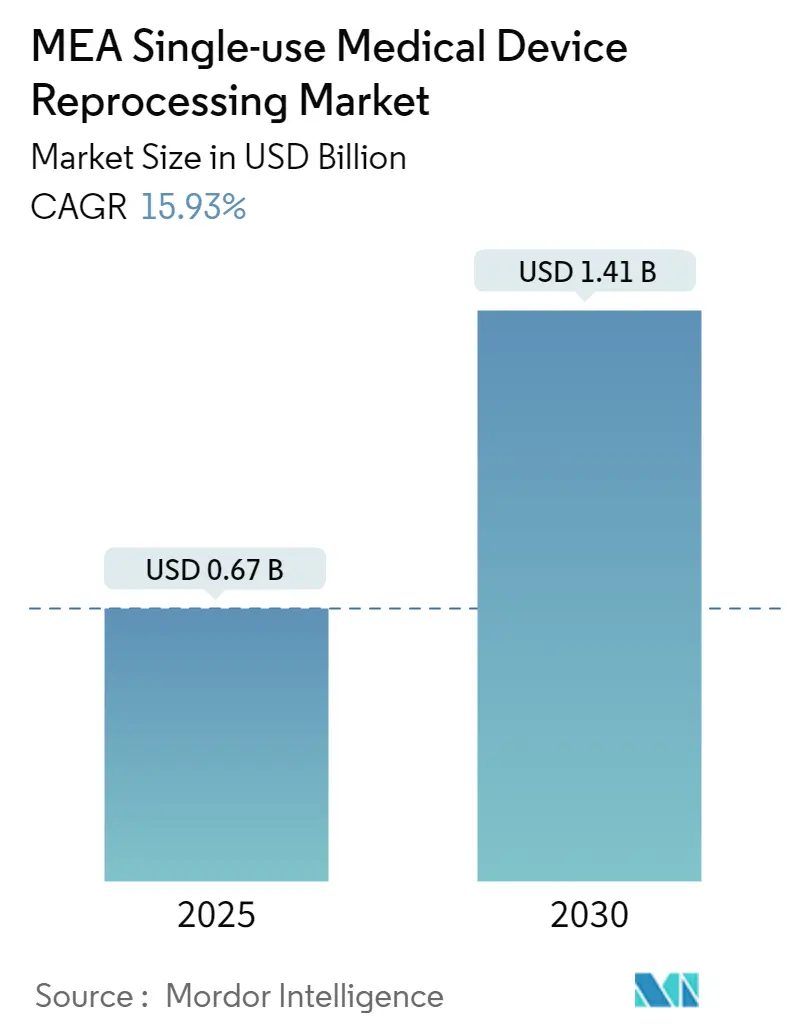
The MEA Single-use Medical Device Reprocessing Market size is estimated at USD 0.67 billion in 2025, and is expected to reach USD 1.41 billion by 2030, at a CAGR of 15.93% during the forecast period (2025-2030).
The Middle East and Africa's medical device reprocessing industry is undergoing significant transformation driven by growing healthcare sustainability initiatives and environmental concerns. According to BioEnergy Consult's March 2022 report, approximately 15-25% of medical waste generated in the Middle East is classified as hazardous or infectious, highlighting the critical need for sustainable healthcare solutions. This has led to major policy shifts, exemplified by the UAE President's declaration of 2023 as the Year of Sustainability, with healthcare groups specifically focusing on improving communities through enhanced medical waste management and reprocessing practices. The implementation of these initiatives has resulted in increased adoption of reprocessing programs across healthcare facilities in the region.
Regulatory frameworks and infrastructure development are evolving to support the growing reprocessing industry. In August 2023, Qatar's Ministry of Public Health implemented new regulations for the systematic disposal of medical waste, mandating separate containment systems and transfer protocols to health centers. This regulatory evolution is supported by significant investments, as evidenced by the World Bank's announcement in March 2023 of a USD 9.13 million grant from the Global Environment Facility to expand healthcare waste management initiatives in the Greater Cairo region. These developments are creating a more structured and efficient environment for medical device reprocessing operations.
The industry is witnessing substantial improvements in reprocessing technologies and operational protocols. Healthcare facilities are increasingly adopting advanced cleaning and sterilization techniques, with many institutions implementing rigorous validation processes for reprocessed devices. The Saudi Food and Drug Authority's March 2023 guidelines emphasize comprehensive documentation, competency requirements, and adherence to manufacturer instructions during reprocessing procedures. These operational improvements are enhancing the quality and reliability of reprocessed devices while ensuring compliance with international standards.
Quality assurance and safety concerns continue to shape market dynamics, particularly regarding material integrity and infection control. Healthcare providers are implementing more stringent testing protocols and validation procedures to address these concerns. According to the South Africa Medical Journal's April 2022 report, effective implementation of reprocessing programs could potentially reduce healthcare costs by USD 540 million annually, demonstrating the significant economic benefits of addressing quality concerns through proper protocols. This focus on quality assurance is driving investments in advanced reprocessing technologies and training programs across the region, contributing to hospital sustainability.
MEA Single-use Medical Device Reprocessing Market Trends and Insights
Cost Savings Through Reprocessing Single-Use Devices
Healthcare facilities across the Middle East and Africa are increasingly adopting reprocessing of single-use medical devices that were originally labeled as single-use, primarily driven by significant cost advantages. The cost of reprocessing a single-use device is approximately half the cost of purchasing a new device, making it an attractive option for healthcare facilities looking to optimize their operational expenses. This cost-effectiveness is particularly impactful as studies have shown that reprocessing of single-use medical devices programs can help healthcare facilities reduce their expenses by approximately USD 540 million annually, while maintaining the same quality and safety standards as new devices.
Strategic activities and partnerships in the region are further enhancing the availability and adoption of cost-effective reprocessing services. For instance, in February 2023, hospitals and surgical centers in Israel entered into strategic partnerships with professional medical device reprocessors to increase the accessibility of reprocessing services through third-party providers. These partnerships are particularly beneficial for healthcare facilities in regions with budget constraints, as they provide access to high-quality reprocessed devices while significantly reducing procurement costs. The growing focus on such collaborative approaches and the establishment of professional reprocessing networks is expected to continue driving reduction in healthcare costs across healthcare facilities in the region.
Rising Need to Reduce the Volume of Medical Waste
The escalating volume of medical waste generation in the Middle East and Africa region has created an urgent need for sustainable waste management solutions, positioning medical device reprocessing as a crucial strategy. Recent data highlights the severity of the situation, with GCC countries collectively generating more than 150 tons of medical waste daily, with Saudi Arabia alone producing over 80 tons per day. The United Arab Emirates generates approximately 21.5 tons of medical waste daily, while Kuwait and Bahrain produce 12 tons and 7 tons respectively, creating significant environmental and health challenges that necessitate immediate attention.
The implementation of new sustainability initiatives and regulations is further driving the adoption of medical device reprocessing as a waste reduction strategy. In January 2023, the UAE President announced the Year of Sustainability, prompting healthcare groups to focus on improving communities through better management of medical waste and reprocessing practices. Additionally, in August 2023, Qatar's Ministry of Public Health implemented new regulations for the separate disposal of medical waste through a specialized system, making the reprocessing procedure more efficient. These initiatives are particularly significant given that between 15-25% of the total medical waste created in the Middle East is considered hazardous or infectious, making proper waste management through reprocessing not just an environmental concern but also a critical public health issue.
Segment Analysis: By Device Type
Class II Devices Segment in MEA Single-use Medical Device Reprocessing Market
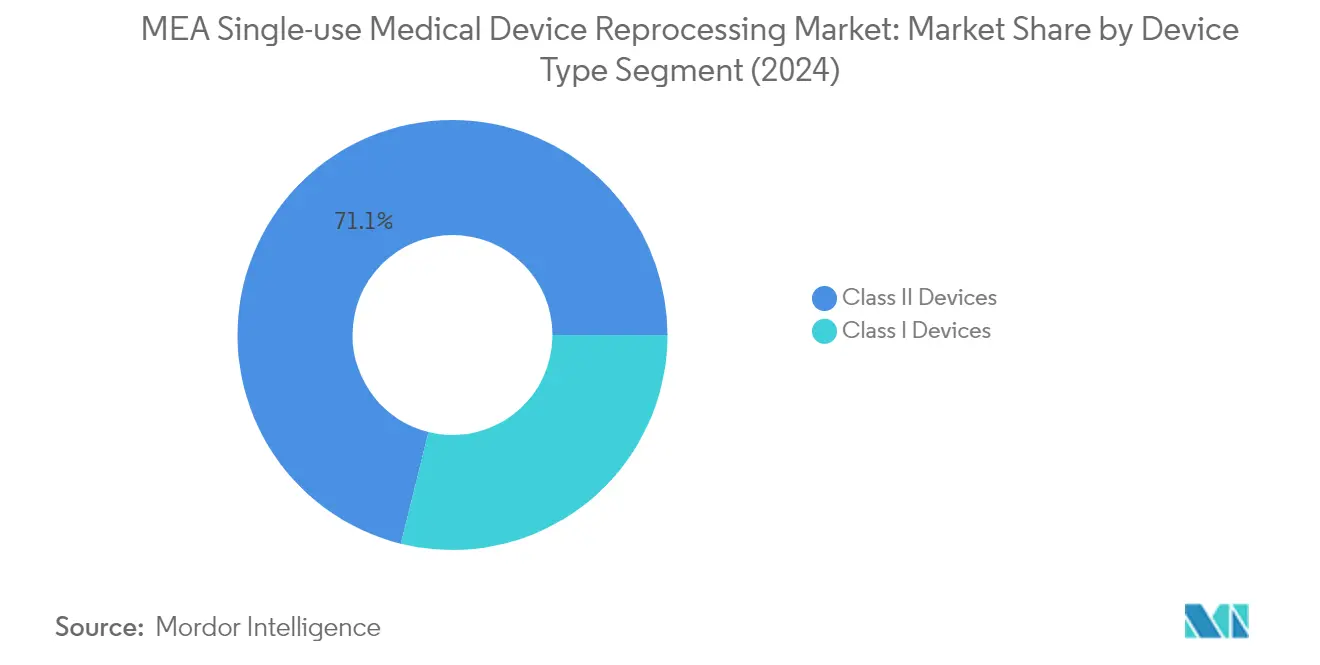
The Class II devices segment has established itself as the dominant force in the MEA single-use medical device reprocessing market, commanding approximately 71% of the total market share in 2024. This segment encompasses critical medical devices such as pulse oximeter sensors, sequential compression sleeves, catheters, and guidewires, which are extensively used in various medical procedures across healthcare facilities in the Middle East and Africa. The segment's leadership position is primarily attributed to the growing focus on these devices for medical device reprocessing, driven by their high procurement costs and frequent usage in medical procedures. Additionally, the segment is experiencing the fastest growth trajectory in the market, with an expected growth rate of approximately 16% during the forecast period 2024-2029, primarily due to the increasing adoption of reprocessing practices for these devices to reduce healthcare costs and medical waste generation in the region.
Remaining Segments in Device Type
The Class I devices segment represents the other major category in the MEA single-use medical device reprocessing market, encompassing essential medical tools such as laparoscopic graspers, scissors, forceps, scalpels, and tourniquet cuffs. This segment plays a crucial role in the market as these devices are fundamental to various surgical and medical procedures. The reprocessing of Class I devices is particularly significant in healthcare facilities looking to optimize their operational costs while maintaining quality healthcare delivery. The segment's growth is supported by the increasing awareness about medical device cleaning and the cost-effectiveness of reprocessing these relatively simpler devices compared to Class II devices. Furthermore, the importance of medical device sterilization in ensuring safety and compliance further emphasizes the segment's relevance in the market.
MEA Single-use Medical Device Reprocessing Market Geography Segment Analysis
Single-use Medical Device Reprocessing Market in GCC
The GCC region, comprising the United Arab Emirates, Saudi Arabia, Qatar, Oman, Kuwait, and Bahrain, dominates the Middle East & Africa medical device reprocessing market, holding approximately 47% of the market share. The region's leadership position is primarily driven by its advanced healthcare infrastructure and robust adoption of single-use device reprocessing technologies. The Kingdom of Saudi Arabia and the United Arab Emirates stand out as particularly developed markets within the GCC, featuring state-of-the-art reprocessing facilities and strong adherence to international quality standards. The region's healthcare facilities demonstrate a growing commitment to healthcare sustainability, particularly in reducing medical waste through reprocessing initiatives. The presence of key market players and their established distribution networks further strengthens the GCC's market position. Additionally, the region's focus on cost-effective healthcare solutions and environmental sustainability has led to increased adoption of reprocessing practices across various healthcare institutions. The GCC's regulatory environment, while still evolving, shows promising developments in supporting the growth of medical device reprocessing, particularly in major healthcare hubs like Dubai and Riyadh.
Single-use Medical Device Reprocessing Market in South Africa
South Africa represents a significant market in the Middle East & Africa region, demonstrating a robust historical growth rate of approximately 16% from 2019 to 2024. The market's development is characterized by increasing adoption of reprocessing practices across both public and private healthcare sectors. South Africa's healthcare system, which is one of the most developed in Africa, has been instrumental in driving the adoption of medical device reprocessing technologies. The country's medical facilities are increasingly recognizing the economic and environmental benefits of reprocessing single-use devices. The presence of local reprocessing service providers and their partnerships with international companies has enhanced the market's sophistication. South Africa's regulatory framework for medical device reprocessing has evolved to align with international standards, providing a structured environment for market growth. The country's healthcare institutions are particularly focused on maintaining high-quality standards in reprocessing practices while achieving cost efficiencies. Furthermore, South Africa serves as a regional hub for single-use device reprocessing, influencing practices across neighboring countries.
Single-use Medical Device Reprocessing Market in Rest of Middle East & Africa
The Rest of Middle East & Africa region, encompassing countries like Israel, Egypt, Iran, Syria, Jordan, Lebanon, and Nigeria, is projected to grow at approximately 15% during the 2024-2029 period. This market segment demonstrates significant potential for expansion, driven by increasing awareness of sustainable healthcare practices and cost-optimization needs. The region's healthcare facilities are gradually adopting reprocessing practices, particularly in countries with more developed healthcare systems like Israel and Egypt. The market in these countries is characterized by a mix of local reprocessing practices and partnerships with international reprocessing service providers. Healthcare institutions in these regions are increasingly focusing on implementing proper protocols and standards for medical device reprocessing. The growing burden of healthcare costs in these countries has made reprocessing an attractive option for healthcare facilities. Additionally, the increasing focus on reducing medical waste and environmental impact has encouraged healthcare providers to consider reprocessing as a viable solution. The market also benefits from knowledge transfer and technological adoption from more developed markets in the region, enhancing the hospital supply chain efficiency.
Competitive Landscape
Top Companies in MEA Single-use Medical Device Reprocessing Market
The competitive landscape is characterized by established multinational corporations like Stryker Corporation, Johnson & Johnson, Cardinal Health, and Getinge AB leading the market through comprehensive product portfolios and a strong regional presence. These companies are focusing on expanding their medical device lifecycle management capabilities through technological advancements in cleaning and sterilization processes while simultaneously strengthening their quality control measures to meet stringent regulatory requirements. Strategic partnerships with healthcare facilities and investment in local distribution networks have become crucial differentiators in the market. Companies are also emphasizing sustainability initiatives and cost-effectiveness in their reprocessing programs to align with growing environmental concerns and healthcare budget constraints. The market is witnessing an increased focus on specialized training programs for reprocessing technicians and the development of standardized protocols to ensure consistent quality across reprocessed devices.
Market Structure Shows Concentrated Player Base
The MEA single-use medical device reprocessing market exhibits a relatively concentrated structure dominated by global medical device manufacturers who have integrated reprocessing capabilities into their service offerings. These established players leverage their existing relationships with healthcare providers, technical expertise, and robust quality management systems to maintain their market positions. The market shows a limited presence of local specialists, with most regional activities being conducted through subsidiaries or authorized distributors of global companies. The high barriers to entry, including stringent regulatory requirements and the need for significant technical expertise, have contributed to this consolidated market structure.
The competitive dynamics are shaped by the presence of both diversified healthcare conglomerates and specialized reprocessing service providers. While conglomerates like Johnson & Johnson and Stryker Corporation benefit from their broad healthcare portfolios and established brand reputation, specialized players like NEScientific focus on developing innovative medical device cleaning technologies for specific device categories. The market has seen limited merger and acquisition activity, with companies primarily focusing on organic growth through capacity expansion and technology enhancement rather than consolidation.
Innovation and Compliance Drive Future Success
Success in the MEA single-use medical device reprocessing market increasingly depends on companies' ability to balance innovation with regulatory compliance while maintaining cost-effectiveness. Market incumbents are strengthening their positions by investing in advanced cleaning and medical device sterilization services, developing comprehensive quality management systems, and building stronger relationships with healthcare providers through value-added services. New entrants and smaller players are finding opportunities by focusing on specific device categories or regional markets where they can develop specialized expertise and establish a strong local presence.
The market's future competitive landscape will be shaped by several critical factors, including the ability to address growing concerns about device safety and quality, adaptation to evolving regulatory frameworks, and development of sustainable reprocessing solutions. Companies need to focus on building trust with healthcare providers through transparent quality assurance processes and documented cost savings. The increasing emphasis on environmental sustainability in healthcare operations presents opportunities for companies to differentiate themselves through eco-friendly reprocessing solutions and waste reduction programs. Additionally, the development of specialized training programs and technical support services will become increasingly important in maintaining a competitive advantage. Furthermore, integrating medical equipment reprocessing into the hospital supply chain can offer significant cost efficiencies and sustainability benefits.
MEA Single-use Medical Device Reprocessing Industry Leaders
Stryker Corporation
Johnson & Johnson
Cardinal Health
NEScientific Inc.
Arjo
- *Disclaimer: Major Players sorted in no particular order



Recent Industry Developments
- June 2022: The Ministry of Health (MOH) launched a workshop in Oman on endoscope reprocessing in collaboration with WHO. The event involved an exhibition displaying state-of-the-art equipment and techniques for medical device reprocessing.
- March 2022: Getinge AB, a company focused on developing automated endoscope reprocessors, established a Getinge training center in Dubai on the premises of the medtech company's distributor, MPC Healthcare. In this center, visitors can experience and interact with the company's sterile reprocessing solutions.
MEA Single-use Medical Device Reprocessing Market Report Scope
As per the scope of the report, single-use device (SUD) reprocessing includes the cleaning, disinfection, sterilization, testing, and restoration of the technical and functional safety of used devices. The reprocessing of SUDs makes it possible for hospitals to maintain patient care quality while saving substantial amounts of money. The Middle East and Africa single-use medical device reprocessing market is segmented by device type (class I devices (laparoscopic graspers, scissors, forceps, scalpels, tourniquet cuffs, and other class I devices) and class II devices (pulse oximeter sensors, sequential compression sleeves, catheters, guidewires, and other class II devices)) and geography (GCC, South Africa, and Rest of Middle East and Africa). The report offers the value (in USD) for the above segments.
| Class I Devices | Laparoscopic Graspers |
| Scissors | |
| Forceps | |
| Scalpels | |
| Tourniquet Cuffs | |
| Other Class I Devices | |
| Class II Devices | Pulse Oximeter Sensors |
| Sequential Compression Sleeves | |
| Catheters and Guidewires | |
| Other Class II Devices |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa |
| By Device Type | Class I Devices | Laparoscopic Graspers |
| Scissors | ||
| Forceps | ||
| Scalpels | ||
| Tourniquet Cuffs | ||
| Other Class I Devices | ||
| Class II Devices | Pulse Oximeter Sensors | |
| Sequential Compression Sleeves | ||
| Catheters and Guidewires | ||
| Other Class II Devices | ||
| Geography | Middle East and Africa | GCC |
| South Africa | ||
| Rest of Middle East and Africa | ||


Key Questions Answered in the Report
How big is the MEA Single-use Medical Device Reprocessing Market?
The MEA Single-use Medical Device Reprocessing Market size is expected to reach USD 0.67 billion in 2025 and grow at a CAGR of 15.93% to reach USD 1.41 billion by 2030.
What is the current MEA Single-use Medical Device Reprocessing Market size?
In 2025, the MEA Single-use Medical Device Reprocessing Market size is expected to reach USD 0.67 billion.
Who are the key players in MEA Single-use Medical Device Reprocessing Market?
Stryker Corporation, Johnson & Johnson, Cardinal Health, NEScientific Inc. and Arjo are the major companies operating in the MEA Single-use Medical Device Reprocessing Market.
Which is the fastest growing region in MEA Single-use Medical Device Reprocessing Market?
Middle East and Africa is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in MEA Single-use Medical Device Reprocessing Market?
In 2025, the Middle East and Africa accounts for the largest market share in MEA Single-use Medical Device Reprocessing Market.
What years does this MEA Single-use Medical Device Reprocessing Market cover, and what was the market size in 2024?
In 2024, the MEA Single-use Medical Device Reprocessing Market size was estimated at USD 0.56 billion. The report covers the MEA Single-use Medical Device Reprocessing Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the MEA Single-use Medical Device Reprocessing Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:








