Single-use Medical Device Reprocessing Market Analysis by Mordor Intelligence
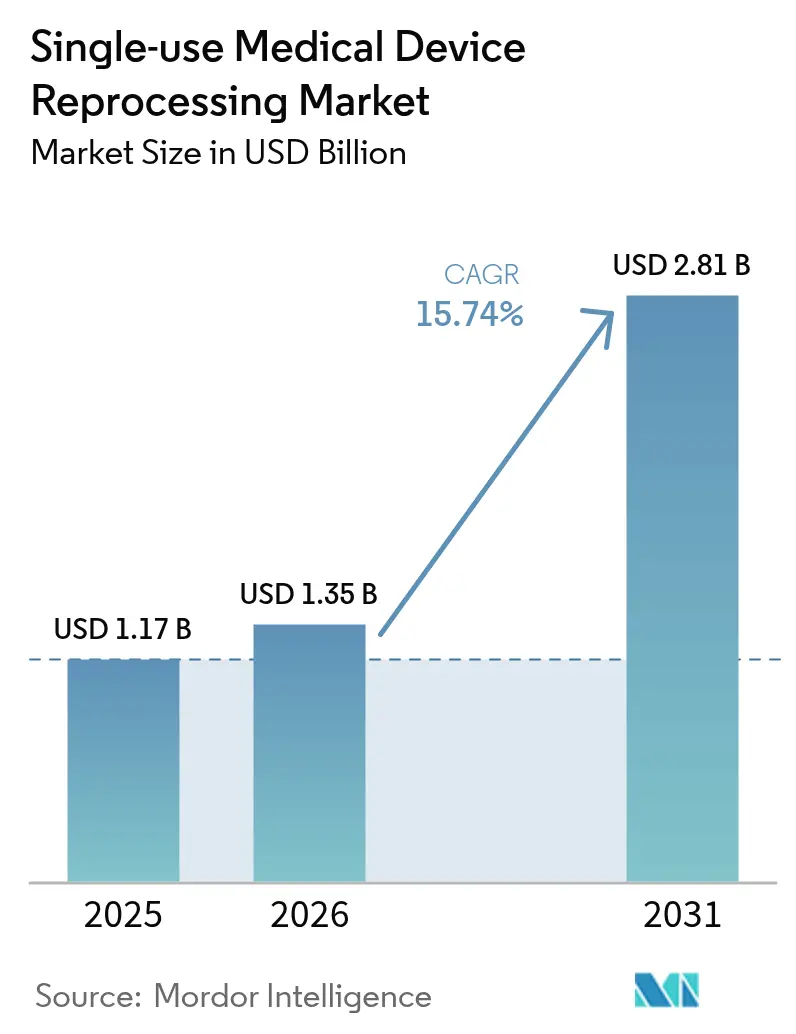
The single-use medical device reprocessing market size is expected to grow from USD 1.17 billion in 2025 to USD 1.35 billion in 2026 and is forecast to reach USD 2.81 billion by 2031 at 15.74% CAGR over 2026-2031. Regulatory clarity from the U.S. FDA’s May 2024 guidance on remanufacturing and growing acceptance of vaporized hydrogen peroxide sterilization have reduced compliance uncertainty and broadened the universe of devices considered safe for reprocessing[1]Source: U.S. Food and Drug Administration, “FDA Issues Final Guidance to Clarify ‘Remanufacturing’ of Devices,” fda.gov . Hospital procurement teams view reprocessing as a line-item lever for margin preservation amid inflation and reimbursement headwinds, especially after documented savings of USD 451 million in 2024 across 17 countries[2]Source: Dan Vukelich, “Earth Day News: Hospitals and Surgical Centers Save USD 451 Million,” amdr.org . Sustainability mandates, Scope-3 carbon accounting and antitrust enforcement against restrictive OEM contracts are accelerating device-level adoption, while AI-enabled traceability platforms and automated sterilizers reinforce patient-safety confidence .
Key Report Takeaways
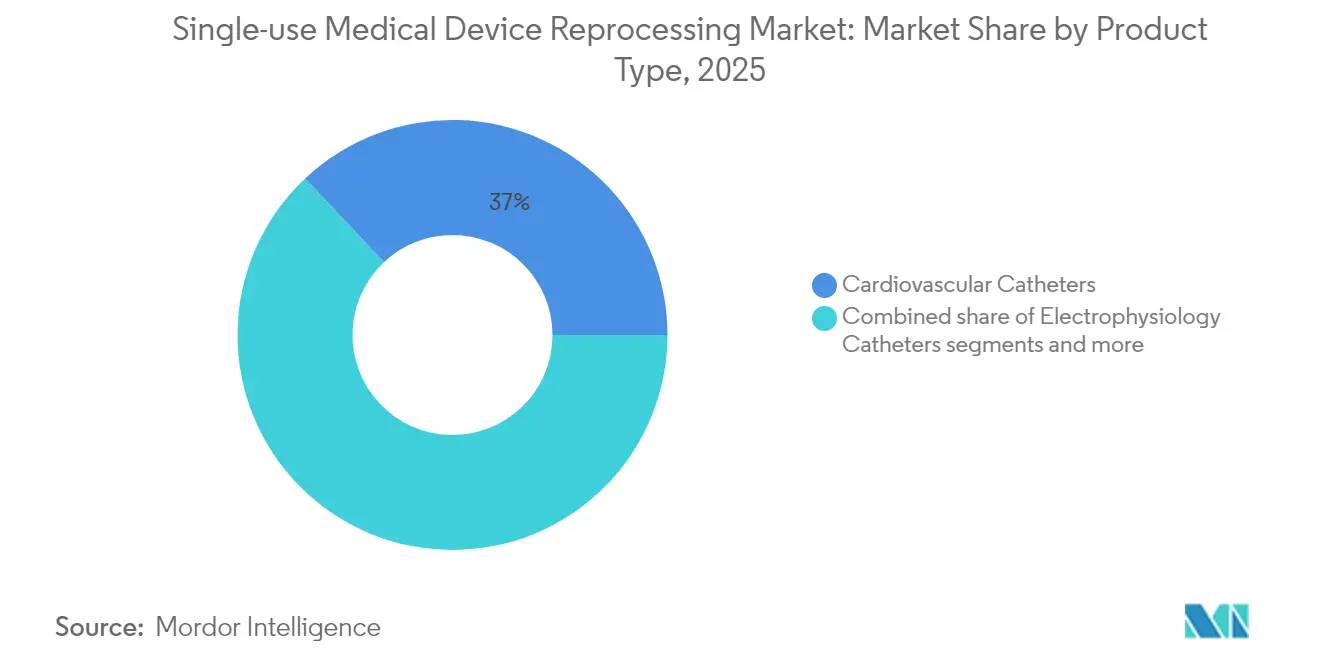
- By product type, cardiovascular catheters led with 37.02% of single-use medical device reprocessing market share in 2025; electrophysiology catheters are projected to register the fastest 15.92% CAGR through 2031 .
- By service provider, third-party commercial reprocessors held 84.12% of the single-use medical device reprocessing market size in 2025, while the segment is poised to expand at a 16.05% CAGR between 2026 and 2031 .
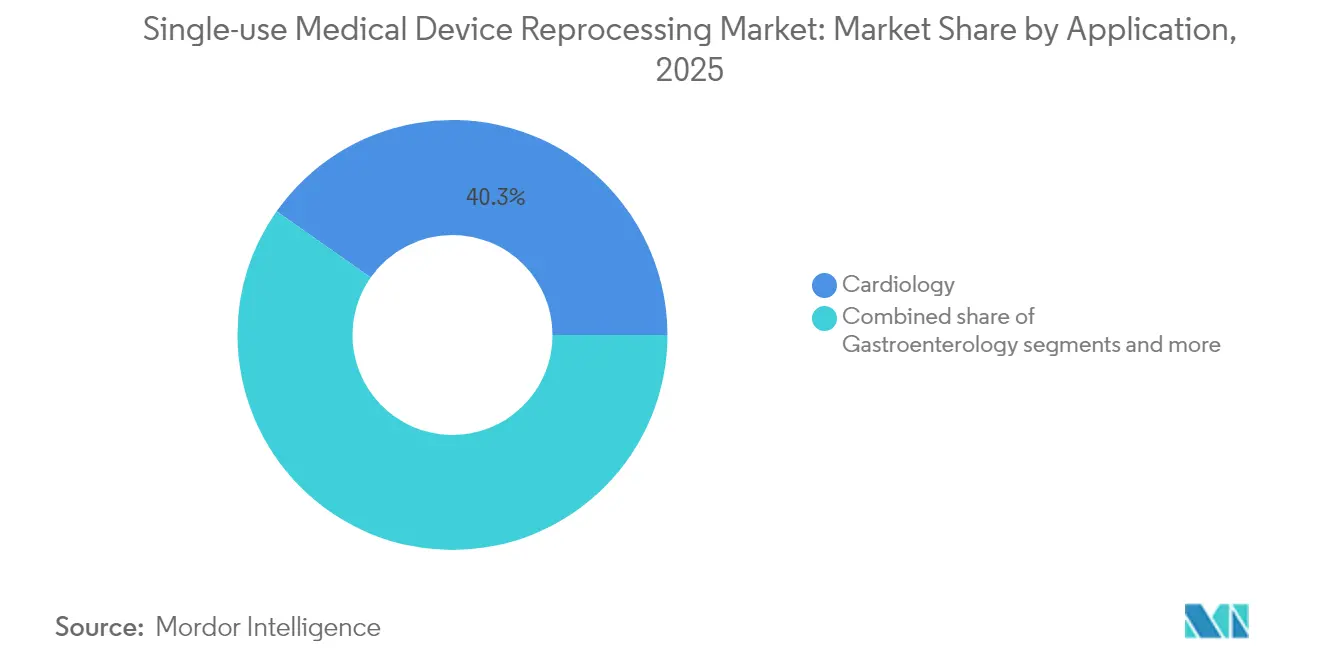
- By application, cardiology accounted for 40.25% share of the single-use medical device reprocessing market size in 2025; gastroenterology applications show the highest 16.28% CAGR outlook to 2031 .
- By end user, hospitals and surgical centers represented 63.75% demand in 2025, whereas ambulatory surgical centers are advancing at a 16.63% CAGR through 2031 .
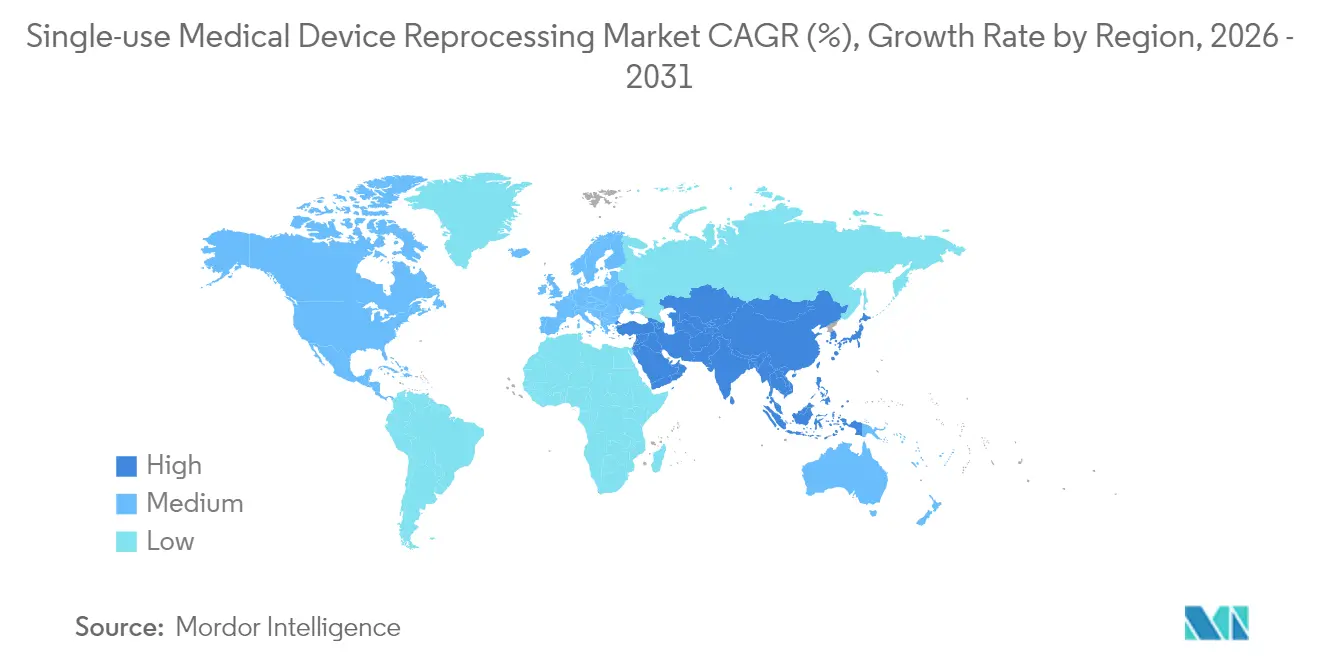
- By geography, North America commanded 43.10% revenue in 2025, yet Asia-Pacific is forecast to expand the single-use medical device reprocessing market at 16.88% CAGR to 2031 .
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Single-use Medical Device Reprocessing Market Trends and Insights
Driver Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Cost-containment pressure on hospitals | +4.2% | Global, with acute impact in North America & Europe | Medium term (2-4 years) |
| Regulatory approvals & clearances for reprocessed SUDs | +3.8% | North America & EU primary, expanding to APAC | Long term (≥ 4 years) |
| Sustainability & waste-reduction mandates | +2.9% | EU leading, North America following, APAC emerging | Long term (≥ 4 years) |
| ESG reporting tying Scope-3 emissions to procurement | +2.1% | Global corporate markets, concentrated in developed economies | Medium term (2-4 years) |
| Antitrust rulings curbing OEM restrictive contracts | +1.8% | North America primary, potential EU spillover | Short term (≤ 2 years) |
| Supply-chain resilience post-pandemic PPE shortages | +1.5% | Global, with emphasis on strategic stockpiling regions | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Cost-containment Pressure on Hospitals
Shrinking operating margins have moved reprocessing from optional to essential in hospital supply-chain playbooks, often delivering 40-60% device-level savings versus OEM list prices . Medicare payment reforms and private-payer contracting heightened visibility of disposable device spend, prompting the Veterans Health Administration to revisit its own reprocessing restrictions in 2025 . Chief financial officers increasingly embed reprocessing ROIs in annual capital-allocation models, translating to systematic adoption across multi-hospital networks . The scale of savings is now material to bond-rating agencies evaluating nonprofit health-system liquidity, reinforcing management commitment . As inflation persists, financial stewardship is expected to underpin at least one-third of new account conversions through 2027 .
Regulatory Approvals & Clearances for Reprocessed SUDs
The FDA’s 2024 remanufacturing guidance clarified boundaries between servicing and reprocessing, reducing legal ambiguity for third-party operators . Vaporized hydrogen peroxide earned recognition as an established sterilization modality, diversifying validated methods beyond ethylene oxide . Japan embedded single-use device re-manufacturing into its QMS ordinance with staggered compliance deadlines through 2024, setting a template for other APAC regulators . The FDA’s 2025 approvals of VARIPULSE and Sphere-9 catheter systems, each containing reusable components, signaled growing confidence in mixed-use platforms . These milestones collectively expand the single-use medical device reprocessing market addressable base beyond cardiology into complex electrophysiology segments .
Sustainability & Waste-reduction Mandates
National health systems in the EU and United Kingdom now treat reprocessing as a compliance lever for landfill diversion and carbon-reduction targets rather than a discretionary green initiative . Over 70% of U.S. hospitals reported formal operating-room waste programs in 2024, with reprocessing identified as the single largest contributor to avoided landfill tonnage . The United Kingdom’s plan to eliminate avoidable single-use medical products by 2045 explicitly lists device reprocessing among endorsed strategies . Several EU jurisdictions are evaluating waste taxes indexed to kilogram output, potentially boosting the economic rationale for reprocessing by 2026 . With healthcare estimated to contribute 4.4% of global greenhouse gases, every 40% emission reduction per reprocessed unit tangibly improves providers’ Scope-3 profiles .
ESG Reporting Tying Scope-3 Emissions to Procurement
Rating agencies and investors increasingly scrutinize hospital Scope-3 disclosures, linking supply-chain emissions to cost of capital for large systems . Proprietary carbon calculators from leading reprocessors now quantify per-device CO₂ avoidance, enabling procurement teams to monetize the sustainability premium in vendor scorecards . Large integrated delivery networks bundle carbon metrics with price when awarding multi-year supply contracts, elevating reprocessing from tactical savings tool to strategic ESG differentiator . Health-system CFOs highlight Scope-3 performance in bond-offering documents to tap green-bond investor pools, creating financial upside for aggressive device-level reprocessing adoption . Collectively, these pressures are projected to drive 18–20% of new reprocessing account wins through 2028 .
Restraint Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| OEM lobbying & restrictive labelling practices | -2.8% | Global, concentrated in markets with strong OEM presence | Medium term (2-4 years) |
| Device design limits to multiple re-use cycles | -1.9% | Global, technology-dependent rather than geography-specific | Long term (≥ 4 years) |
| EU MDR Article 17 cross-border fragmentation | -1.4% | European Union primary, potential regulatory spillover effects | Long term (≥ 4 years) |
| AI-enabled traceability exposing reprocessing failures | -0.8% | Developed markets with advanced healthcare IT infrastructure | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
OEM Lobbying & Restrictive Labeling Practices
Johnson & Johnson’s 2025 antitrust loss, accompanied by a USD 442 million penalty, underscores systemic OEM resistance to reprocessing adoption . Manufacturers continue to leverage “single-use” labels to sow legal uncertainty, particularly in emerging markets with nascent regulatory oversight . Trade associations funded by OEMs lobby against expanded device-eligibility lists, delaying clinical uptake in high-volume categories like laparoscopic instruments . Even where antitrust scrutiny curbs overt contract restrictions, soft barriers such as staff-training withdrawal can still impede provider confidence . The resulting legal environment is expected to shave nearly three percentage points off projected CAGR in the near term .
EU MDR Article 17 Cross-border Fragmentation
European Union's Medical Device Regulation Article 17 creates a complex patchwork of national implementations that fragment the European reprocessing market and increase compliance costs. Individual member states retain authority to restrict or prohibit reprocessing, resulting in regulatory arbitrage that complicates pan-European reprocessing strategies. Germany's Bundesrat consideration of banning CE reprocessing after over 10 years of practice demonstrates regulatory instability that creates investment uncertainty. Cross-border device movement for reprocessing faces varying national interpretations of manufacturer obligations, creating logistical complexity that favors larger reprocessors with multi-jurisdictional compliance capabilities. The European Commission's December 2022 study on Article 17 implementation revealed significant variation in national approaches, suggesting continued fragmentation rather than harmonization.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Product Type: Cardiovascular Dominance Drives Market Maturity
The cardiovascular catheter category delivered 37.02% of single-use medical device reprocessing market size in 2025, sustained by well-documented clinical equivalency studies and standardized sterilization workflows . Electrophysiology catheters are pacing the field with a 15.92% CAGR, catalyzed by FDA approvals for VARIPULSE and Sphere-9 systems that incorporate reusable components . Laparoscopic instruments benefit from robotic-surgery scale, yet complex geometries demand automated cleaning tunnels available chiefly at large commercial reprocessors . Gastroenterology device growth hinges on sterilization breakthroughs like ULTRA GI hydrogen-peroxide gas-plasma cycles, which mitigate past infection risks . Orthopedic external-fixation hardware remains niche due to patient-custom configurations and extended wear times, while general-surgery tools provide steady volume but face pricing commoditization pressures .
Technological advances, notably AI-based device-tracking tags and cloud analytics, now allow product-level performance benchmarking across reuse cycles, fostering surgeon trust in reprocessed alternatives . Enhanced inspection optics and non-destructive integrity testing continue to elevate quality assurance, helping cardiovascular and laparoscopic devices maintain near-parity with new OEM units in failure rates . As category life-cycle emissions enter formal ESG scorecards, providers prioritize high-volume disposable categories such as catheters for earliest conversion, reinforcing leadership of cardiovascular subsegments in the single-use medical device reprocessing market .
Note: Segment shares of all individual segments available upon report purchase
By Service Provider: Third-party Consolidation Accelerates
Third-party operators accounted for 84.12% of single-use medical device reprocessing market share in 2025, reflecting scale advantages in sterilization, validation and logistics . The sector’s 16.05% CAGR through 2031 is fueled by payer and regulator preference for ISO-certified specialist facilities over resource-strained in-house units . Consolidation continues, exemplified by Medline’s 2024 acquisition of Ecolab’s surgical solutions business, integrating reprocessing into a full-line distribution model that simplifies provider procurement.
Hospitals evaluating internal programs confront capital outlays for sterilizers, traceability software and quality testing that exceed USD 5 million per site, tipping cost-benefit calculations toward outsourcing . Regulatory amendments harmonizing FDA QSR with ISO 13485 in 2026 are expected to heighten documentation burdens, disadvantaging smaller in-house units lacking dedicated regulatory teams . As commercial partners adopt AI vision-inspection and robotic packing lines, throughput efficiencies generate 5–8-point margin advantages, widening the gap versus hospital-run operations. Accordingly, most top-100 U.S. IDNs now operate hybrid models where only very low-volume instruments remain on-site, while high-volume cath-lab and EP devices ship to third-party plants weekly.
By Application: Cardiology Leadership with Gastroenterology Acceleration
Cardiology procedures accounted for 40.25% of single-use medical device reprocessing market size in 2025, built on decades of clinical studies proving reprocessed catheter safety . Ablation volume growth and antitrust rulings that dismantle OEM service-withholding policies should maintain momentum through 2031 . Gastroenterology shows the highest 16.28% CAGR forecast, thanks to newly validated vaporized-hydrogen-peroxide sterilization cycles for duodenoscopes that remove infection-control barriers .
Orthopedics remains limited by low procedure standardization and patient-specific instrumentation, but trauma centers still unlock savings on external-fixation constructs with predictable geometries . Urology applications benefit from increased adoption of disposable ureteroscopes, creating demand for validated secondary reuse cycles once lumen-cleaning robotics reach commercial maturity in 2027. General surgery’s growth parallels operating-room sustainability mandates but faces price ceiling constraints due to commoditized device sets .
Note: Segment shares of all individual segments available upon report purchase
By End User: ASC Growth Outpaces Hospital Adoption
Hospitals and integrated delivery networks comprised 63.75% of 2025 demand, yet growth rate trails the overall market as decision hierarchies extend evaluation cycles . In contrast, ambulatory surgical centers will post a 16.63% CAGR through 2031, driven by fee-for-service migration and investor-owned ASC chains prioritizing rapid payback on device expense lines . ASCs leverage reprocessing to sidestep inventory-carrying costs and mitigate vendor-backorder risk, often signing exclusive deals with a single commercial partner to streamline logistics .
Academic medical centers, while early adopters of sustainability programs, face unique research protocol constraints that slow full transition to reprocessed instrumentation . Specialty cath labs capitalize on high device-reuse potential, negotiating volume-based rebates that can exceed USD 2 million annually at top U.S. heart institutes. The combined ASC–clinic segment is expected to capture 38% of incremental single-use medical device reprocessing market revenue over the next five years, reflecting procedure site-of-care shifts accelerated by payer incentives .
Geography Analysis
North America generated 43.10% of 2025 revenue, underpinned by FDA guidance certainty, antitrust enforcement and well-developed third-party networks . The U.S. market also benefits from hospital sustainability charters that align reprocessing with ESG key-performance indicators . Canada’s public-funded health system adopts reprocessing to offset budget caps, while Mexico’s medical-device cluster in Baja California offers near-shore sterilization capacity expansion for cross-border providers .
Asia-Pacific is on track to record a 16.88% CAGR to 2031, led by Japan’s QMS rule harmonization and China’s hospital-modernization programs, which target 70% device-reuse certification in tier-1 cities by 2028 . India’s Ayushman Bharat scheme expands insurance coverage, compelling public hospitals to stretch fixed budgets, thereby elevating reprocessing in procurement tenders starting 2026 . South Korea and Australia, both early electron-beam sterilization adopters, pilot AI-tracked catheter reuse to meet national zero-waste targets by 2035 .
Europe’s outlook is tempered by Article 17 fragmentation that introduces divergent reprocessing rules among member states, inflating compliance costs by up to 25% for cross-border operators . Germany’s potential CE-reprocessing ban could remove USD 90 million in annual revenue if enacted in 2026, although Denmark and the Netherlands have issued guidance enabling reprocessing under strict quality-management oversight . The United Kingdom, outside EU frameworks, formally targets elimination of avoidable single-use medical goods by 2045, positioning reprocessing as a central compliance mechanism . France’s health ministry launched a limited pilot in 2024 to evaluate duodenoscope reprocessing economics, possibly informing national policy in 2027 .
Competitive Landscape
The single-use medical device reprocessing market remains moderately fragmented, yet scale-driven consolidation is accelerating as ISO-quality compliance and advanced sterilization investments raise entry barriers . Stryker’s Sustainability Solutions division served more than 3,000 U.S. hospitals and delivered USD 238 million in customer savings during 2023, leveraging AI-enabled track-and-trace dashboards to differentiate service levels . Medline’s acquisition of Ecolab’s surgical solutions portfolio in 2024 extended end-to-end logistics control from original manufacture through reprocessing, enhancing negotiating power with hospitals seeking bundled sourcing .
Independent specialty players capitalize on electrophysiology and gastroenterology niches where device complexity historically limited reuse; Innovative Health’s successful antitrust verdict against Johnson & Johnson validates the pathway for market access challenges when OEMs withhold support . New entrants featuring cloud-native quality-management systems and modular sterilization pods aim to service rural hospitals lacking volume for traditional hub-and-spoke models .
Looking forward, the 2026 FDA QSR–ISO 13485 alignment should simplify dual-compliance burdens, favoring agile mid-tier firms while maintaining rigorous patient-safety thresholds . Systematic digitization of sterilizer performance metrics and predictive maintenance algorithms will likely underpin next-wave differentiation, enabling real-time client dashboards that tie device-reuse cycles to carbon-savings analytics . Anticipated OEM divestitures of internal reprocessing units could inject additional consolidation targets, potentially elevating top-five market share above the current 55% threshold by 2028 .
Single-use Medical Device Reprocessing Industry Leaders
Stryker Corporation
Medline Industries Inc.
Innovative Health
Johnson & Johnson (Sterilmed Inc.)
Arjo
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- June 2025: Johnson & Johnson MedTech ordered to pay USD 442 million for antitrust violations tied to withholding clinical support for hospitals using reprocessed catheters
- June 2022: The Association of Medical Device Reprocessors launched 'Global Regulatory Standards for 'Single-Use' Medical Device Reprocessing and Remanufacturing,' the first roadmap to help Notified Bodies, Ministries of Health, and regulatory authorities of medical devices to unlock these benefits for hospitals and health systems worldwide.
Global Single-use Medical Device Reprocessing Market Report Scope
As per the scope of the report, single-use device (SUD) reprocessing includes cleaning, disinfection, sterilization, testing, and restoration of the technical and functional safety of the used device. Reprocessing of SUDs makes it possible for hospitals to maintain patient care quality while saving substantial amounts of money.
The single-use medical device reprocessing market is segmented by device type (class I devices (laparoscopic graspers, scalpels, tourniquet cuffs, and other class I devices) and class II devices (pulse oximeter sensors, sequential compression sleeves, catheters, and guidewires, and other class II devices)) and geography. (North America, Europe, Asia-Pacific, Middle East Africa, and South America). The market report also covers the estimated market sizes and trends for 17 countries across major regions globally.
The report offers the value (in USD) for the above segments.
| Cardiovascular Catheters |
| Electrophysiology Catheters |
| Laparoscopic Instruments |
| Gastroenterology Devices |
| Orthopedic External Fixation Devices |
| General Surgery Instruments |
| Third-party /Commercial Reprocessors |
| In-house /Hospital Reprocessing Units |
| Cardiology |
| Gastroenterology |
| Orthopedics |
| Urology |
| General Surgery |
| Hospitals & Surgical Centers |
| Ambulatory Surgical Centers |
| Specialty Clinics & Cath Labs |
| Academic & Research Institutes |
| North America | United States |
| Canada | |
| Mexico | |
| South America | Brazil |
| Argentina | |
| Rest of South America | |
| Europe | Germany |
| France | |
| United Kingdom | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| South Korea | |
| Australia | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| Saudi Arabia | |
| Rest of Middle East and Africa |
| By Product Type (Value, USD) | Cardiovascular Catheters | |
| Electrophysiology Catheters | ||
| Laparoscopic Instruments | ||
| Gastroenterology Devices | ||
| Orthopedic External Fixation Devices | ||
| General Surgery Instruments | ||
| By Service Provider (Value, USD) | Third-party /Commercial Reprocessors | |
| In-house /Hospital Reprocessing Units | ||
| By Application (Value, USD) | Cardiology | |
| Gastroenterology | ||
| Orthopedics | ||
| Urology | ||
| General Surgery | ||
| By End User (Value, USD) | Hospitals & Surgical Centers | |
| Ambulatory Surgical Centers | ||
| Specialty Clinics & Cath Labs | ||
| Academic & Research Institutes | ||
| By Geography (Value, USD) | North America | United States |
| Canada | ||
| Mexico | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||
| Europe | Germany | |
| France | ||
| United Kingdom | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| South Korea | ||
| Australia | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| Saudi Arabia | ||
| Rest of Middle East and Africa | ||


Key Questions Answered in the Report
What is the projected value of the single-use medical device reprocessing market in 2031?
It is forecast to reach USD 2.81 billion by 2031, up from USD 1.35 billion in 2026 at a 15.74% CAGR.
Which product type currently dominates device reprocessing?
Cardiovascular catheters hold 37.02% revenue share, supported by long-standing clinical validation.
Why are ambulatory surgical centers adopting reprocessing faster than hospitals?
ASCs prioritize margin management and have streamlined governance, enabling a 16.63% CAGR through 2031.
How do environmental regulations influence adoption?
Scope-3 carbon reporting and landfill-reduction mandates position reprocessing as a compliance pathway that also generates device-level savings.
What impact did the 2025 Johnson & Johnson antitrust ruling have?
The USD 442 million penalty curtailed OEM contract restrictions and opened catheter reprocessing opportunities for independent providers.
Page last updated on:











