Clinical Trial Management Systems Market Size and Share
Clinical Trial Management Systems Market Analysis by Mordor Intelligence
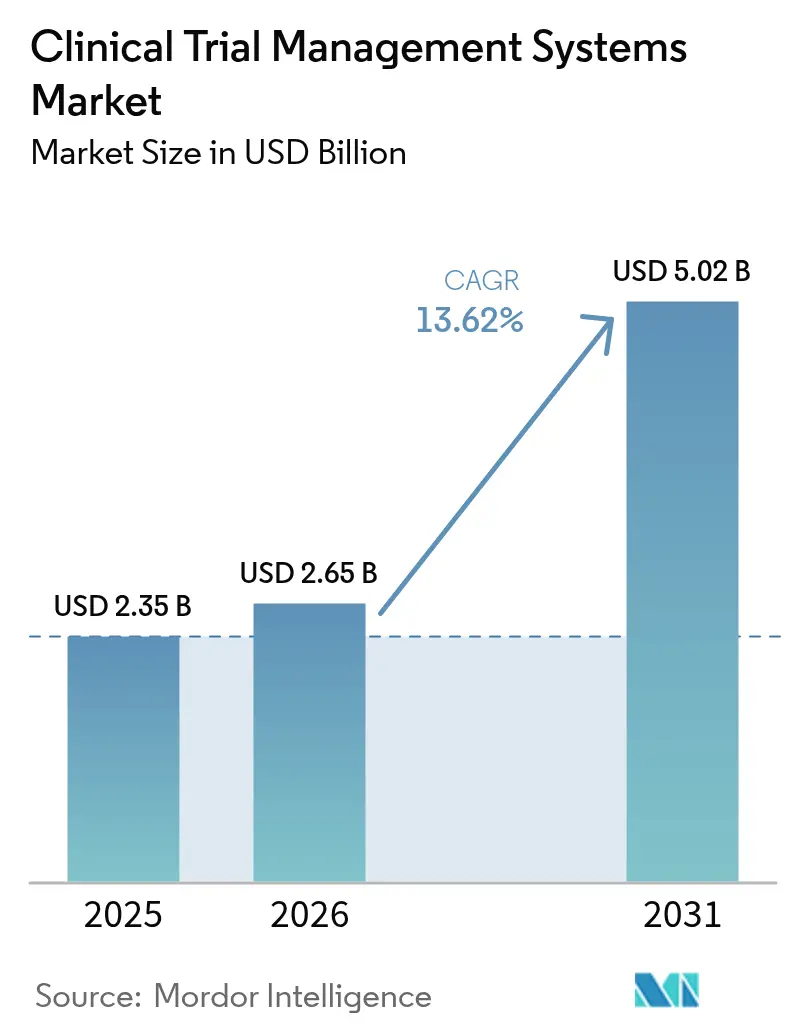
The Clinical Trial Management Systems Market size is projected to expand from USD 2.35 billion in 2025 and USD 2.65 billion in 2026 to USD 5.02 billion by 2031, registering a CAGR of 13.62% between 2026 to 2031.
Surging adoption of cloud-native platforms, expanding decentralized trial models, and new ICH-GCP E6(R3) real-time oversight mandates are accelerating investment decisions. Sponsors are consolidating disparate study tools, patient tracking, document management, and monitoring dashboards, into unified CTMS suites to cut cycle times and improve data integrity. Vendors are embedding artificial-intelligence modules that forecast enrollment risk, automate protocol-deviation alerts, and recommend corrective actions, helping sponsors protect milestone-based financing. Meanwhile, geopolitical data-sovereignty requirements are fracturing global hosting strategies, nudging multinationals toward region-specific CTMS tenants that comply with local privacy laws. Competitive intensity remains moderate as cloud newcomers challenge on-premise incumbents with usage-based pricing and rapid feature delivery cadences.
Key Report Takeaways
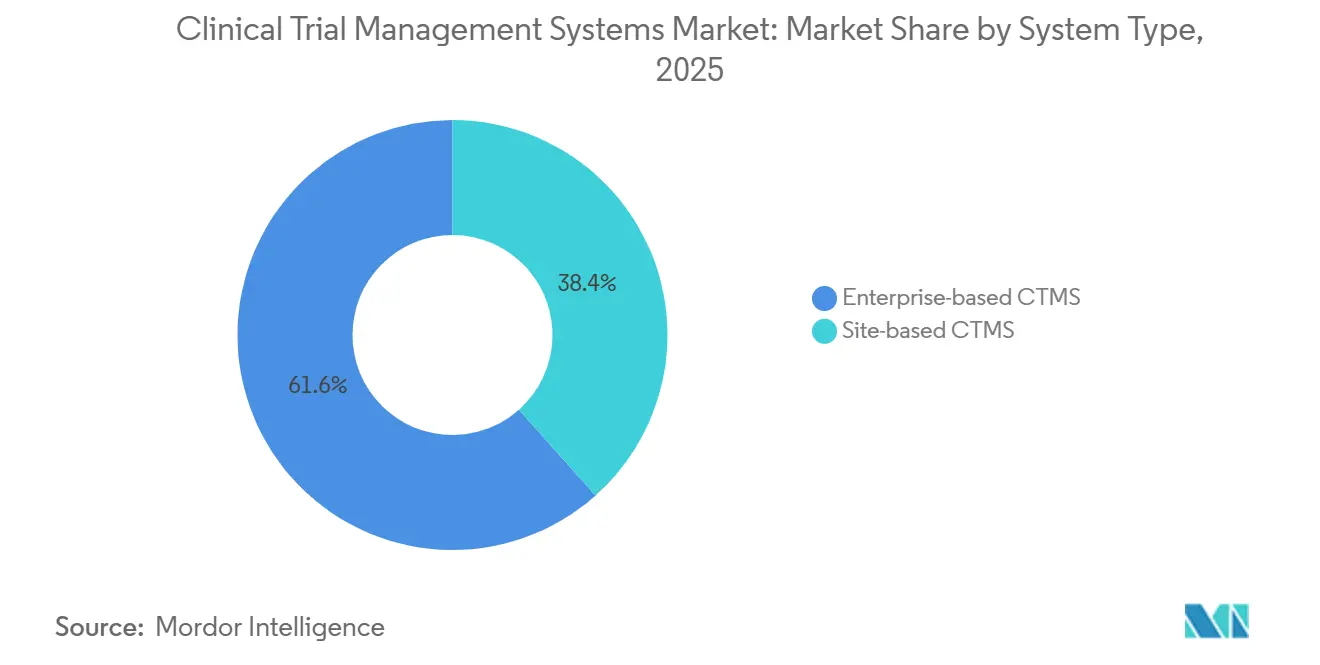
- By system type, enterprise platforms led with 61.58% of revenue in 2025, while site-based solutions are projected to expand at a 15.89% CAGR through 2031.
- By delivery mode, web-hosted deployments captured 54.66% of 2025 sales, whereas cloud SaaS is advancing at a 17.48% CAGR to 2031.
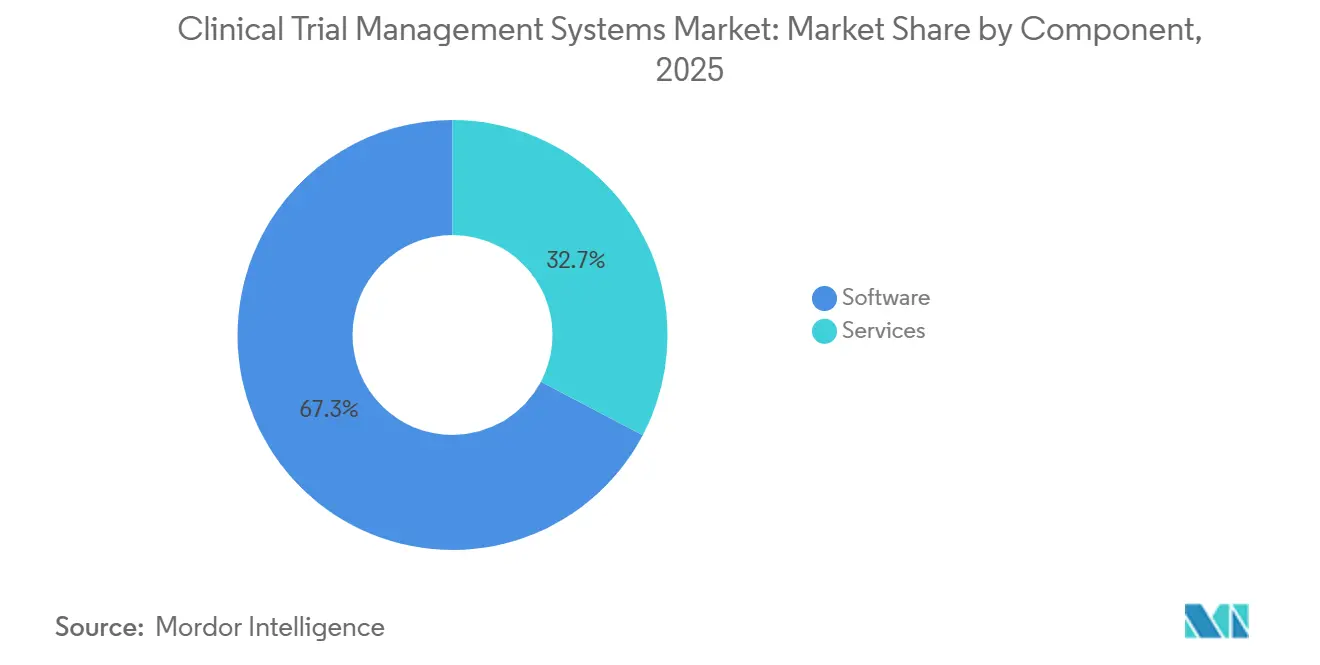
- By component, software licenses accounted for 67.27% of revenue in 2025; professional services represent the fastest-growing slice at a 16.32% CAGR over the forecast period.
- By end user, pharmaceutical and biotechnology sponsors held 39.52% of 2025 spend, while contract research organizations are on track for a 17.43% CAGR through 2031.
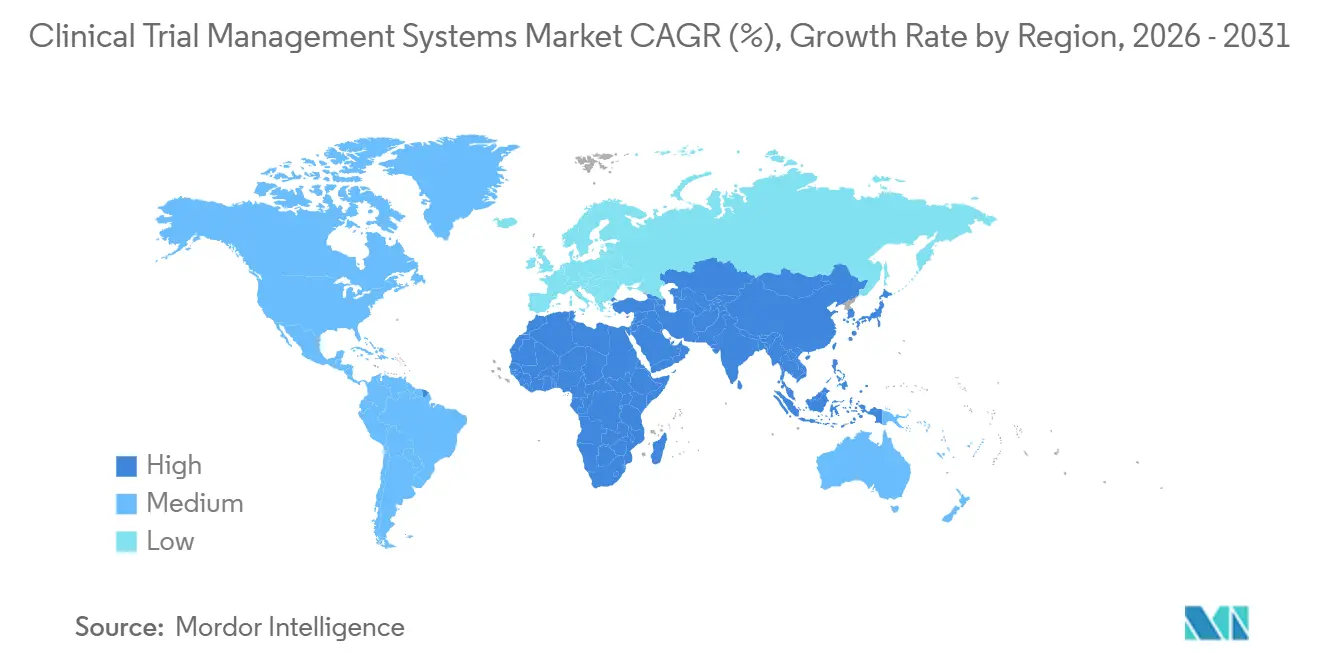
- By geography, North America commanded 49.68% of global share in 2025, yet Asia-Pacific is poised for the highest regional growth at a 15.24% CAGR to 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Clinical Trial Management Systems Market Trends and Insights
Driver Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Surge in cloud-based CTMS adoption for decentralized trials | 3.2% | Global, with early traction in North America and Western Europe | Medium term (2–4 years) |
| Rising volume and complexity of global clinical trials | 2.8% | Global, particularly Asia-Pacific and North America | Long term (≥ 4 years) |
| Regulatory mandates for real-time oversight (ICH-GCP E6-R3) | 2.5% | Global, led by ICH member regions (US, EU, Japan) | Short term (≤ 2 years) |
| CRO outsourcing boom among mid-size biopharma | 2.1% | North America and Europe, expanding to Asia-Pacific | Medium term (2–4 years) |
| AI-enabled predictive analytics improves enrollment velocity | 1.9% | North America and Europe, pilot deployments in Asia-Pacific | Medium term (2–4 years) |
| U.S.–China tech trade barriers pushing regional CTMS hosting | 1.1% | China, with spillover effects in APAC and select MEA markets | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Surge in Cloud-Based CTMS Adoption for Decentralized Trials
Decentralized designs rely on telehealth visits, home nursing, and wearable sensors, creating continuous data streams that on-premise systems cannot scale to ingest. Vendors such as Veeva report 28% year-over-year growth in Vault CTMS biotech subscriptions for fiscal 2025 as sponsors coordinate remote nursing and electronic consent workflows. FDA guidance finalized in 2024 requires auditable electronic workflows for off-site activities, a criterion best met by multi-tenant SaaS that ships version-controlled modules validated against 21 CFR Part 11.[1]Robert M. Califf, “Decentralized Clinical Trials Guidance,” U.S. Food and Drug Administration, fda.gov Oncology and rare-disease trials where eligible patients are geographically dispersed now default to cloud CTMS to curb screen-failure rates and compress enrollment timelines. These capabilities underpin the widening preference for cloud infrastructure across the clinical trial management system market.
Rising Volume and Complexity of Global Clinical Trials
Interventional registrations on ClinicalTrials.gov exceeded 480,000 by December 2025, up 9% from December 2024.[2]Rebecca J. Williams, “ClinicalTrials.gov Trends, Charts, and Maps: December 2025 Snapshot,” U.S. National Library of Medicine, clinicaltrials.gov Basket and umbrella protocols layer multiple disease cohorts into a single master protocol, driving exponential increases in task notifications, protocol amendments, and country-specific ethics workflows. Europe’s Clinical Trials Information System, fully enforced from 2024 onward, mandates structured XML uploads, a process automated by modern CTMS templates, to avoid validation errors.[3]Emer Cooke, “Clinical Trials Information System User Guide,” European Medicines Agency, ema.europa.eu As sponsors juggle 15-plus countries per phase-3 study, modular CTMS interfaces that embed multilingual informed-consent libraries and region-specific import-permit trackers have become indispensable, reinforcing demand across the clinical trial management system market.
Regulatory Mandates for Real-Time Oversight (ICH-GCP E6-R3)
The ICH issued its E6(R3) good-clinical-practice guideline in August 2024, pivoting from periodic on-site inspections toward continuous, centralized monitoring. Sponsors must now define critical-to-quality factors at study launch and surveil them in real time. CTMS suites answer this requirement by flagging protocol deviations within dashboards and auto-escalating serious adverse events to safety desks, preventing regulatory holds that can stall capital raises. Converging standards, FDA, EMA, and ISO 14155, cement CTMS as a compliance linchpin, solidifying its role inside the clinical trial management system market.
CRO Outsourcing Boom Among Mid-Size Biopharma
IQVIA’s 2025 report shows CRO revenue from biotech clients climbing 19% year-over-year, outpacing growth from large pharmaceutical companies. CROs deploy validated enterprise CTMS instances and onboard sponsor protocols via multi-tenant workspaces, spreading license and validation costs across dozens of clients. Labcorp added 1,200 new sponsor tenants during 2025, proof that variable-cost outsourcing resonates with cash-constrained biotech pipelines. This momentum places CROs among the fastest-expanding stakeholders within the clinical trial management system market.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High implementation and validation costs for SME sponsors | -1.8% | Global, most acute in North America and Europe | Short term (≤ 2 years) |
| Persistent data-privacy and cross-border-transfer restrictions | -1.5% | EU (GDPR), China (PIPL), Brazil (LGPD), with global spillover | Long term (≥ 4 years) |
| Shortage of CTMS-literate clinical-ops talent in emerging regions | -1.2% | Asia-Pacific (excluding Japan), Middle East & Africa, Latin America | Medium term (2–4 years) |
| Fragmented legacy IT stacks slowing interoperability | -1.0% | Global, particularly among large pharma with decades-old EDC systems | Medium term (2–4 years) |
| Source: Mordor Intelligence | |||
High Implementation and Validation Costs for SME Sponsors
Full-scope CTMS rollouts cost SMEs USD 0.5–2 million upfront, primarily for infrastructure, software licenses, and computer-system validation tied to 21 CFR Part 11 and EMA Annex 11. Validation commonly extends six to twelve months, during which sponsors must run duplicate paper workflows, delaying first-patient-in dates. SaaS subscriptions defer capex but escalate opex as user seats grow, limiting affordability for single-study biotechs and investigator-initiated trials. This cost drag tempers near-term penetration in the clinical trial management system market.
Persistent Data-Privacy and Cross-Border Restrictions
GDPR prohibits personal-data transfers to non-adequate jurisdictions without standardized clauses, creating carve-out CTMS instances for EU subjects. China’s PIPL compels onshore storage and security assessments before any outbound transfer. Brazil’s LGPD mirrors these requirements, and India’s Digital Personal Data Protection Act is slated to impose similar mandates once detailed rules publish. Maintaining siloed CTMS tenants inflates infrastructure complexity and slows consolidated analysis across the clinical trial management system market.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By System Type: Enterprise Platforms Anchor Spend as Site-Based Gains Momentum
Enterprise platforms accounted for 61.58% of 2025 revenue, underscoring big-pharma reliance on consolidated portfolios. These deployments integrate financial modules and quality systems to govern more than 200 concurrent trials. Conversely, site-based products are set to grow at a 15.89% CAGR through 2031 as decentralized designs hand greater autonomy to investigator sites. In oncology networks, community infusion clinics prefer lightweight CTMS that dovetail with electronic medical-record workflows, boosting vendor reach beyond the top-tier academic centers that dominated earlier procurement cycles. The clinical trial management system market size for site-based tools is projected to expand significantly as regulatory guidance now permits read-only sponsor access, lowering audit barriers. Over time, interoperability standards are likely to weave both deployment models into a single data fabric that minimizes duplicate entry.
Enterprise platforms will keep commanding budget priority for phase-3 registrational studies that involve multi-continent footprints and require uniform data visibility. Yet budget-constrained biotechs piloting first-in-human studies increasingly funnel spend toward modular, site-centric applications that go live within weeks and avoid enterprise change-control processes. As such, dual-platform coexistence will characterize the clinical trial management system market through the forecast window, with purchasing decisions hinging on trial complexity, site dispersion, and sponsor head-count scale.
By Delivery Mode: Web-Hosted Holds the Lead, but Cloud SaaS Accelerates
Web-hosted deployments held 54.66% of 2025 sales, a residual from early browser-based migrations. Cloud SaaS, however, is forecast to surge at 17.48% CAGR to 2031 as multitenant architectures roll out quarterly feature updates without user-side patching. The clinical trial management system market size attributed to SaaS models is expanding fastest among mid-size sponsors that lack dedicated IT staff. Regulatory guidance issued in 2024 clarified that logically segregated multitenant clouds can meet Part 11 validation if encryption, audit logging, and annual SOC 2 audits are in place, eliminating a lingering adoption barrier.
Despite the momentum, certain sponsors retain web-hosted tenants to comply with sovereign data laws that restrict cloud region choices. These hybrid patterns will persist, particularly where national regulators scrutinize foreign cloud providers. Even so, total cost-of-ownership math overwhelmingly favors SaaS, suggesting an eventual inversion in the delivery-mode revenue mix across the clinical trial management system market.
By Component: Software Dominates, Services Outpace on Growth
Licenses captured 67.27% of revenue in 2025. Yet services, validation, integration, and training, are expanding at a 16.32% CAGR, reflecting the skills gap around risk-based monitoring and AI enrollment modules. Validation providers generate high-margin workstreams drafting traceability matrices and executing operational-qualification scripts. Sponsors also contract change-management partners to train global site staff, particularly when rolling out predictive dashboards that require statistical literacy. Consequently, the clinical trial management system market size tied to services is widening in step with platform sophistication.
Software vendors increasingly bundle consulting hours within subscription tiers, blurring traditional revenue lines. However, pure-play service firms remain pivotal when large pharma embark on multi-year migrations from legacy on-premise estates. Integration blueprints that stitch CTMS records to electronic data capture, electronic trial master file, and pharmacovigilance systems underpin the next efficiency wave inside the clinical trial management system market.
By End User: Pharma Leads, CROs Surge
Pharmaceutical and biotech sponsors accounted for 39.52% of 2025 spend, reflecting their regulatory accountability and preference for direct system control. Nevertheless, contract research organizations are poised for a 17.43% CAGR through 2031 as venture-backed biotechs outsource operational heavy lifting to conserve burn rates. The clinical trial management system market size booked by CROs will therefore eclipse many single-sponsor segments over time.
Device manufacturers and academic institutes collectively supply the remainder, with digital therapeutic developers adopting CTMS to meet post-market evidence obligations. Platform trials funded by government grants are often the first adopters of adaptive-randomization features, foreshadowing broader toolchain convergence. Overall, roughly 60% of new CTMS study setups in 2026 originate from organizations that lack internal statistical programming teams, reinforcing outsourced operating models across the clinical trial management system market.
Geography Analysis
North America retained 49.68% of 2025 revenue, anchored by dense biotech clusters in Boston, the San Francisco Bay Area, and Research Triangle Park. The FDA’s 2024 decentralized-trial guidance accelerated demand for remote-ready CTMS, and the National Institutes of Health’s USD 1.5 billion Clinical Trials Transformation Initiative funds technology modernization at academic centers, further boosting the clinical trial management system market. Canada offers 15%-plus refundable R&D tax credits, spurring cross-border study placements, while Mexico’s 2024 ethics reforms shortened site approvals, though rural broadband gaps slow site adoption.
In Europe, Germany, France, and the United Kingdom collectively host half of EU phase-3 activity. The EMA’s requirement that all submissions flow through CTIS from 2024 onward amplified CTMS auto-population features. Central and Southern European countries, including Italy and Spain, gained share by streamlining multi-site approvals, nudging wider regional uptake within the clinical trial management system market.
Asia-Pacific is projected to post the highest regional CAGR at 15.24% through 2031. China’s Drug Administration Law amendments and strategic goal to fulfil 50% of global innovative drug filings domestically are central catalysts. Japan expanded English-language consultation services in 2024, easing cross-border collaboration, while India’s digital trial-approval portal reaccelerated site activations post-COVID. South Korea’s cell-therapy clusters and Australia’s 43.5% refundable tax offset sustain early-phase migration into the region, positioning Asia-Pacific as the long-term volume engine of the clinical trial management system market.
Competitive Landscape
The top vendors includes Veeva Systems, Oracle, IQVIA, Medidata (Dassault Systèmes), and Parexel, the market indicates moderate concentration. Cloud-native product roadmaps, seamless eClinical integrations, and therapeutic verticalization shape differentiation. Veeva cross-sells Vault CTMS into its CRM base, while Oracle leverages Clinical One’s AI patent (US11234567B2) to automate deviation detection. IQVIA exploits CRO scale to embed proprietary CTMS in turnkey outsourcing packages.
White-space opportunities exist in site-level SaaS, AI enrollment orchestration, and middleware that bridges legacy EDC schemas. Open-source entrants such as Castor EDC and OpenClinica appeal to investigator-initiated studies requiring cost sensitivity. Compliance credentials, ISO 27001, SOC 2, HITRUST, command premium pricing as regulators intensify auditing, particularly across multi-region deployments inside the clinical trial management system market.
M&A and strategic alliances continue. Medidata certified SimpleTrials as a verified partner in August 2025 to widen mid-market reach, while Veeva launched eSource in January 2026 to capture on-site data at the point of care. Partnerships between CROs and eClinical tech specialists, exemplified by Trialt and Medrio in February 2026, signal growing platform convergence trends aimed at compressing study start-up cycles and reducing total cost of ownership.
Clinical Trial Management Systems Industry Leaders
Oracle Corp.
Dassault Systmes (Medidata)
Veeva Systems
IQVIA Technologies
Parexel International
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2026: Trialt formed a strategic partnership with Medrio to integrate Medrio’s electronic data capture software, strengthening Trialt’s data-management capabilities.
- January 2026: Veeva Systems released Veeva eSource within SiteVault to eliminate paper at research sites by enabling direct data capture.
- August 2025: SimpleTrials CTMS joined Viedoc’s Verified Partner Program, enhancing integrated CTMS-EDC workflows for mid-sized sponsors.
Research Methodology Framework and Report Scope
Market Definitions and Key Coverage
Our study defines the global Clinical Trial Management System (CTMS) market as all purpose-built software platforms and allied implementation services that plan, track, and report operational, financial, and regulatory activities across single-site, multi-site, or decentralized clinical trials. The boundary spans on-premise, web, and cloud deployments that deliver core modules such as study planning, participant management, budgeting, and reporting.
Scope Exclusions: Stand-alone electronic trial master file (eTMF) solutions, electronic data capture, and generic project-management tools are kept outside the count to avoid double counting.
Segmentation Overview
- By System Type
- Enterprise-based CTMS
- Site-based CTMS
- By Delivery Mode
- Web-based (Hosted) CTMS
- Cloud-based (SaaS) CTMS
- On-premise CTMS
- By Component
- Software
- Services
- By End User
- Pharmaceutical & Biotechnology Companies
- Contract Research Organizations
- Medical Device Manufacturers
- Academic & Research Institutes
- By Geography
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Rest of Europe
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia-Pacific
- Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
- South America
- Brazil
- Argentina
- Rest of South America
- North America
Detailed Research Methodology and Data Validation
Primary Research
Interviews and structured surveys with trial operations heads at sponsors, CRO IT directors, product managers at CTMS vendors, and regional regulators across North America, Europe, and Asia Pacific validated adoption rates, average subscription pricing, and service attach ratios, while filling gaps in under-reported emerging regions.
Desk Research
We collated foundational data from tier-one sources such as the World Health Organization ICTRP database, ClinicalTrials.gov, EMA trial registry, the Pharmaceutical Research and Manufacturers of America statistics portal, and filings submitted to the US SEC. Annual reports and 10-Ks from major sponsors, contract research organizations, and pure-play CTMS vendors helped us approximate trial volumes and software revenue splits. Paid intelligence platforms, notably D&B Hoovers for company financials and Dow Jones Factiva for press coverage, supplied directional figures. The sources listed here are illustrative; many additional documents informed the evidence base.
Market-Sizing & Forecasting
A top-down model starts with active and planned trial counts by phase and geography, which are then multiplied by weighted penetration rates for enterprise and site-level CTMS. Results are corroborated with selective bottom-up checks such as sampled vendor revenue roll-ups and average selling price times user seat tallies. Key inputs include new trial initiations, decentralized trial share, software-as-a-service pricing erosion, CRO outsourcing intensity, and regulatory technology mandates. Multivariate regression on trial volume, R&D spend, and cloud IT spend underpins the 2025-2030 forecast, while scenario analysis tests upside and downside adoption curves.
Data Validation & Update Cycle
Outputs pass multi-step variance checks against independent series before senior analyst sign-off. Reports refresh every twelve months, with interim updates triggered by material events such as major regulatory guidance or platform mergers, and a final data sweep occurs directly before client delivery.
Why Mordor's Clinical Trial Management Systems Baseline Commands Confidence
Published CTMS estimates often diverge because firms adopt different functional scopes, pricing ladders, and refresh cadences.
Our disciplined variable selection and explicit segment boundaries keep the baseline steady, yet transparent.
Benchmark comparison
| Market Size | Anonymized source | Primary gap driver |
|---|---|---|
| USD 2.44 Bn (2025) | Mordor Intelligence | - |
| USD 2.00 Bn (2024) | Global Consultancy A | Counts only web-based solutions and classifies eTMF separately |
| USD 1.41 Bn (2024) | Regional Consultancy B | Uses static 2020 pricing and omits implementation services |
| USD 1.26 Bn (2024) | Industry Journal C | Limits geography to North America and Europe, missing APAC growth |
The comparison shows that our balanced inclusion of deployment modes, service revenues, and truly global trial activity delivers a dependable reference point for decision-makers, while still allowing users to trace every assumption back to observable metrics and repeatable steps.


Key Questions Answered in the Report
How fast is the clinical trial management system market expected to grow through 2031?
It is projected to advance at a 13.62% CAGR from 2026 to 2031, reaching USD 5.02 billion in value.
Which delivery model is gaining the most traction?
Cloud-based SaaS deployments are expanding at a 17.48% CAGR as sponsors favor elastic scalability and automated updates.
Why are contract research organizations increasing CTMS spend?
CROs consolidate multi-sponsor workloads onto enterprise CTMS platforms, spreading license costs and accelerating study start-up, resulting in a 17.43% CAGR outlook.
What regulatory shift is driving real-time monitoring features?
The ICH-GCP E6(R3) guideline finalized in 2024 mandates continuous risk-based oversight, pushing vendors to integrate real-time deviation and safety alerting.
Which region offers the fastest growth opportunity?
Asia-Pacific, led by China, India, and South Korea, is forecast to post a 15.24% CAGR due to supportive reforms and expanding trial site infrastructure.
Page last updated on:









