Fetal Bovine Serum Market Size and Share
Fetal Bovine Serum Market Analysis by Mordor Intelligence
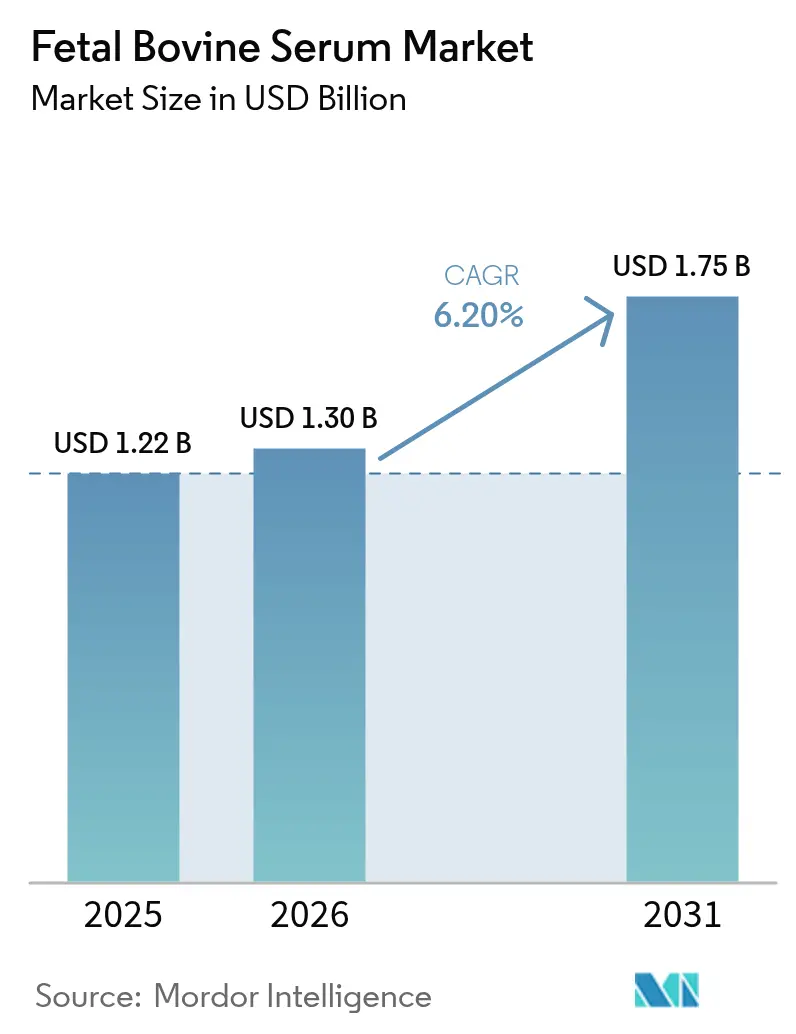
The fetal bovine serum market size is projected to expand from USD 1.22 billion in 2025 and USD 1.30 billion in 2026 to USD 1.75 billion by 2031, registering a CAGR of 6.2% between 2026 and 2031. Structural demand comes from cell- and gene-therapy pipelines whose clinical assets climbed to 3,049 in Q3 2025, locking in serum use well beyond the current validation cycle. Tight raw-material supply reinforces pricing power; the U.S. cattle inventory fell to 87.2 million head in January 2025, the lowest since 1951. Specialized grades such as charcoal/dextran-stripped serum command premium pricing because they resolve hormone-related assay noise, while stem-cell-qualified lots are gaining share as regenerative-medicine trials scale. Competitive strategy has bifurcated: global suppliers are buying collection networks to secure inputs, whereas regional traders compete on origin traceability.
Key Report Takeaways
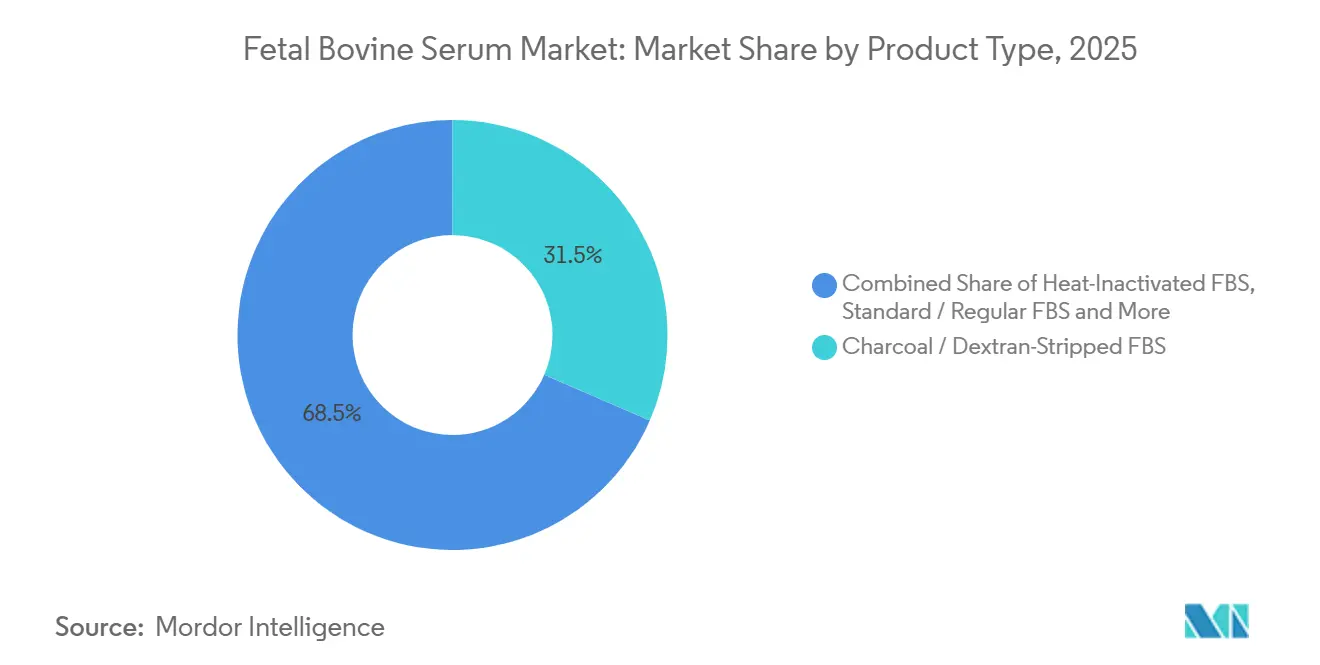
- By product type, charcoal/dextran-stripped serum led with 31.55% of the fetal bovine serum market share in 2025, while stem-cell-qualified FBS is advancing at a 7.25% CAGR to 2031.
- By application, biopharmaceutical production accounted for 32.53% share of the fetal bovine serum market size in 2025, cell-culture maintenance & expansion is advancing at a 6.75% CAGR to 2031.
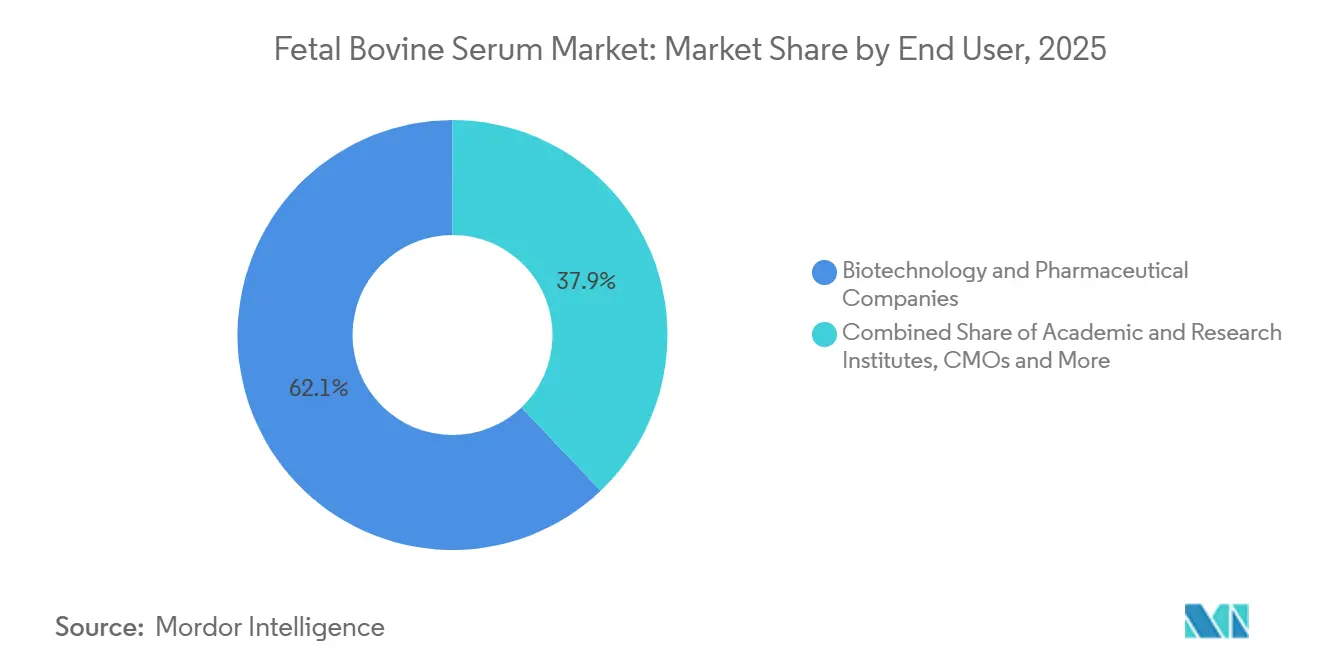
- By end user, biotechnology and pharmaceutical companies controlled 62.15% of 2025 revenue, while academic institutes are advancing at a 6.82% CAGR to 2031.
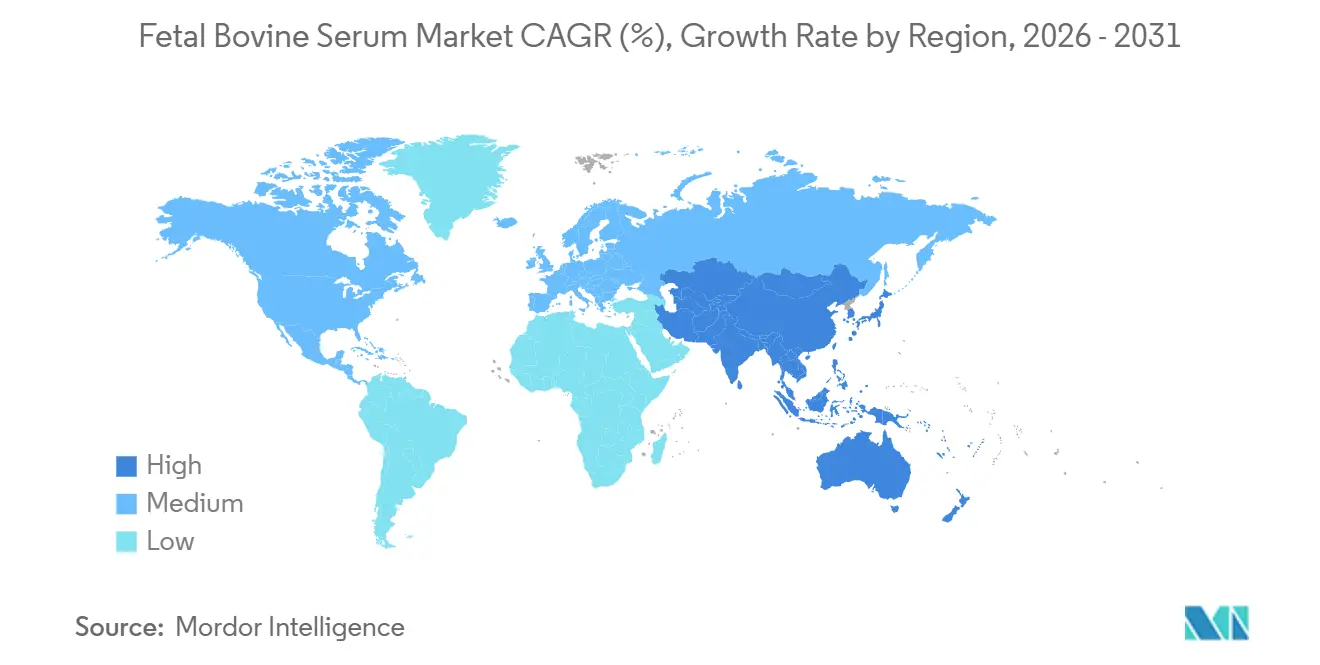
- By geography, North America captured 41.55% of 2025 revenue; Asia-Pacific is projected to grow at 7.22% through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Fetal Bovine Serum Market Trends and Insights
Driver Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rapid biopharma scale-up post-COVID-19 | +1.8% | Global, with concentration in North America, Europe, and Asia-Pacific | Medium term (2-4 years) |
| Expanding cell & gene-therapy pipelines | +1.5% | North America and Europe lead; Asia-Pacific emerging | Long term (≥ 4 years) |
| Growth in animal & human vaccine output | +1.0% | Global, with emphasis on Asia-Pacific and South America | Medium term (2-4 years) |
| Rise of contract cell-culture manufacturing (CMOs/CROs) | +0.9% | North America and Asia-Pacific | Medium term (2-4 years) |
| Cultured-meat R&D funding surge | +0.4% | North America, Europe, and select Asia-Pacific hubs | Long term (≥ 4 years) |
| U.S. cattle-herd decline tightening FBS supply | +0.6% | Global supply impact, most acute in North America | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Rapid Biopharma Scale-Up Post-COVID-19
Capacity built for pandemic biologics is still online and requires routine seed-train maintenance with FBS-supplemented media. A USD 1.6 billion single-use bioreactor expansion announced by Cytiva in 2025 will use serum for process-development runs until at least 2029. Samsung Biologics’ Plant 5 brought 180,000 liters of perfusion volume online in late 2024 and confirms that seed trains remain serum dependent even when production reactors use defined media[1]Samsung Biologics IR Team, “Investor Presentation Q4 2025,” Samsung Biologics, samsungbiologics.com. CMOs, which held 38% of global biologics capacity in 2025, follow client-mandated protocols that specify FBS grades and lot testing, effectively locking in baseline volume. These conditions create a floor under fetal bovine serum market demand despite innovation in serum-free systems.
Expanding Cell & Gene-Therapy Pipelines
Six new cell-therapy indications cleared the U.S. FDA in 2025, each requiring commercial-scale ex vivo culture steps that consume serum. Novartis disclosed that Kymriah sites used 12,000 liters of stem-cell-qualified FBS in 2024, a 19% rise over 2023. Regulatory precedent discourages mid-trial media switches; comparability risks deter sponsors from abandoning validated serum processes. The International Society for Cell & Gene Therapy’s 2025 guidelines call for FBS lot pre-qualification, further embedding serum into late-stage trials. As a result, the fetal bovine serum market receives durable volumes from advanced-therapy pipelines.
Growth in Animal & Human Vaccine Output
Global influenza vaccine capacity climbed to 1.53 billion doses in 2024, up from 1.42 billion in 2023, and primary cell lines used for master-seed preparation still require serum. India’s Serum Institute consumes 8,000 liters of FBS annually for cell-bank maintenance. Brazil’s agriculture ministry documented a 12% rise in foot-and-mouth disease doses in 2024, which lifts veterinary vaccine serum needs. Large animal-health firms keep legacy lines on FBS because revalidation is costly and regulators have not mandated a switch, sustaining incremental growth in the fetal bovine serum market.
Rise of Contract Cell-Culture Manufacturing
Lonza’s USD 1.2 billion CDMO acquisition in 2025 highlights the premium on flexible cell-culture platforms accepting client-specified media. Charles River Laboratories recorded 16% biologics-testing revenue growth in Q3 2025, driven by FBS lot testing for CMO customers. WuXi Biologics qualified 47 serum lots in 2024 to meet diverse sponsor specs. Each additional CMO in a development chain multiplies procurement touchpoints, pushing up fetal bovine serum market volumes even as individual sponsors plan serum-free conversions.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Price volatility linked to beef-industry cycle | -0.8% | Global, most acute in North America and South America | Short term (≤ 2 years) |
| Ethical concerns & regulatory scrutiny | -0.6% | Europe and North America lead; Asia-Pacific following | Medium term (2-4 years) |
| Acceleration of serum-free-media adoption | -1.2% | Global, with fastest uptake in Europe and North America | Long term (≥ 4 years) |
| Recombinant albumin substitutes gaining traction | -0.5% | North America and Europe | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Ethical Concerns & Regulatory Scrutiny
The European Medicines Agency now requires individual-herd traceability for serum used in advanced-therapy medicinal products, raising compliance costs for small suppliers[2]European Medicines Agency, “Guideline on TSE/BSE Testing for ATMPs,” EMA, ema.europa.eu. USDA-APHIS proposed similar oversight for U.S. exporters in June 2025. The University of California system demands justification for every FBS purchase in grant-funded research. These measures do not eliminate demand but slow the fetal bovine serum industry growth rate as developers weigh long-term reputational risk.
Acceleration of Serum-Free-Media Adoption
Thermo Fisher’s serum-free media revenue outpaced FBS sales in 2025 by a ratio of 24% to 9%. Merck KGaA bought Mirus Bio for USD 290 million to couple transfection reagents with defined media, easing the path away from serum. Sartorius derived 22% of 2025 bioprocess revenue from chemically defined platforms and targets 35% by 2028. Early-stage projects move first because switching costs are low, placing a ceiling on very long-term expansion of the fetal bovine serum market.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Product Type: Specialized Grades Capture Premium Pricing
Stem-cell-qualified serum is forecast to grow 7.25% annually, outstripping the overall fetal bovine serum market size, because regenerative trials demand rigorous contaminant testing[3]National Institutes of Health, “Stem Cell Research Funding FY 2025,” NIH, nih.gov. Charcoal/dextran-stripped lots already control 31.55% of 2025 revenue and remain entrenched in hybridoma and endocrine research. Heat-inactivated and dialyzed variants serve routine culture and metabolic assays, while exosome-depleted grades gain traction after 2025 ISEV guidelines. Gamma-irradiated and low-IgG options protect viral-vaccine and immunology workflows, reinforcing a price hierarchy within the fetal bovine serum market.
Commodity-grade serum faces margin pressure as defined media substitutes arrive, yet premium grades retain bargaining power. ATCC’s 2025 authentication standards require full certificates of analysis, disadvantaging low-cost suppliers. Mesenchymal stem-cell-qualified lots remain supply constrained because fewer than ten producers meet multilineage-differentiation specs. Embryonic stem-cell protocols are shifting to defined media, but legacy feeder-dependent lines still specify FBS. This bifurcation signals widening spread in the fetal bovine serum market between cost-sensitive and performance-critical niches.

By Application: Biopharmaceutical Production Anchors Demand
Biopharmaceutical production delivered 32.53% of 2025 revenue and underpins current fetal bovine serum market size. However, its share may plateau because new facilities start directly with serum-free platforms. Cell-culture maintenance and expansion is projected to grow 6.75% through 2031 as CMOs scale client-bank inventories. Vaccine manufacturing, both human and veterinary, retained 18% of volume in 2025, with India and Brazil leading orders.
Stem-cell research gained momentum on the back of USD 2.1 billion in NIH awards in fiscal 2025, lifting specialized FBS volumes. Diagnostics and IVF remain niche yet price-inelastic, demanding ultra-low endotoxin lots for assay reproducibility and embryo viability. Hybridoma antibody production is slowly transitioning to defined media, but historical cell banks ensure persistent though tapering contributions to fetal bovine serum market demand.
By End User: Biotech and Pharma Dominance Masks Academic Growth
Biotechnology and pharmaceutical companies owned 62.15% of 2025 revenue, reflecting legacy manufacturing lines that still require FBS seed trains. Academic and research institutes are expected to post a 6.82% CAGR because grant-funded exploratory work stays with validated serum protocols. CMOs procure in bulk, passing costs through to sponsors, which keeps them less price sensitive than captive labs.
Cell banks such as ATCC continue to draw modest but steady lots for master-cell collection. Diagnostic labs and veterinary clinics contribute minor but stable orders. The segmentation confirms that the fetal bovine serum market draws its resilience from a mix of regulatory inertia in commercial plants and experimentation needs in academia.

Geography Analysis
North America held 41.55% of 2025 revenue, but drought-driven cattle-herd contraction threatens supply even as local biologics demand rises. Few Midwest abattoirs with GMP-grade cold chains create single-point-of-failure risk, nudging buyers toward South American collections secured by acquisitions such as Thermo Fisher’s 2025 deal. Serum-free adoption is fastest in the United States and Canada, implying regional growth will trail the global fetal bovine serum market by the late forecast period.
Asia-Pacific is projected to grow 7.22% through 2031 on expanding biosimilar output and vaccine capacity. China relies on imported serum because local suppliers struggle with TSE compliance, while India’s vaccine hubs keep seed-train steps on FBS. Japan’s conditional approval pathway boosts regenerative-medicine applications, and South Korean CMOs add bioreactor capacity that still starts with serum seed trains. Australia and New Zealand collect high-grade serum but lack herd volume to satisfy regional growth, driving cross-border logistics complexity.
Europe maintains demand through mature pharma clusters and strict traceability rules that favor vertically integrated suppliers. Germany and the United Kingdom anchor R&D needs for specialty grades, while France sustains vaccine lines that remain serum dependent. South America is primarily a supply region; Brazil’s 220 million-head herd underpins export flows though domestic demand is moderate. Africa and the Middle East contribute marginal consumption because local biomanufacturing infrastructure is limited.
Competitive Landscape
The top five suppliers—Thermo Fisher Scientific, Merck KGaA, Sartorius, Cytiva, and Corning—accounted for a significant share of 2025 revenue, indicating moderate concentration in the fetal bovine serum market. Vertically integrated players control collection, processing, and global distribution, allowing them to meet new traceability mandates at lower incremental cost. Large vendors are also hedging with serum-free portfolios; Sartorius derived 22% of 2025 bioprocess revenue from chemically defined media and plans capacity additions in Germany.
Regional suppliers, numbering 30 to 40, compete on origin claims and spot availability but struggle to meet enhanced regulatory audits under EMA and FDA guidance. Exosome-depleted and stem-cell-qualified niches offer margin opportunity because fewer than ten producers achieve consistent specs defined by ISEV and ISCT guidelines. Upstream control is strategic; Thermo Fisher’s South American buyout protects access as North American cattle herds shrink.
Technological differentiation is emerging through blockchain lot tracking and inline contaminant analytics, which resonate with risk-averse pharma buyers. Recombinant albumin suppliers such as Albumedix present long-run substitution risk, but limited production scale and high cost keep immediate impact modest. Overall, the fetal bovine serum industry is set for gradual consolidation as traceability and biosecurity costs squeeze small traders.
Fetal Bovine Serum Industry Leaders
Thermo Fisher Scientific Inc.
Merck KGaA (Sigma-Aldrich)
Sartorius AG
Danaher Corp. (Cytiva)
Corning Incorporated
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- January 2026: Sartorius committed EUR 120 million (USD 130 million) to expand serum-free media production in Göttingen, targeting a 50% capacity lift by Q4 2027.
- November 2024: Gemini Bioproducts acquired selected FBS product rights and inventory from Bio-Techne to broaden its premium serum portfolio.
Global Fetal Bovine Serum Market Report Scope
Fetal bovine serum (FBS) is the liquid fraction of clotted blood from fetal calves, depleted cells, fibrin, and clotting factors. It contains a high amount of nutritional and macromolecular factors essential for cell growth. FBS also contains a variety of small molecules like amino acids, sugars, lipids, and hormones. It is commonly used for biotechnological research, including cancer research, the development of biopharmaceuticals, and the production of human and animal vaccines. The fetal bovine serum market is segmented by product type, application, end-user, and geography.
The segmentation of the fetal bovine serum market by product type includes standard/regular FBS, heat-inactivated FBS, charcoal/dextran-stripped FBS, dialyzed FBS, chromatographically-purified (low-IgG) FBS, stem-cell-qualified FBS, exosome-depleted FBS, and gamma-irradiated FBS. By application, the market is segmented into biopharmaceutical production, vaccine manufacturing (human and animal), cell-culture maintenance and expansion, stem-cell research and therapy, diagnostics/IVD, IVF and reproductive medicine, exosome studies, and antibody production and hybridoma. By end user, the segmentation includes biotechnology and pharmaceutical companies, academic and research institutes, CMOs and CROs, cell banks and biorepositories, diagnostic laboratories, and veterinary clinics and research. By geography, the market is divided into North America, Europe, Asia-Pacific, the Middle East and Africa, and South America. The report offers the value (in USD) for the above-mentioned segments.
| Standard / Regular FBS | |
| Heat-Inactivated FBS | |
| Charcoal / Dextran-Stripped FBS | |
| Dialyzed FBS | |
| Chromatographically-Purified (Low-IgG) FBS | |
| Stem-Cell-Qualified FBS | Embryonic SC-Qualified |
| Mesenchymal SC-Qualified | |
| Exosome-Depleted FBS | |
| Gamma-Irradiated FBS |
| Biopharmaceutical Production |
| Vaccine Manufacturing (Human & Animal) |
| Cell-Culture Maintenance & Expansion |
| Stem-Cell Research & Therapy |
| Diagnostics / IVD |
| IVF & Reproductive Medicine |
| Exosome Studies |
| Antibody Production & Hybridoma |
| Biotechnology & Pharmaceutical Companies |
| Academic & Research Institutes |
| CMOs & CROs |
| Cell Banks & Biorepositories |
| Diagnostic Laboratories |
| Veterinary Clinics & Research |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product Type | Standard / Regular FBS | |
| Heat-Inactivated FBS | ||
| Charcoal / Dextran-Stripped FBS | ||
| Dialyzed FBS | ||
| Chromatographically-Purified (Low-IgG) FBS | ||
| Stem-Cell-Qualified FBS | Embryonic SC-Qualified | |
| Mesenchymal SC-Qualified | ||
| Exosome-Depleted FBS | ||
| Gamma-Irradiated FBS | ||
| By Application | Biopharmaceutical Production | |
| Vaccine Manufacturing (Human & Animal) | ||
| Cell-Culture Maintenance & Expansion | ||
| Stem-Cell Research & Therapy | ||
| Diagnostics / IVD | ||
| IVF & Reproductive Medicine | ||
| Exosome Studies | ||
| Antibody Production & Hybridoma | ||
| By End User | Biotechnology & Pharmaceutical Companies | |
| Academic & Research Institutes | ||
| CMOs & CROs | ||
| Cell Banks & Biorepositories | ||
| Diagnostic Laboratories | ||
| Veterinary Clinics & Research | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the projected value of the fetal bovine serum market in 2031?
It is forecast to reach USD 1.75 billion by 2031 on a 6.2% CAGR.
Which product grade currently leads sales?
Charcoal/dextran-stripped serum held 31.55% of 2025 revenue.
Why does cell- and gene-therapy manufacturing sustain serum demand?
Validated clinical protocols rely on FBS for ex vivo expansion, and switching would require costly revalidation.
Which region is expected to register the fastest growth?
Asia-Pacific is projected to expand at a 7.22% CAGR between 2026 and 2031.
Page last updated on:









