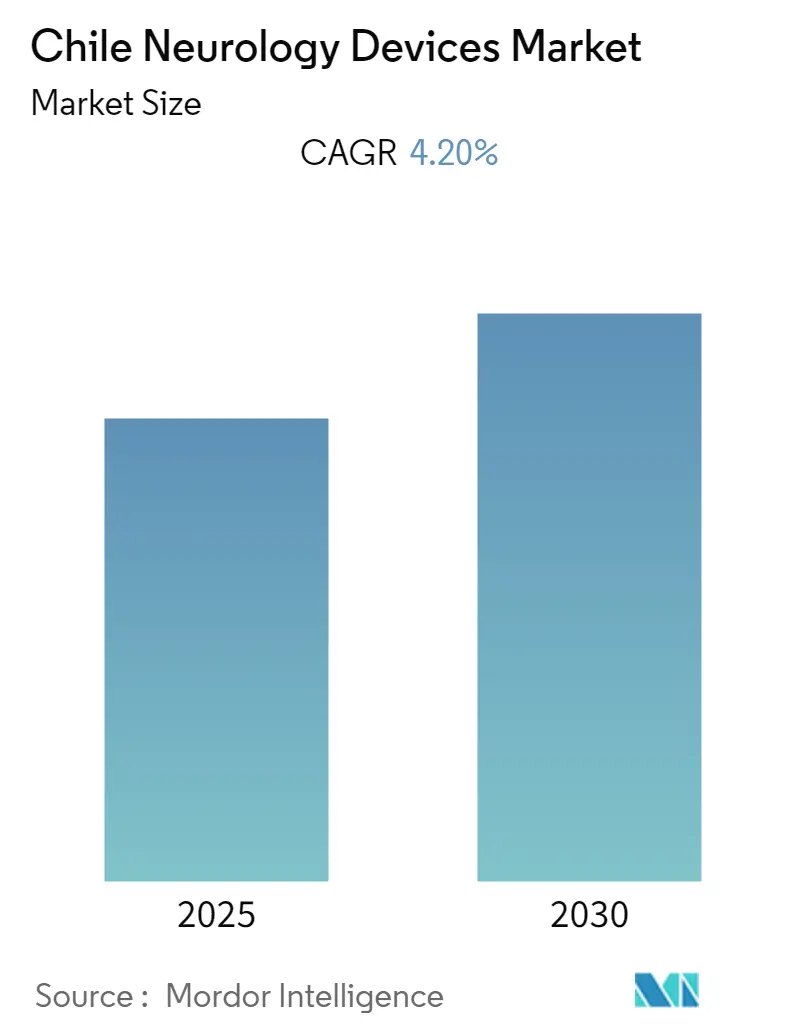
Chile Neurology Devices Market Analysis by Mordor Intelligence
The Chile Neurology Devices Market is expected to register a CAGR of 4.2% during the forecast period.
The COVID-19 pandemic significantly impacted neurology care in Chile during the early phase, which in turn impacted the usage of neurology devices as well. For instance, according to an article published by PubMed in April 2021, a study was conducted in a university hospital in Chile, which showed that at least 96 patients admitted due to COVID-19 had one or more neurological complications. Thus, the COVID-19 pandemic significantly impacted the market growth initially; however, as the pandemic has currently subsided, neurology care is going back to normal in the country. Hence, the studied market is expected to have proper growth during the forecast period of the study.
The key factors propelling the growth of the studied market are the increasing incidence of neurological disorders, the increasing R&D expenditure, and huge investments by private players in neurology devices.
The rising prevalence of neurological disorders such as Parkinson's disease, Alzheimer's disease, epilepsy, and brain diseases, among others, and the increasing research related to them, is the major factor driving the market growth. For instance, according to the data updated by ClinicalTrials.gov in April 2023, there are currently 2 active clinical trials of Parkinson's disease in Chile. Thus, the rise in research and development in Chile related to neurological disorders such as Parkinson's disease is expected to boost the market growth.
Moreover, according to an article published by the Office of the Assistant Secretary for Planning and Evaluation in December 2022, a harmonized cognitive assessment protocol (HCAP) project has been implemented in various countries for assessing cognitive functions and aging in Alzheimer's patients. Chile is included in the list of countries.
Thus, the aforementioned factors, such as the rising prevalence of neurological disorders and the increasing initiatives by various organizations, are expected to boost the market growth during the forecast period of the study. However, the high cost of equipment and stringent regulatory scenario are expected to impede market growth.
Chile Neurology Devices Market Trends and Insights
Sacral Nerve Stimulation Devices Segment is Expected to Have a Significant Market Share Over the Forecast Period
The sacral nerve stimulation devices are mostly used for patients for whom drug therapy or other types of medications are not successful. It is performed with the help of a small device, which is capable of sending electrical impulses to the targeted sacral nerves located in the lower back region. These devices are found to be successful in treating bladder problems. Hence, with increasing incidences of overactive bladder and bowel incontinence, the sacral nerve stimulation devices segment is expected to grow during the forecast period.
According to an article published by a few Chilean authors in SpringerLink in February 2022, electric nerve stimulation procedures such as sacral nerve stimulation are widely used for the treatment of overactive bladder in patients. Thus, the increasing use of sacral nerve stimulation in the treatment of overactive bladder is expected to boost segment growth during the forecast period of the study.
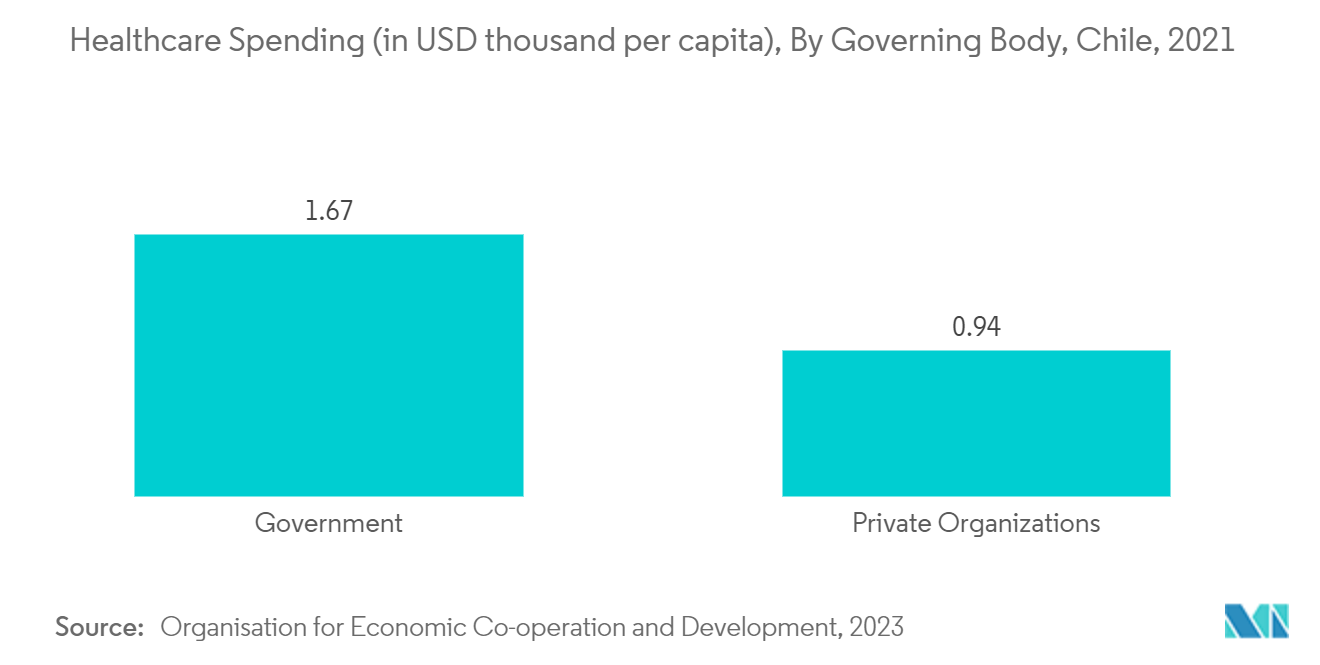
Furthermore, according to the data published by the United Nations Population Fund Dashboard in 2022, an estimated 13% of the population in Chile will be aged 65 years and older in 2022. Thus, the rising geriatric population is expected to enhance the market growth, as old age is often associated with diseases that require sacral nerve stimulation treatment. Moreover, the rising healthcare spending in the country is also expected to boost segment growth.
Hence, the aforementioned factors, such as the rising prevalence of overactive bladder and urinary incontinence and the rising healthcare expenditure, are expected to boost segment growth during the forecast period of the study.


Deep Brain Stimulation Devices Segment is Expected to Have a Significant Growth During the Forecast Period
Deep brain stimulation (DBS) is a neurosurgical technique that regulates neuron activity by using internal pulse generators to through electrodes in specific target areas of the brain. These devices are used for the treatment of various neurological diseases such as dystonia, Alzheimer's disease, Parkinson's disease, and epilepsy, among others. Thus, the rising prevalence of such neurological disorders in Chile and the rising geriatric population is expected to boost the segment growth.
For instance, according to an article published by Chilean authors in MDPI in September 2022, Drug-induced oromandibular dystonia encompasses a wide variety of hyperkinetic and hypokinetic extrapyramidal involuntary movements, manifesting in the form of dystonia in patients, and it is often found that oromandibular movement disorders were consistent with dystonia. Thus, the prevalence of dystonia in the country associated with a variety of movements is expected to boost segment growth, as deep brain stimulation is often used for the treatment of neurological disorders such as dystonia.
Furthermore, the rising geriatric population in the country is also expected to enhance segment growth, as elderly people are often associated with a high prevalence of neurological disorders. For instance, according to the data updated by the UN World Population Prospects in 2022, an estimated 3,477 thousand people were more than 60 years old in Chile in 2021.
Hence, the aforementioned factors, such as the rising prevalence of dystonia and Parkinson's disease and the rising geriatric population, are expected to boost the segment growth during the forecast period of the study.


Competitive Landscape
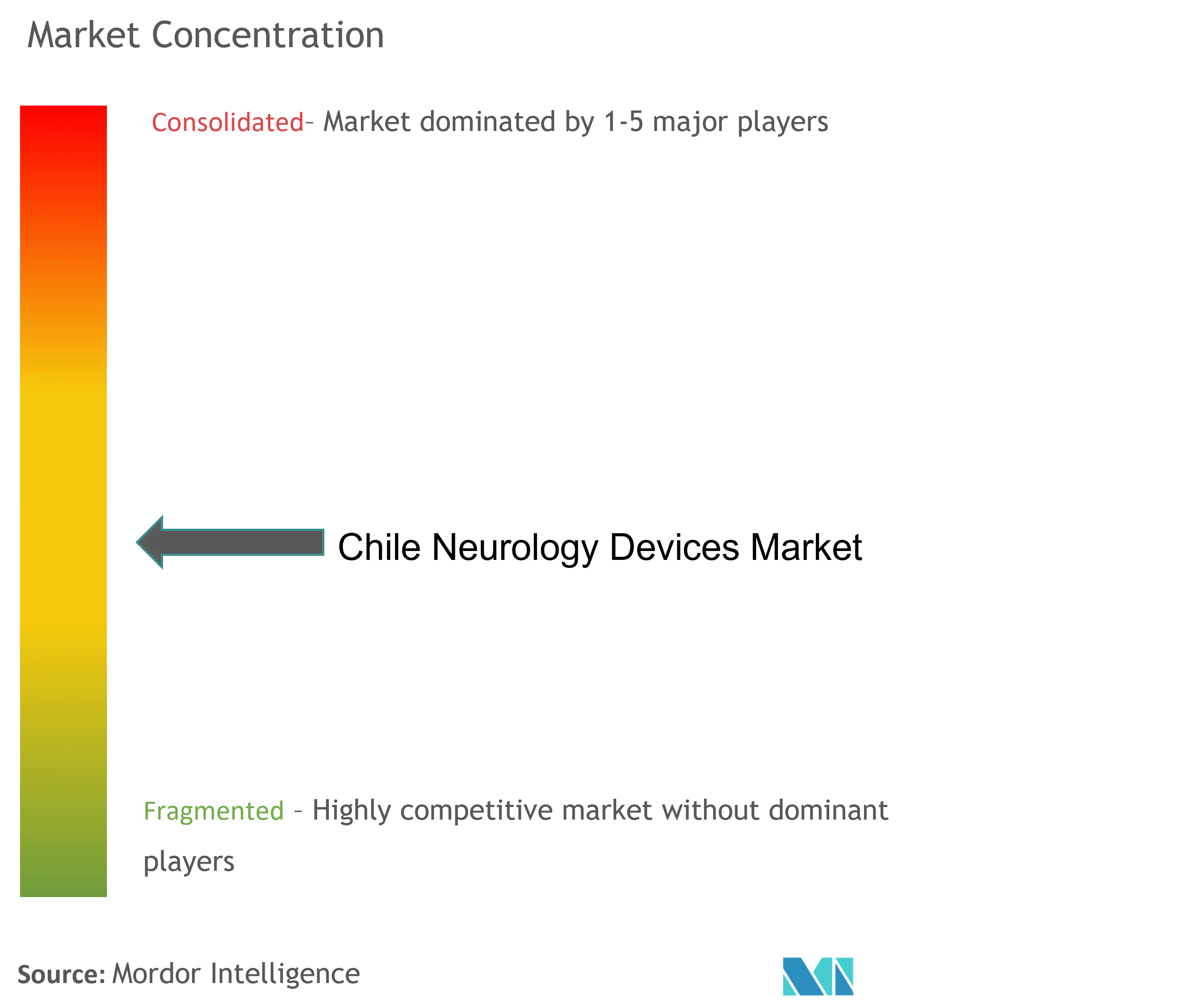
The Chile neurology devices market is moderately fragmented in nature. There are market participants involved in R&D for the development of advanced stimulation and neuromodulation techniques with minimally invasive procedures that are preferable to the patients. Furthermore, many private companies are showing great interest in developing novel neurological devices to earn huge profits. Some of the key market players are B. Braun SE, Medtronic PLC, Abbott Laboratories, Nihon Kohden Corporation, and Stryker Corporation, among others.
Chile Neurology Devices Industry Leaders
Stryker Corporation
Medtronic PLC
Abbott Laboratories
Nihon Kohden Corporation
B. Braun SE
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- July 2022: A clinical trial was in the recruiting stage in Chile by the Universidad Católica del Maule to check the efficacy of automated mechanical peripheral stimulation in Parkinson's disease patients.
- March 2022: A clinical trial was in the recruiting stage in Chile by the Corporación de Rehabilitación Club de Leones Cruz del Sur for assessing transcranial electrical stimulation and auditory stimulation during walking in Parkinson's disease patients.


Chile Neurology Devices Market Report Scope
As per the scope of the report, neurological devices are medical devices that help to diagnose, prevent, and treat a variety of neurological disorders and conditions, such as Alzheimer's disease, Parkinson's disease, major depression, and traumatic brain injury. The Chile neurology devices market is segmented by type of device (cerebrospinal fluid management device, interventional neurology device (interventional/surgical simulators, neuro thrombectomy devices, carotid artery stents, embolic coils, and support devices)), neurosurgery device (neuro endoscopes, stereotactic systems, aneurysm clips, and other neurosurgery devices), neurostimulation device (spinal cord stimulation devices, deep brain stimulation devices, sacral nerve stimulation devices, and other neurostimulation devices), and other types of devices). The report offers the value (in USD) for the above segments.
| Cerebrospinal Fluid Management Devices | |
| Interventional Neurology Devices | Interventional/Surgical Simulators |
| Neurothrombectomy Devices | |
| Carotid Artery Stents | |
| Embolic Coils | |
| Support Devices | |
| Neurosurgery Devices | Neuroendoscopes |
| Stereotactic Systems | |
| Aneurysm Clips | |
| Other Neurosurgery Devices | |
| Neurostimulation Devices | Spinal Cord Stimulation Devices |
| Deep Brain Stimulation Devices | |
| Sacral Nerve Stimulation Devices | |
| Other Neurostimulation Devices | |
| Other Types of Devices |
| By Type of Device | Cerebrospinal Fluid Management Devices | |
| Interventional Neurology Devices | Interventional/Surgical Simulators | |
| Neurothrombectomy Devices | ||
| Carotid Artery Stents | ||
| Embolic Coils | ||
| Support Devices | ||
| Neurosurgery Devices | Neuroendoscopes | |
| Stereotactic Systems | ||
| Aneurysm Clips | ||
| Other Neurosurgery Devices | ||
| Neurostimulation Devices | Spinal Cord Stimulation Devices | |
| Deep Brain Stimulation Devices | ||
| Sacral Nerve Stimulation Devices | ||
| Other Neurostimulation Devices | ||
| Other Types of Devices | ||


Key Questions Answered in the Report
What is the current Chile Neurology Devices Market size?
The Chile Neurology Devices Market is projected to register a CAGR of 4.2% during the forecast period (2025-2030)
Who are the key players in Chile Neurology Devices Market?
Stryker Corporation, Medtronic PLC, Abbott Laboratories, Nihon Kohden Corporation and B. Braun SE are the major companies operating in the Chile Neurology Devices Market.
What years does this Chile Neurology Devices Market cover?
The report covers the Chile Neurology Devices Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Chile Neurology Devices Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
Chile Neurology Devices Market Report
Statistics for the 2025 Chile Neurology Devices market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Chile Neurology Devices analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.





