Global Central Nervous System Biomarkers Market Size and Share
Global Central Nervous System Biomarkers Market Analysis by Mordor Intelligence
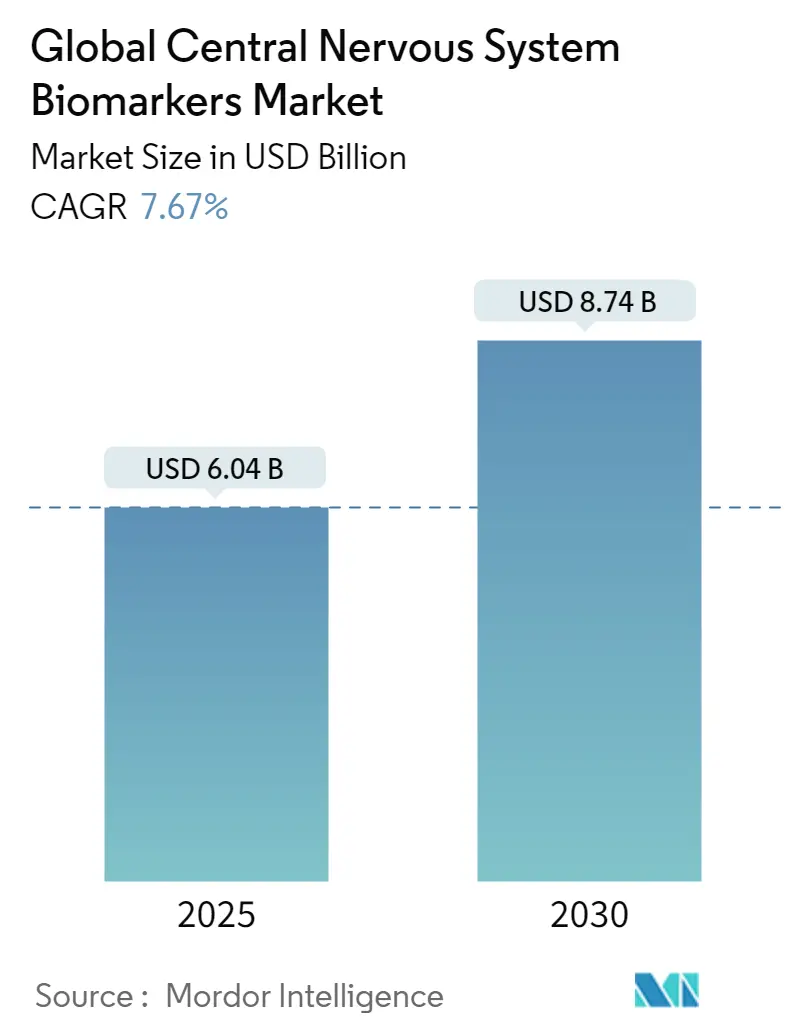
The Global Central Nervous System Biomarkers Market size is estimated at USD 6.04 billion in 2025, and is expected to reach USD 8.74 billion by 2030, at a CAGR of 7.67% during the forecast period (2025-2030).
The neurological biomarkers market is experiencing significant transformation driven by the increasing prevalence of neurological disorders globally. The rising burden of conditions like Alzheimer's disease and dementia is creating unprecedented demand for early detection and monitoring solutions. According to Alzheimer's Europe projections, dementia cases in France alone are expected to double to approximately 2,236,682 by 2050, highlighting the growing need for effective central nervous system biomarkers-based diagnostics. This demographic shift is compelling healthcare systems worldwide to adopt more sophisticated diagnostic approaches, particularly in developed nations where aging populations are most concentrated.
The industry is witnessing a paradigm shift towards personalized medicine and precision diagnostics, fundamentally changing how neurological conditions are diagnosed and treated. Healthcare providers are increasingly moving away from traditional one-size-fits-all approaches in favor of more targeted, neurological biomarkers-based diagnostic strategies. This transition is particularly evident in the field of multiple sclerosis and Parkinson's disease diagnostics, where CNS biomarkers are becoming essential tools for disease stratification and treatment monitoring. Recent statistics from Argentina's Institute for Neurological Research indicate a prevalence of approximately 200 cases of Parkinson's disease per 100,000 population, demonstrating the significant market potential for biomarker-based diagnostics.
Technological integration and automation are revolutionizing biomarker testing capabilities across laboratories and healthcare facilities. Advanced laboratory information management systems (LIMS) and artificial intelligence-driven analytics are enhancing the accuracy and efficiency of biomarker detection and analysis. The industry is seeing increased adoption of automated platforms that can process multiple biomarker tests simultaneously, reducing turnaround times and improving cost-effectiveness. This technological evolution is particularly beneficial for large-scale screening programs and research initiatives.
The market is experiencing a notable shift towards non-invasive biomarker testing methods, with blood-based and imaging biomarkers gaining significant traction. According to recent Canadian healthcare statistics, with over 500,000 Canadians living with dementia, there is a growing emphasis on developing and validating novel, less invasive neurological biomarkers testing approaches. This trend is driving innovation in blood-based biomarker development and imaging technologies, making diagnostic procedures more patient-friendly while maintaining high accuracy levels. The industry is witnessing increased collaboration between diagnostic companies and research institutions to develop and validate these next-generation biomarker testing platforms.
Global Central Nervous System Biomarkers Market Trends and Insights
Rise in the Development in Proteomics, Genomics, and Imaging Systems
The advancement in proteomics, genomics, and imaging systems has significantly transformed the evolution of newer neurological biomarkers for central nervous system disorders. Proteomics technologies have made substantial progress in various areas of drug discovery and development through the comparative assessment of normal and diseased tissues. Proteomics research demonstrates great potential in discovering relevant protein neurological biomarkers for both diagnostics and therapies targeting the central nervous system. These technologies play a crucial role in earlier disease diagnosis, prognosis, and monitoring central nervous disease development, with proteomics and metabolomics approaches enabling personalized treatment options for various neurological conditions.
The development in genomics has led to improved awareness regarding genetic regulatory pathways related to CNS biomarkers. Measurable DNA and RNA characteristics serve as indicators of normal biological processes, pathogenic processes, and therapeutic responses. The advancement in imaging systems has introduced sophisticated techniques like MRI perfusion, which allows for the quantitative assessment of tumor microvasculatures. This technique utilizes the DSC-MRI method to measure relative cerebral blood volume as a non-invasive imaging biomarker of tissue microvascular density, revolutionizing the way neurological conditions are diagnosed and monitored.
Rising Investments from Governments and Private Players
Government and private sector investments have become instrumental in accelerating research pertaining to central nervous system biomarkers. For instance, Brown University established a new Center for Alzheimer's Disease Research with support from two generous gifts totaling USD 30 million, aiming to build a world-class research program focused on early detection and treatment of Alzheimer's disease and related dementias using CNS biomarkers. Similarly, the National Institutes of Health awarded USD 7.2 million to a Cleveland Clinic-led research team to improve the accuracy of multiple sclerosis diagnoses, supporting a multi-center study to evaluate whether a new biomarker known as the central vein sign can serve as a reliable diagnostic marker.
The investment landscape has witnessed significant participation from venture capital firms and pharmaceutical companies. Hikma Pharmaceuticals PLC's venture capital arm, Hikma Ventures, invested in Altoida Inc., following the company's USD 6.3 million Series-A round, to help predict brain diseases with up to 94% accuracy. Additionally, BrainStorm Cell Therapeutics received a USD 495,330 grant from the National Multiple Sclerosis Society to support biomarker studies through its clinical trial testing cell therapy, NurOwn, in patients with progressive multiple sclerosis. These investments have been crucial in driving innovation and research in CNS biomarkers development.
Increasing Research and Development with Successful Clinical Trials of Biomarkers
The field of CNS biomarkers has witnessed remarkable advancement through successful clinical trials and research initiatives. Denovo Biopharma LLC achieved a significant milestone when the United States Food and Drug Administration authorized its investigational new drug application of DB104 (liafensine) for treatment-resistant depression, enabling the start of a global Phase 2B clinical trial. This represents the first-ever genetic biomarker-guided clinical trial conducted for central nervous system diseases, marking a revolutionary approach in CNS disorder treatment.
LEXEO Therapeutics has demonstrated promising results in their ongoing Phase 1/2 clinical trial of LX1001, an AAV-based investigational gene therapy designed to deliver the protective apolipoprotein E2 gene to the CNS of APOE4 homozygous Alzheimer's disease patients. Additionally, Lysogene reported positive biomarker data from their ongoing AAVance Phase 2/3 clinical trial with LYS-SAF302 for the treatment of MPS IIIA, demonstrating the biological activity of their investigational therapy through reductions relative to baseline levels. These successful clinical trials have not only validated the effectiveness of various biomarkers but have also opened new avenues for therapeutic interventions in CNS disorders, contributing to the growth of the central nervous system biomarkers market.
Segment Analysis: By Type of CNS Biomarker
Safety Biomarker Segment in Central Nervous System Biomarkers Market
The Safety Biomarker segment maintains its dominant position in the central nervous system biomarkers market, accounting for approximately 39% of the total market share in 2024. Safety biomarkers play a crucial role in indicating the likelihood, presence, or extent of toxicity as an adverse effect before or after exposure to medical products or environmental agents. These biomarkers have gained widespread acceptance by regulatory agencies and are increasingly implemented in early clinical trials, playing a key role in decision-making processes and facilitating the progression of promising therapeutics from preclinical through clinical development. The segment's leadership is further strengthened by extensive research initiatives and collaborations between major pharmaceutical companies and research institutions focusing on developing and validating novel safety biomarkers for CNS disorders.
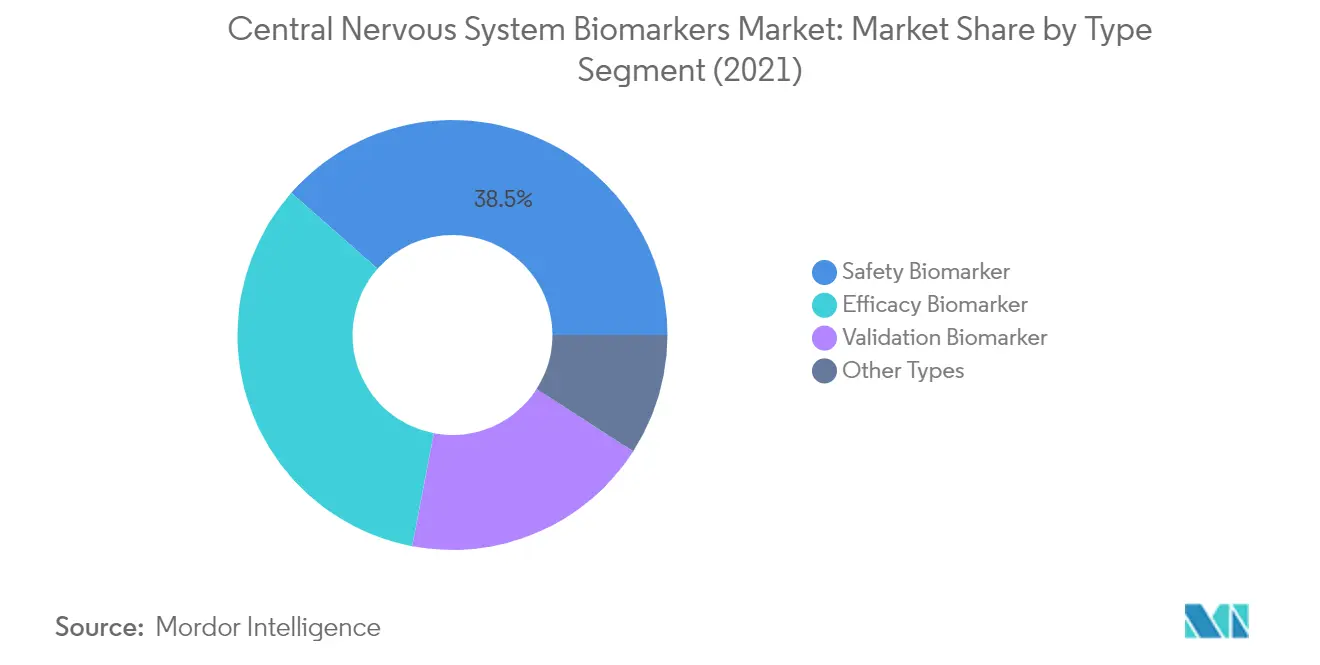
Efficacy Biomarker Segment in Central Nervous System Biomarkers Market
The Efficacy Biomarker segment is projected to experience the highest growth rate of approximately 10% during the forecast period 2024-2029. This accelerated growth is driven by the increasing demand for biomarkers that can effectively predict the clinical efficacy of drugs given to patients with neurological disorders. The segment's expansion is supported by advancing proteomics and genomics technologies, enabling more precise measurement of drug responses and treatment outcomes. Additionally, the rising focus on personalized medicine and the need for more accurate therapeutic monitoring in neurological conditions has created a strong foundation for the development and adoption of efficacy biomarkers in clinical settings.
Remaining Segments in CNS Biomarker Types
The Validation Biomarker and Other Types of CNS biomarkers segments continue to play vital roles in the market's ecosystem. Validation biomarkers are particularly important in establishing scientific frameworks and providing evidence for the physiologic, toxicologic, and pharmacologic significance of test results. These biomarkers are essential for confirming the accuracy and consistency of diagnostic and therapeutic approaches in CNS disorders. The Other Types segment encompasses emerging biomarker categories and novel approaches in development, including predictive and prognostic biomarkers, which are gaining importance in early disease detection and treatment response monitoring.
Segment Analysis: By Application
Drug Discovery and Development Segment in Central Nervous System Biomarkers Market
The Drug Discovery and Development segment dominates the central nervous system biomarkers market, holding approximately 49% of the market share in 2024. This significant market position is driven by the increasing use of biomarkers in clinical trials and drug development processes for neurological disorders. Biomarkers assist in diagnosis, demonstrate target engagement, support disease modification, and monitor safety parameters during drug development. The segment's growth is further supported by the rising investments in research and development activities by pharmaceutical companies focusing on CNS disorders. Additionally, the increasing adoption of biomarker-guided drug development approaches has shifted attrition to earlier stages of the development process, making drug discovery more efficient and cost-effective.
Personalized Medicine Segment in Central Nervous System Biomarkers Market
The Personalized Medicine segment is projected to witness the fastest growth in the neurological biomarkers market during the forecast period 2024-2029. This rapid growth is attributed to the increasing adoption of precision medicine approaches in treating neurological disorders. The segment's expansion is driven by the rising application of biomarkers as diagnostic tools for recognizing precise biological markers and developing breakthrough products for neurological diseases. Furthermore, the growing emphasis on tailored therapeutic approaches and the increasing success of biomarker-based patient stratification in clinical trials are contributing to the segment's accelerated growth. The advancement in biomarker research for neurodegenerative diseases has also opened new avenues for personalized treatment approaches, particularly in conditions like Alzheimer's and Parkinson's disease.
Remaining Segments in CNS Biomarkers Market by Application
The Other Applications segment, which includes disease risk assessment and diagnostics, plays a crucial role in the CNS biomarkers market. This segment focuses on using biomarkers for early detection of brain changes and understanding risk factors involved in various neurological conditions. The segment encompasses applications in research studies, clinical practice, and disease diagnostics, particularly in neuroinflammatory diseases where biomarkers facilitate timely diagnosis and proper disease management. The growing importance of diagnostic biomarkers in detecting early signs of neurological disorders and monitoring disease progression continues to drive innovation and development in this segment.
Segment Analysis: By End User
Research Centers Segment in Central Nervous System Biomarkers Market
Research centers dominate the central nervous system biomarkers market, accounting for approximately 44% of the total market share in 2024. This significant market position is driven by extensive research activities and substantial funding for biomarker development in neurological disorders. Major research institutions worldwide are actively engaged in studying novel biomarkers for early detection and monitoring of conditions like Alzheimer's disease, Parkinson's disease, and multiple sclerosis. The segment is also experiencing the fastest growth rate of around 9% from 2024-2029, fueled by increasing investments in R&D infrastructure and the rising focus on precision medicine approaches in neurology. Research centers are particularly crucial in validating new biomarkers through clinical trials and establishing standardized protocols for their implementation in diagnostic and therapeutic applications. The expansion of collaborative research networks and the integration of advanced technologies like artificial intelligence in biomarker discovery are further strengthening the segment's market position.
Diagnostic Laboratories Segment in Central Nervous System Biomarkers Market
Diagnostic laboratories represent a vital segment in the central nervous system biomarkers market, playing a crucial role in the clinical implementation of validated biomarker tests. These facilities are equipped with sophisticated analytical instruments and automated systems that enable high-throughput screening and precise measurement of various CNS biomarkers. The increasing adoption of standardized testing protocols and quality control measures has enhanced the reliability of biomarker testing in diagnostic laboratories. Many laboratories are expanding their test menus to include novel CNS biomarkers, particularly those related to neurodegenerative disorders and neurological conditions. The segment benefits from growing collaborations with pharmaceutical companies for companion diagnostic development and the rising demand for molecular diagnostic services. Additionally, the integration of digital pathology solutions and laboratory information management systems is improving the efficiency and accuracy of biomarker testing processes.
Remaining Segments in End User Market
The clinics/hospitals segment plays an essential role in the central nervous system biomarkers market by serving as the primary point of patient care and biomarker test utilization. These healthcare facilities are increasingly incorporating biomarker testing into their diagnostic workflows, particularly for neurological disorders that require regular monitoring and treatment adjustment. Hospitals are also participating in clinical validation studies for new biomarkers and contributing to the development of clinical guidelines for their use. The segment benefits from direct access to patient populations and the ability to correlate biomarker results with clinical outcomes. Many hospitals are establishing specialized neurology centers equipped with advanced diagnostic capabilities, including biomarker testing facilities, to provide comprehensive care for patients with neurological disorders.
Global Central Nervous System Biomarkers Market Geography Segment Analysis
Central Nervous System Biomarkers Market in North America
The North American central nervous system biomarkers market is experiencing robust growth driven by advanced healthcare infrastructure, significant research and development activities, and a strong presence of key market players. The United States, Canada, and Mexico are the key countries in this region, each contributing uniquely to the market's development. The region benefits from extensive clinical trials, an increasing prevalence of neurological disorders, and substantial healthcare expenditure across these nations. The presence of sophisticated diagnostic laboratories and research centers further strengthens the market position in North America.
Central Nervous System Biomarkers Market in United States
The United States dominates the North American central nervous system biomarkers market, holding approximately 86% of the regional market share in 2024. The country's market leadership is attributed to its advanced healthcare system, substantial research funding, and the presence of major pharmaceutical and biotechnology companies. The growing burden of central nervous system diseases, coupled with an aging population and increasing demand for efficient diagnostic products, drives market growth. The country's robust healthcare infrastructure, including numerous diagnostic laboratories and research centers, further supports market expansion. Additionally, the strong presence of key market players and their continuous focus on research and development activities contributes significantly to market development.
Growth Dynamics in United States Central Nervous System Biomarkers Market
The United States also leads in terms of growth rate, with an expected CAGR of approximately 9% during 2024-2029. This growth is fueled by increasing investments in biomarker research, rising adoption of personalized medicine, and technological advancements in proteomics and genomics. The country's focus on developing novel biomarkers through extensive research programs and clinical trials supports this growth trajectory. The expansion of diagnostic capabilities and increasing integration of CNS biomarkers-based approaches in clinical practice further accelerates market growth. The strong emphasis on early disease detection and monitoring through biomarkers continues to drive innovation and market expansion in the United States.
Competitive Landscape
Top Companies in Central Nervous System Biomarkers Market
The competitive landscape is characterized by major players like Bio-Rad Laboratories, Thermo Fisher Scientific, Merck KGaA, Eli Lilly, and BioMérieux leading the central nervous system biomarkers market through continuous innovation and strategic initiatives. Companies are heavily investing in research and development to expand their biomarker portfolios, particularly focusing on neurodegenerative diseases like Alzheimer's and Parkinson's. The industry witnesses frequent collaborations with academic institutions and research organizations to enhance technological capabilities and develop novel diagnostic solutions. Market leaders are strengthening their global presence through robust distribution networks and strategic partnerships, while simultaneously focusing on automation-compatible reagent systems and associated products. Companies are also emphasizing the development of predictive neurological biomarkers and expanding their manufacturing capabilities to meet growing market demands.
Market Dominated by Diversified Global Players
The central nervous system biomarkers market exhibits a moderately consolidated structure dominated by large multinational corporations with diverse product portfolios spanning multiple therapeutic areas. These established players leverage their extensive research capabilities, global distribution networks, and strong financial positions to maintain market leadership. The market is characterized by a mix of pure-play biomarker companies and large pharmaceutical conglomerates, with the latter having significant advantages in terms of research funding and market access. The competitive dynamics are further shaped by regional players who focus on specific geographic markets or specialized biomarker applications.
The industry has witnessed significant merger and acquisition activity, with larger companies acquiring smaller, innovative firms to expand their technological capabilities and product offerings. Companies are increasingly focusing on vertical integration strategies, combining diagnostic capabilities with therapeutic development to create comprehensive solutions. The market also sees frequent strategic alliances between diagnostic companies and pharmaceutical firms, particularly in the development of companion diagnostics and personalized medicine applications. Regional players are gaining prominence through niche specialization and local market expertise, though global players continue to dominate the overall market landscape.
Innovation and Collaboration Drive Future Success
Success in the CNS biomarkers market increasingly depends on companies' ability to develop innovative, cost-effective diagnostic solutions while maintaining high accuracy and reliability. Market players must focus on expanding their intellectual property portfolios and developing proprietary technologies to differentiate themselves in a competitive landscape. The ability to provide comprehensive solutions, including both diagnostic tools and therapeutic applications, is becoming increasingly important. Companies need to invest in digital capabilities and automation to improve testing efficiency and reduce costs, while maintaining strong relationships with healthcare providers and research institutions.
Future market success will require companies to navigate complex regulatory environments while maintaining flexibility in product development and commercialization strategies. Players must focus on developing biomarkers that address unmet clinical needs while ensuring compatibility with existing diagnostic platforms. The ability to provide value-added services, such as data analytics and interpretation support, will become increasingly important for maintaining competitive advantage. Companies must also consider the growing importance of personalized medicine and adapt their strategies to address the increasing demand for targeted diagnostic solutions. Building strong partnerships with healthcare providers and maintaining close relationships with key opinion leaders will be crucial for long-term success in this evolving market.
Global Central Nervous System Biomarkers Industry Leaders
G-Biosciences
Merck KGaA
Thermo Fisher Scientific Inc.
Bio-Rad Laboratories, Inc.
Biomeriux (Banyan Biomarkers Inc.)
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- In November 2021, Eisai Co. Ltd and Biogen Inc. announced a presentation about exploring the use of plasma-based biomarkers in the Phase 3 AHEAD 3-45 study of lecanemab (BAN2401), an investigational anti-amyloid beta (Aβ) protofibril antibody for Alzheimer's disease.
- In October 2021, Diadem announced the publication of clinical data showing its AlzoSure biomarker test is able to predict the progression of Alzheimer's disease years before symptoms are displayed.
Global Central Nervous System Biomarkers Market Report Scope
As per the scope of the report, central nervous system (CNS) biomarkers are used clinically to screen for, diagnose, or monitor the activity of CNS-related diseases to guide molecularly targeted therapy or assess therapeutic response. Biomarkers help understand the development of chronic diseases and their relationship with environmental chemicals and identify subjects who are at high risk of developing diseases. The central nervous system biomarkers market is segmented by type of CNS biomarkers (safety biomarker, efficacy biomarker, validation biomarker, and other types of CNS biomarkers), application (drug discovery and development, personalized medicine, and other applications), end user (diagnostic labs, clinics/hospitals, and research centers), and geography (North America, Europe, Asia-Pacific, Middle-East and Africa, and South America). The report offers the value (in USD million) for the above segments.
| Safety Biomarkers |
| Efficacy Biomarkers |
| Validation Biomarkers |
| Other Types of CNS Biomarkers |
| Drug Discovery and Development |
| Personalized Medicine |
| Other Applications |
| Diagnostic Labs |
| Clinics/Hospitals |
| Research Centers |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle-East and Africa | GCC |
| South Africa | |
| Rest of Middle-East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Type of CNS Biomarkers | Safety Biomarkers | |
| Efficacy Biomarkers | ||
| Validation Biomarkers | ||
| Other Types of CNS Biomarkers | ||
| By Application | Drug Discovery and Development | |
| Personalized Medicine | ||
| Other Applications | ||
| By End User | Diagnostic Labs | |
| Clinics/Hospitals | ||
| Research Centers | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle-East and Africa | GCC | |
| South Africa | ||
| Rest of Middle-East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
How big is the Global Central Nervous System Biomarkers Market?
The Global Central Nervous System Biomarkers Market size is expected to reach USD 6.04 billion in 2025 and grow at a CAGR of 7.67% to reach USD 8.74 billion by 2030.
What is the current Global Central Nervous System Biomarkers Market size?
In 2025, the Global Central Nervous System Biomarkers Market size is expected to reach USD 6.04 billion.
Who are the key players in Global Central Nervous System Biomarkers Market?
G-Biosciences, Merck KGaA, Thermo Fisher Scientific Inc., Bio-Rad Laboratories, Inc. and Biomeriux (Banyan Biomarkers Inc.) are the major companies operating in the Global Central Nervous System Biomarkers Market.
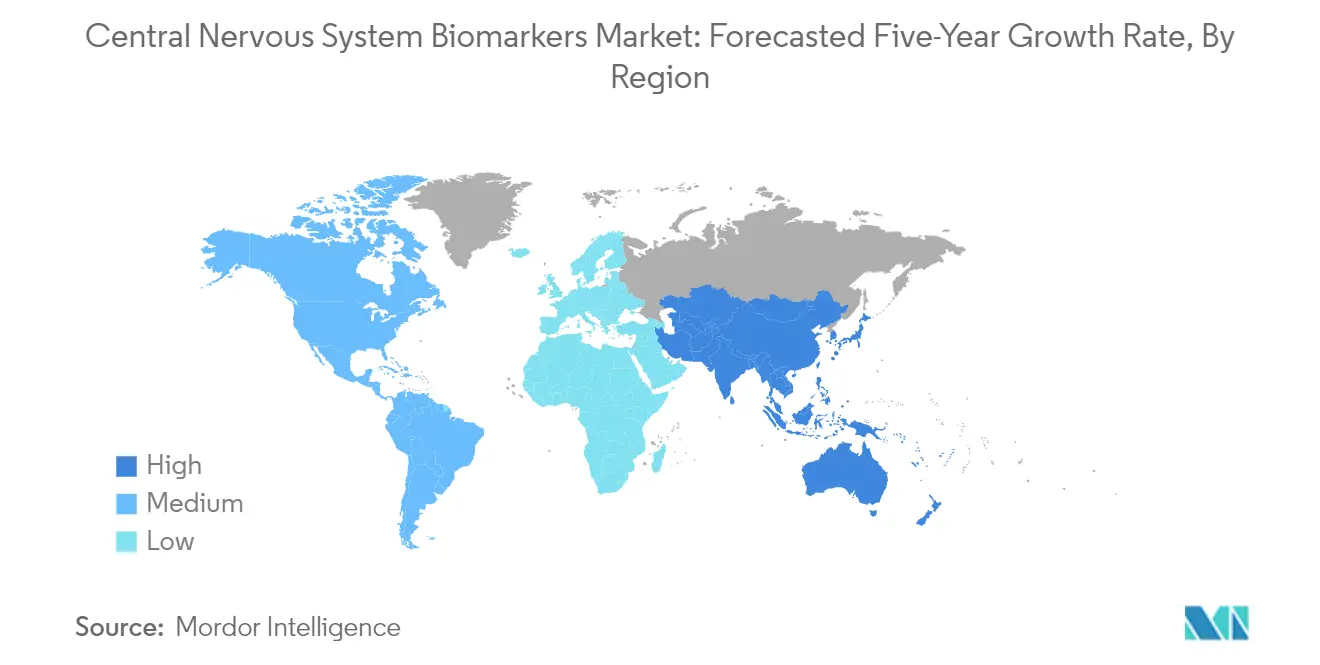
Which is the fastest growing region in Global Central Nervous System Biomarkers Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Global Central Nervous System Biomarkers Market?
In 2025, the North America accounts for the largest market share in Global Central Nervous System Biomarkers Market.
What years does this Global Central Nervous System Biomarkers Market cover, and what was the market size in 2024?
In 2024, the Global Central Nervous System Biomarkers Market size was estimated at USD 5.58 billion. The report covers the Global Central Nervous System Biomarkers Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Global Central Nervous System Biomarkers Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:








