Cell-free Protein Expression Market Size and Share
Cell-free Protein Expression Market Analysis by Mordor Intelligence
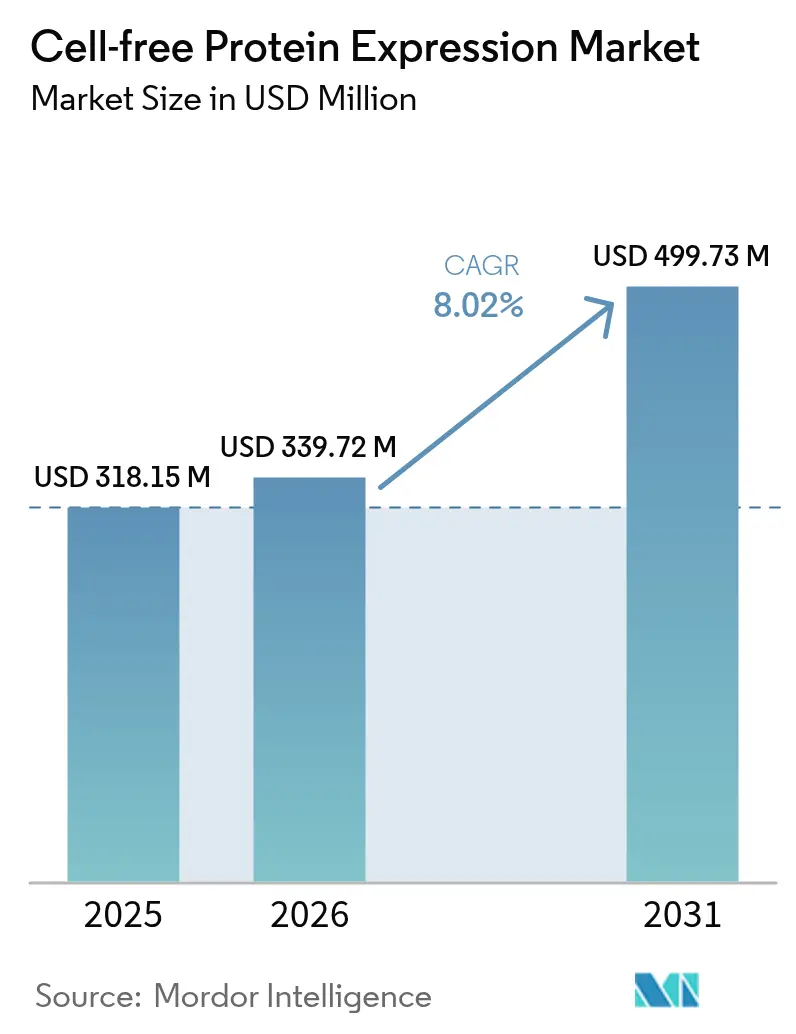
The Cell-free Protein Expression Market size is expected to increase from USD 318.15 million in 2025 to USD 339.72 million in 2026 and reach USD 499.73 million by 2031, growing at a CAGR of 8.02% over 2026-2031.
Funding for distributed biomanufacturing, especially in the United States and Japan, is reshaping procurement priorities toward portable kits that shorten design–build–test loops for synthetic-biology projects. Government preparedness mandates after pandemic-era supply shocks, together with new FDA guidance clarifying existing biologics rules for cell-free therapeutics, have reduced regulatory risk and attracted pharmaceutical investment.[1]U.S. Food and Drug Administration, “Chemistry, Manufacturing, and Controls Information for Biological Products,” fda.gov Continuous-exchange cell-free (CECF) systems are now scaling beyond multi-gram runs, allowing niche biologics to be produced economically without fermentation start-up delays.[2]Zachary A. Sun, “Continuous-Exchange Cell-Free Systems,” Nature Chemical Biology, nature.com At the same time, freeze-dried reaction kits remain the fastest-growing product type because they remove cold-chain dependence and enable point-of-care protein printing in remote settings.
Key Report Takeaways
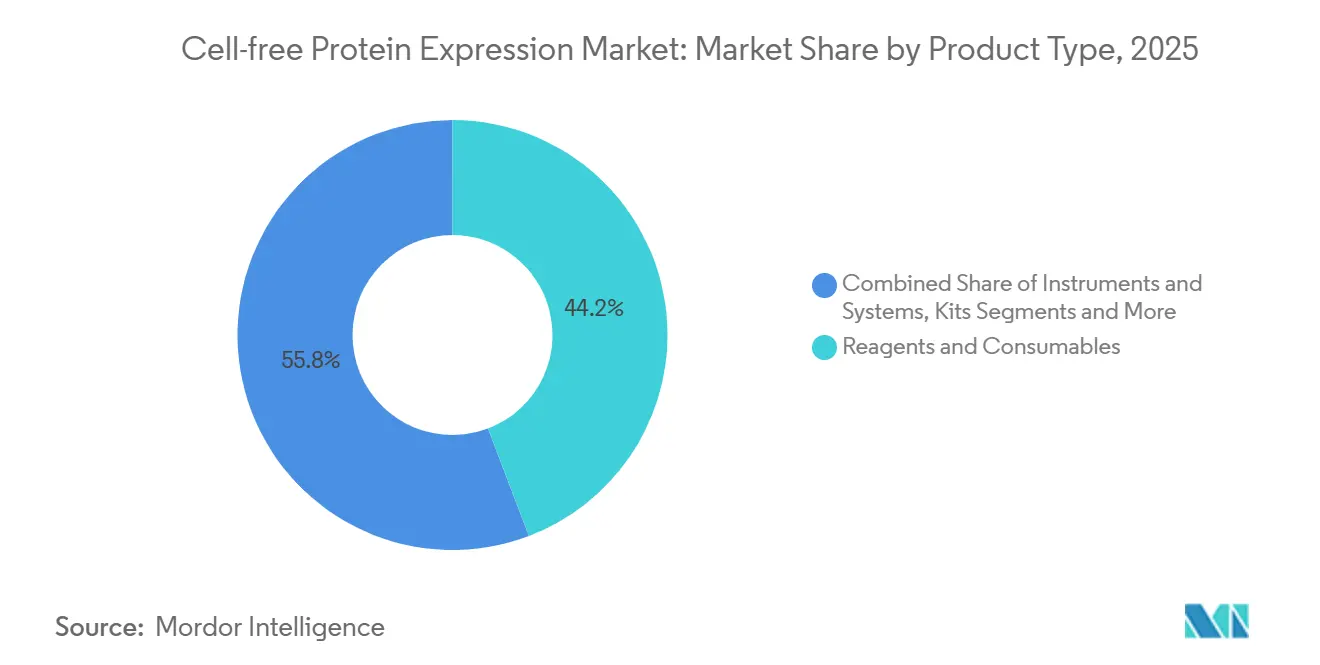
- Reagents and consumables led with 44.22% of cell-free protein expression market share in 2025, while kits are forecast to expand at an 11.52% CAGR through 2031.
- E. coli lysates commanded 51.75% share of the cell-free protein expression market size in 2025, yet insect-cell extracts are projected to post a 10.74% CAGR to 2031.
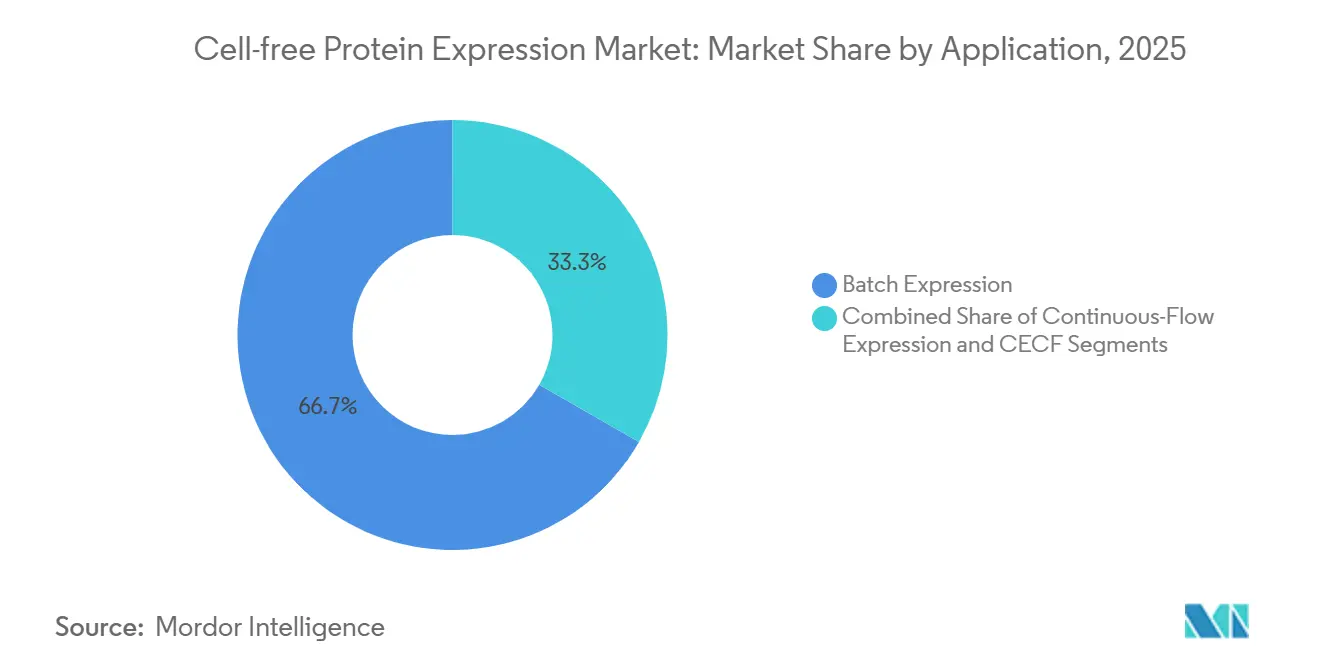
- Batch expression accounted for 66.72% revenue in 2025; CECF is the fastest-growing mode at 10.89% CAGR through 2031.
- Liquid formats held 78.21% of 2025 volume, but lyophilized variants are set to grow 11.48% CAGR, widening access to portable biomanufacturing.
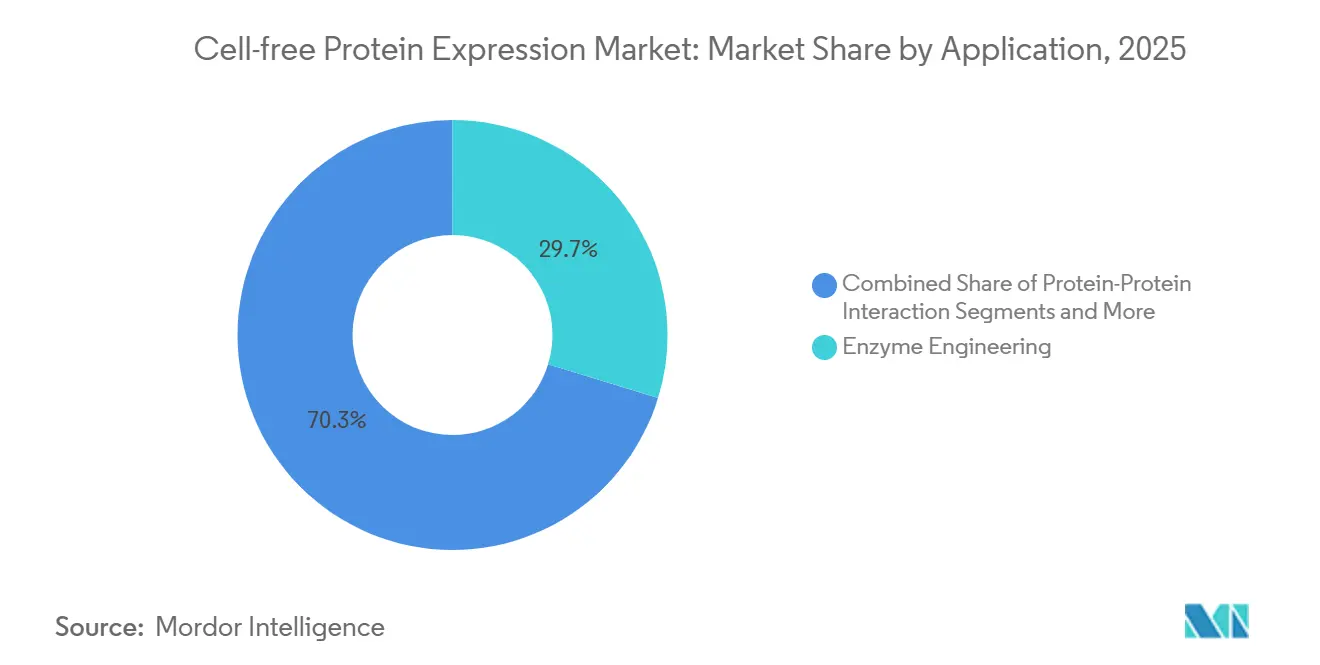
- High-throughput production captured 29.73% share in 2025 and is projected to register an 11.39% CAGR between 2026 and 2031.
- Contract research organizations recorded the highest projected CAGR among end users at 10.24% over 2026-2031.
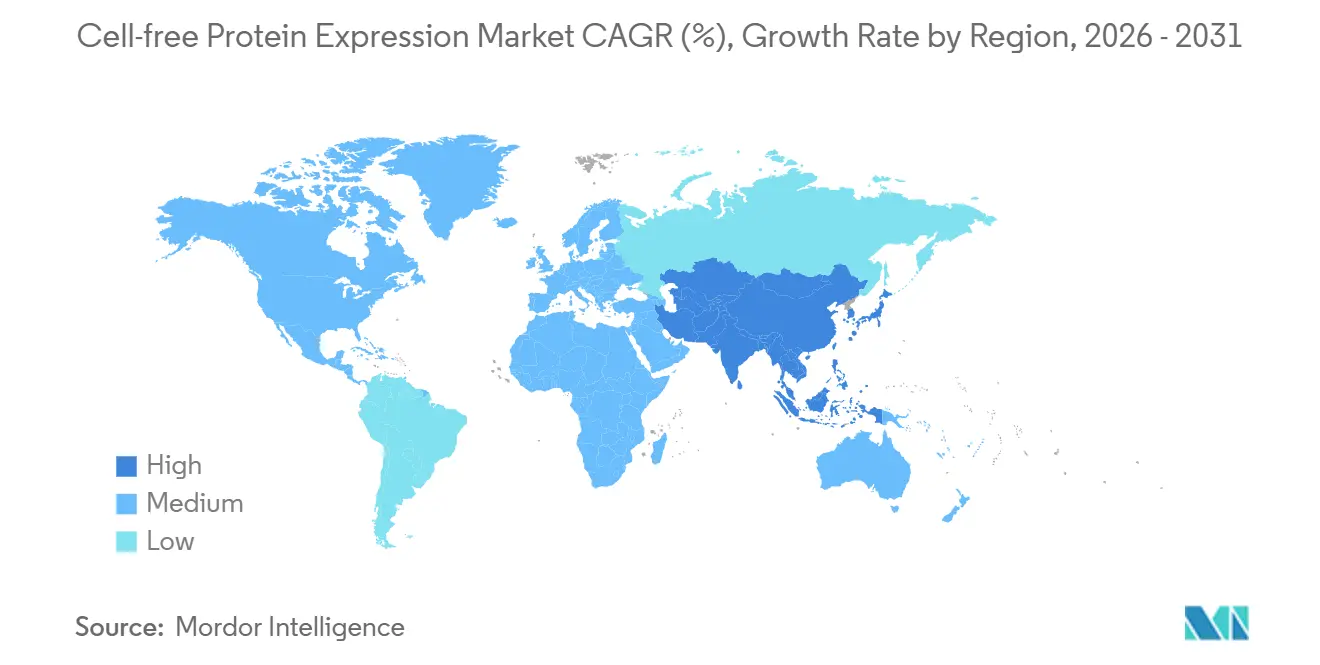
- North America led with 39.54% revenue in 2025, whereas Asia-Pacific is forecast to expand at 10.28% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Cell-free Protein Expression Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rapid Adoption for High-Throughput Protein Engineering & Synthetic Biology | +1.8% | Global, led by North America & Europe | Short term (≤ 2 years) |
| Rising Demand for Biologics & Personalized Therapeutics | +1.5% | North America, Europe, Asia-Pacific | Medium term (2-4 years) |
| Advantages Over Cell-Based Systems (Speed, Toxic Protein Handling) | +1.3% | Global | Short term (≤ 2 years) |
| Government On-Demand Biomanufacturing Preparedness Initiatives | +1.1% | North America, Europe | Medium term (2-4 years) |
| Freeze-Dried Kits Enabling Decentralized Protein Printing | +1.0% | Global, early adoption in North America & Asia-Pacific | Long term (≥ 4 years) |
| AI/ML-Driven Yield Optimization & Automation Integration | +0.9% | North America, Europe, China, Japan | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Rapid Adoption for High-Throughput Protein Engineering & Synthetic Biology
Cell-free workflows collapse cloning, colony picking, and scale-up into a same-day process, letting researchers screen hundreds of variants in 96- or 384-well plates while living-cell systems would require weeks.[3]Christopher A. Voigt, “Design-Build-Test Cycles in Synthetic Biology,” Proceedings of the National Academy of Sciences, pnas.org DARPA’s USD 87 million BioMaDE awards in 2024 funded modular lysate platforms that prototype enzymes and biosensors for defense applications. Commercial kits bundling lysate and energy mixes standardize performance across labs and lower entry barriers. Seamless integration with Golden Gate and Gibson assembly now transforms a digital DNA design into functional protein inside 24 hours, fundamentally shortening discovery cycles in academia and start-ups.
Rising Demand for Biologics & Personalized Therapeutics
More than 3,800 biologics were in clinical development in 2025, creating urgency for flexible platforms that crank out patient-specific proteins within days. Cell-free systems meet turn-around targets for personalized cancer vaccines, something CHO cells cannot achieve at clinical scale. Sutro Biopharma’s XpressCF, built on E. coli lysates, advanced to Phase II trials in 2025, proving regulators will review cell-free-produced therapeutics when process controls comply with cGMP. FDA guidance in 2024 cemented that host-free manufacturing falls under existing biologic rules so long as analytical characterization is robust. Hospitals in low-resource regions now explore freeze-dried kits to print therapeutic enzymes on site, sidestepping cold-chain dependencies.
Advantages Over Cell-Based Systems (Speed, Toxic Protein Handling)
Membrane and toxic proteins that cripple living hosts express efficiently in open lysates, unlocking drug-target classes previously off-limits. Wheat-germ and insect-cell extracts replicate native lipid environments, enabling structural biologists to elucidate GPCR and ion-channel structures. Non-canonical amino acids can be inserted directly because no cell wall filters substrates, supporting site-specific conjugation strategies used in antibody–drug conjugates. Thermo Fisher’s PURExpress kit, which uses purified translation components, boosted yields 40% in 2025 by removing endogenous nucleases. Batch reactions deliver usable protein within four hours, and continuous-flow reactors sustain synthesis days longer, shrinking early discovery timelines.
Government On-Demand Biomanufacturing Preparedness Initiatives
The U.S. DoD awarded USD 23 million in 2024 to develop backpack-size reactors for field vaccine and antitoxin synthesis. NIST followed with standards to quantify lysate potency, a prerequisite for regulatory acceptance. Japan earmarked JPY 3.2 billion in 2025 to stand up regional hubs able to formulate influenza vaccines inside 72 hours. These projects create a captive market segment that values portability and shelf life more than cost per milligram, favoring freeze-dried kits over stainless-steel bioreactors.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High Reagent Costs & Scalability Challenges | −1.2% | Global | Short term (≤ 2 years) |
| Limited Post-Translational Modification Capability | −0.9% | Global, acute for mammalian therapeutics | Medium term (2-4 years) |
| Fragmented IP Landscape & Patent Thickets | −0.7% | North America, Europe, Asia-Pacific | Long term (≥ 4 years) |
| Regulatory Pathway Uncertainty for Therapeutic Proteins | −0.6% | Global, most acute in emerging markets | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
High Reagent Costs & Scalability Challenges
Energy cocktails, nucleotides, and lysate prep account for up to 70% of production cost, often topping USD 500 per milligram for mammalian extracts, dwarfing fermentation economics. Continuous-exchange reactors improve yields five-fold but still trail fed-batch titers by two orders of magnitude. Merck KGaA shaved 30% off reagent expense in 2024 with its ReadyToProcess CFPS line, yet management admits 10-liter scale remains uneconomical for mainstream antibodies. Absence of scale economies in lysate production confines adoption to high-value or time-critical proteins.
Limited Post-Translational Modification Capability
E. coli systems lack glycosylation enzymes, preventing authentic antibody Fc modifications that govern serum half-life. Insect-cell and mammalian extracts offer partial solutions but at yields 5-10x lower than prokaryotic counterparts. Hybrid approaches that bolt glycosyltransferases onto E. coli lysates can restore N-glycans yet add USD 150 per milligram extra cost. Disulfide-bond formation requires redox-balanced buffers that complicate automation, limiting throughput.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Product Type: Kits Accelerate Democratization
Kits captured growing mindshare as lyophilized formats removed the cold-chain barrier and bundled protocols eliminated troubleshooting. Kits are on track to expand at an 11.52% CAGR between 2026 and 2031, outpacing reagents despite the latter’s 44.22% revenue lead in 2025. This trajectory illustrates how pre-optimized mixes shorten learning curves for small biotechs and academic labs. Instruments that automate pipetting and purification complement kit adoption, as seen with Nuclera’s eProtein platform rollout to 12 pharma labs in 2025. Reagents will remain indispensable for experienced users customizing buffer chemistries, yet kit convenience drives the incremental user base. Accessories and service bundles, including contract lysate prep, grow in tandem because newcomers often outsource complex targets.

By Expression System: Insect-Cell Momentum Builds
The cell-free protein expression market size for insect-cell lysates is expected to climb at 10.74% CAGR, nibbling share from E. coli’s 51.75% dominance in 2025. Demand for eukaryotic glycosylation propels hybrid and insect solutions for vaccine antigens and antibody fragments. Wheat-germ extracts sit in the middle ground, favored for membrane proteins that aggregate in bacteria. Start-ups such as LenioBio combine E. coli yield advantages with exogenous enzymes to approximate mammalian quality at lower cost. Rabbit reticulocyte and mammalian systems persist in niche roles where post-translational fidelity trumps cost.
By Expression Mode: CECF Unlocks Multi-Gram Runs
Batch systems still delivered 66.72% of 2025 revenue because they need only a heat block and microtube, making them the default for quick screens. Yet CECF platforms are forecast to post 10.89% CAGR as pharmaceutical users chase yields above 5 mg/mL, bridging the pre-clinical manufacturing gap. Sutro Biopharma’s CECF-based XpressCF produced antibody–drug conjugates now in Phase II trials, underscoring regulatory viability. Continuous-flow formats serve mid-range needs where extended runs are beneficial but dialysis hardware is acceptable.

By Reaction Format: Lyophilized Leads Portability Wave
Liquid lysates owned 78.21% share in 2025 because freezer capacity is ubiquitous in core labs. However, lyophilized variants are set to expand 11.48% CAGR, supplying defense agencies and rural clinics with shelf-stable “protein printers”. Microfluidic chips integrate synthesis and purification in credit-card footprints, targeting diagnostic OEMs that value automation more than cost per microgram. The cell-free protein expression market share of lyophilized kits will therefore widen as ambient shipping becomes standard for global distribution.
By Application: High-Throughput Workflows Surge
Enzyme engineering retained 29.73% of 2025 revenue, yet high-throughput production is projected to climb at 11.39% CAGR as robotics and AI converge for 96-protein overnight runs. Protein-protein interaction mapping and protein labeling also benefit from open lysate architecture that tolerates co-expression and non-canonical amino acids. Therapeutic protein output, though smaller today, gains legitimacy from clinical-stage programs like Sutro Biopharma, suggesting future expansion once regulatory precedents mature.

By End User: CRO Growth Outpaces In-House Efforts
Pharmaceutical and biotech firms generated 41.27% of 2025 turnover, yet CRO revenue is predicted to grow 10.24% CAGR as outsourcing becomes cheaper than maintaining fermenters for early screens. Academic labs adopt commercial kits thanks to NIH grants covering capital purchases. Diagnostic developers leverage cell-free speed to refresh antigen panels for multiplex tests without large-scale cultures. Other users, including ag-biotech firms, remain exploratory but recognize value where transgenic hosts are impractical.
Geography Analysis
North America commanded 39.54% of 2025 revenue, reflecting NIH and DoD spending on rapid-response platforms. The region also hosts leading kit suppliers and most FDA-regulated clinical programs. Europe follows, buoyed by Horizon Europe’s EUR 200 million grants that subsidize personalized-medicine pilots, particularly in Germany and the United Kingdom.
Asia-Pacific posts the fastest regional growth at 10.28% CAGR, underwritten by China’s RMB 1.2 billion stimulus for domestic lysate production and automation tools. Japan’s Osaka and Fukuoka hubs target 72-hour vaccine turnaround to bolster pandemic preparedness. India funds centers of excellence building low-cost kits for crop-protection enzymes and point-of-care diagnostics.
South America and the Middle East & Africa remain small today, though Brazil explores veterinary vaccines and South Africa pilots rural diagnostic antigen production. Adoption hinges on lowering kit prices and clarifying local biologics regulations.
Competitive Landscape
The market is moderately concentrated. Thermo Fisher Scientific differentiates with PURExpress kits that eliminate background nucleases, while Merck KGaA invests in large-scale CECF reactors for CDMO services. Promega extends shelf life via freeze-drying innovations, capturing military and field-diagnostics contracts. Takara Bio and New England Biolabs emphasize modularity, embedding AI into protocol designers and expanding non-canonical amino-acid compatibility.
Disruptors include Nuclera, which bundles hardware, consumables, and software in subscription form, shifting revenue to recurring reagents. LenioBio’s EcoProX hybrid lysate adds eukaryotic folding enzymes to E. coli background, cutting costs relative to mammalian extracts. Synthelis focuses on membrane proteins with insect-cell lysates, serving vaccine and structural-biology niches.
Patent thickets on energy-regeneration chemistries constrain new entrants, but first-generation patents started to lapse, enabling biosimilar lysates. Vendors that pair AI optimization with robotic execution stand to gain, as pharmaceutical clients favor turnkey platforms over commodity reagents.
Cell-free Protein Expression Industry Leaders
Thermo Fisher Scientific Inc.
Promega Corporation
Merck KGaA (Sigma-Aldrich)
Takara Bio Inc.
New England Biolabs
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2026: New England Biolabs released the Monarch Mag Cell-Free DNA Extraction Kit, enabling sequential isolation of low-abundance cfDNA for biomarker discovery.
- February 2026: Thermo Fisher Scientific launched the Sequential Protein DNA RNA Extraction Kit, allowing researchers to harvest multi-omic material from scarce samples.
- January 2026: Nuclera closed a USD 12 million Series C extension, bringing total funding to USD 87 million to accelerate deployment of its eProtein platform.
Global Cell-free Protein Expression Market Report Scope
As per the scope of this report, cell-free protein expression or in vitro protein synthesis is a process that involves the production of the recombinant proteins in cell lysate solution, without using cell culture or living cells. Cell-free protein expression can be carried out using different cell lysates, such as those of E. coli, rabbit reticulocytes, wheat germs, human cells, and insect cells.
The Cell‑Free Protein Expression Market Report is segmented by Product Type, Expression System, Expression Mode, Reaction Format, Application, End User, and Geography. By Product Type, the market is segmented into Kits, Reagents & Consumables, Instruments and Systems, and Accessories & Services. By Expression System, the market is segmented into E. coli, Wheat Germ, Rabbit Reticulocyte, Insect Cell, Mammalian, and Hybrid/Others. By Expression Mode, the market is segmented into Batch, Continuous‑Flow, and CECF modes. By Reaction Format, the market is segmented into Liquid, Lyophilized, and Microfluidic formats. By Application, the market is segmented into Enzyme Engineering, Protein‑Protein Interaction studies, Protein Labelling, High‑Throughput Production, Therapeutic Protein Production, and Others. By End User, the market is segmented into Pharmaceutical & Biotechnology Companies, Academic & Research Institutes, CROs, Diagnostic Developers, and Others. By Geography, the market is segmented into North America, Europe, Asia‑Pacific, Middle East & Africa, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. Market Forecasts are Provided in Terms of Value (USD).
| Kits |
| Reagents & Consumables |
| Instruments and Systems |
| Accessories & Services |
| E. coli |
| Wheat Germ |
| Rabbit Reticulocyte |
| Insect Cell |
| Mammalian |
| Hybrid / Others |
| Batch Expression |
| Continuous-Flow Expression |
| Continuous-Exchange Cell-Free (CECF) |
| Liquid (Fresh Lysate) |
| Lyophilized (Freeze-Dried) |
| Microfluidic / Chip-Based |
| Enzyme Engineering |
| Protein-Protein Interaction |
| Protein Labelling |
| High-Throughput Production |
| Therapeutic Protein Production |
| Others |
| Pharmaceutical & Biotechnology Companies |
| Academic & Research Institutes |
| Contract Research Organisations (CROs) |
| Diagnostic Developers |
| Others |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| France | |
| United Kingdom | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| South Korea | |
| Australia | |
| Rest of Asia-Pacific | |
| Middle East & Africa | GCC |
| South Africa | |
| Rest of Middle East & Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product Type | Kits | |
| Reagents & Consumables | ||
| Instruments and Systems | ||
| Accessories & Services | ||
| By Expression System | E. coli | |
| Wheat Germ | ||
| Rabbit Reticulocyte | ||
| Insect Cell | ||
| Mammalian | ||
| Hybrid / Others | ||
| By Expression Mode | Batch Expression | |
| Continuous-Flow Expression | ||
| Continuous-Exchange Cell-Free (CECF) | ||
| By Reaction Format | Liquid (Fresh Lysate) | |
| Lyophilized (Freeze-Dried) | ||
| Microfluidic / Chip-Based | ||
| By Application | Enzyme Engineering | |
| Protein-Protein Interaction | ||
| Protein Labelling | ||
| High-Throughput Production | ||
| Therapeutic Protein Production | ||
| Others | ||
| By End User | Pharmaceutical & Biotechnology Companies | |
| Academic & Research Institutes | ||
| Contract Research Organisations (CROs) | ||
| Diagnostic Developers | ||
| Others | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| France | ||
| United Kingdom | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| South Korea | ||
| Australia | ||
| Rest of Asia-Pacific | ||
| Middle East & Africa | GCC | |
| South Africa | ||
| Rest of Middle East & Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
How large will the cell-free protein expression market be by 2031?
It is forecast to reach USD 499.73 million, rising from USD 339.72 million in 2026 at an 8.02% CAGR.
Which product type is growing fastest?
Lyophilized and other kit-based offerings, projected at 11.52% CAGR through 2031.
Why are insect-cell lysates gaining popularity?
They supply eukaryotic post-translational modifications absent in E. coli systems, meeting vaccine and antibody requirements.
What limits wider adoption today?
High reagent costs and incomplete post-translational modification capability slow large-scale manufacturing uptake.
Which region will see the quickest growth?
Asia-Pacific is forecast to expand at 10.28% CAGR thanks to sizable public funding in China, Japan, and India.
Page last updated on:









