Tumor Ablation Market Size
Tumor Ablation Market Analysis
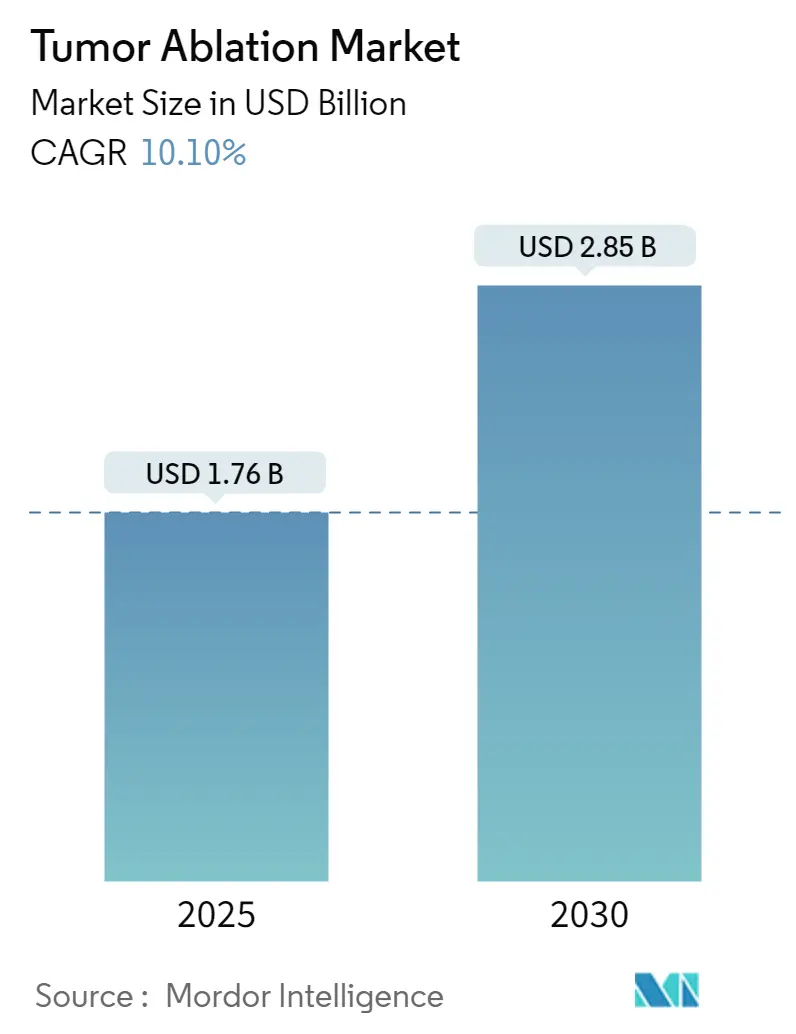
The Tumor Ablation Market size is estimated at USD 1.76 billion in 2025, and is expected to reach USD 2.85 billion by 2030, at a CAGR of 10.1% during the forecast period (2025-2030).
The increasing burden of cancer, technological advancements in ablation devices, and growing demand for minimally invasive surgeries are the major drivers for the Tumor Ablation Market.
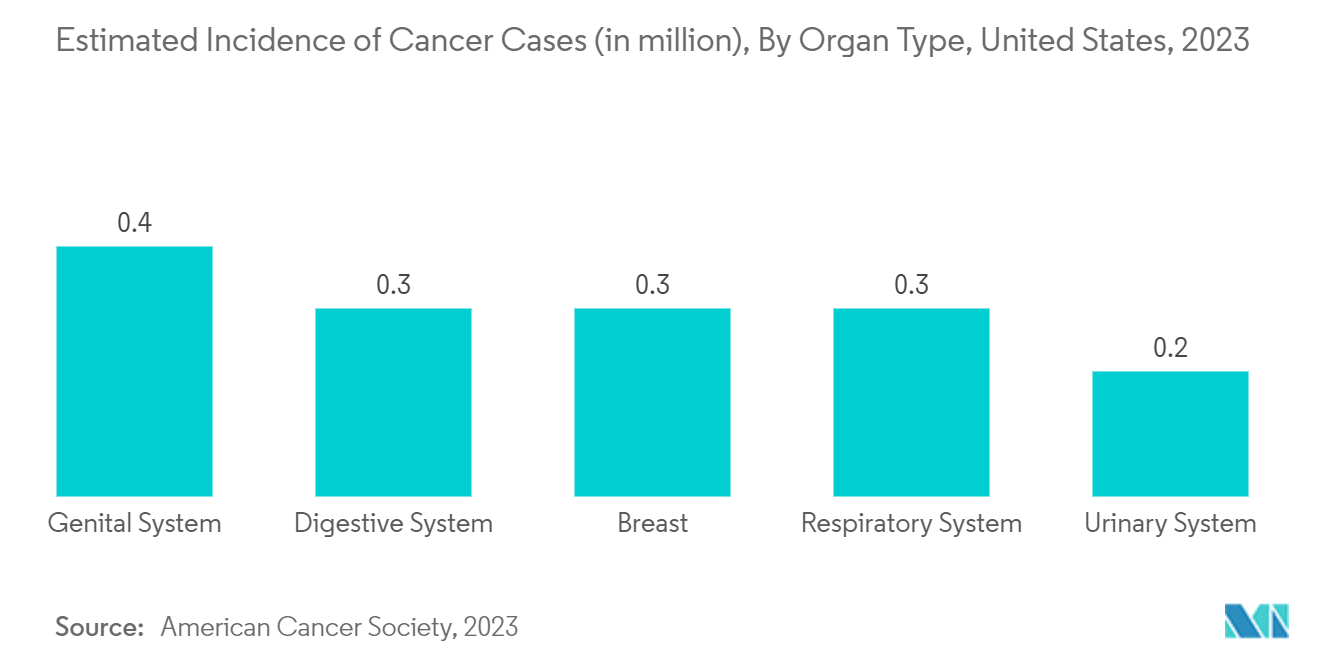
Various cancer therapies such as Tumor Ablation are used as treatment options for the management of several types of cancers, including metastatic disease. As cancer cases increase, the demand for advanced Cancer Ablation therapy rises, which is expected to drive market growth over the forecast period. For instance, according to the American Cancer Society 2023 update, about 1.93 million new cancer cases were estimated to be reported in 2023 compared to 1.9 million cases in 2022 in the United States. Thus, an increase in the burden of cancer is expected to augment the demand for Ablation Devices, thereby boosting market growth over the forecast period.
Similarly, an increase in breast cancer cases is expected to raise the demand for ablation therapies like Laser Ablation of breast tumors, which is a United States Food and Drug Administration and Conformité Européenne approved procedure, thereby facilitating the Tumor Ablation Market Trends over the forecast period. For instance, in September 2023, the Government of Canada reported that about 28,600 Canadian women were diagnosed with breast cancer in 2022, i.e., 25% of all new cancer cases in women in 2022.
Furthermore, multiple potential ablative technologies are being implemented to surmount the current limitations of Thermal Ablation technologies, which are likely to facilitate market growth over the forecast period. For instance, as per an article published in the Korean Journal of Radiology in June 2023, no-touch Radiofrequency Ablation has gained attention and has recently been implemented in local ablation therapy for hepatocellular carcinoma, as it provides improved local tumor control compared to conventional tumor-puncturing radiofrequency ablation. Thus, the adoption of new technologies is likely to bolster the market growth over the forecast period.
Moreover, robotics in Interventional Oncology can be of great help to reduce radiation exposure to operators and can increase procedure accuracy in the future. Robotic systems offering off-plane and multiplanar percutaneous intervention planning, targeting, as well as needle positioning, also using three-dimensional target views, are available for clinical use and can easily support practitioners.
The rising innovations and product launches by key market players over the forecast period are likely to boost market growth. For instance, in April 2023, Compal Electronics, a Taiwanese electronics company, launched a new Radiofrequency Ablation Market (RFA) system for percutaneous, intraoperative coagulation and ablation of soft tissue, including partial or complete ablation of non-resectable liver lesions.
Therefore, due to the increase in cases of cancer and the rise in product launches coupled with technological advancements in Tumor Ablation Devices, the studied market is expected to grow significantly during the study period.
However, a strict regulatory process is expected to restrain the market's growth during the study period.
Tumor Ablation Market Trends
Lung Cancer Segment to is Expected to Witness Healthy Growth Over the Forecast Period
Lung cancers are usually grouped into two main types called small cell and non-small cell (non-small cell includes adenocarcinoma and squamous cell carcinoma). These types of lung cancer grow differently and are treated differently. Non-small cell lung cancer is more common than small cell lung cancer. Lung cancer is a type of cancer that starts when abnormal cells grow in an uncontrolled way in the lungs. It is a serious health issue that can cause severe harm and death, necessitating advanced treatments like Lung Tumor Ablation.
The increasing cases of lung cancer, rising research works, and funding on cancer research are the major drivers for the Lung Tumor Ablation Market. For instance, according to the report published by the American Lung Association in January 2022, there were 654,620 men and women in the United States with a history of lung cancer as of January 1, 2022. In addition, the same source also stated that around 80% of these individuals were 65 years of age or older, reflecting the advanced median age of diagnosis (71 years). Furthermore, more than half (55.0%) of lung cancer survivors were diagnosed within the past 5 years because of the low survival rate. The same source also stated that an estimated 238,340 people (117,550 men and 120,790 women) were diagnosed with lung cancer, and 127,070 people died from the disease in 2023.
Furthermore, increasing lung cancer research funding is also expected to drive the demand for advanced treatment options such as Tumor Ablation. For instance, in December 2022, the Lung Cancer Research Foundation (LCRF) announced the grant of three new areas of funding in addition to its pilot grants: LCRF Research Grant on Early Detection and Pre-Neoplasia in Lung Cancer; LCRF Research Grants on Oncogenic Drivers in Lung Cancer in partnership with several patient-led oncogene groups; and the newly launched LCRF Minority Career Development Award (CDA) for Lung Cancer.
Similarly, in August 2023, the National Comprehensive Cancer Network (NCCN) Oncology Research Program (ORP) announced grants awarded to foster and enhance quality of care, guideline concordance, and innovations in care delivery for patients with limited-stage (LS) and extensive-stage (ES) small cell lung cancer (SCLC). The NCCN ORP will oversee the projects, and the funding will be provided through support from AstraZeneca. Radiofrequency Ablation (RFA) and Microwave Ablation are widely used for the treatment of lung cancer. Radiofrequency ablation uses high-energy radio waves to heat and destroy the lung tumor cells, while microwave ablation uses electromagnetic energy to heat and destroy the tumor cells.
Thus, owing to the increasing cases of cancer and funding on cancer research, considerable segment growth is expected over the forecast period.
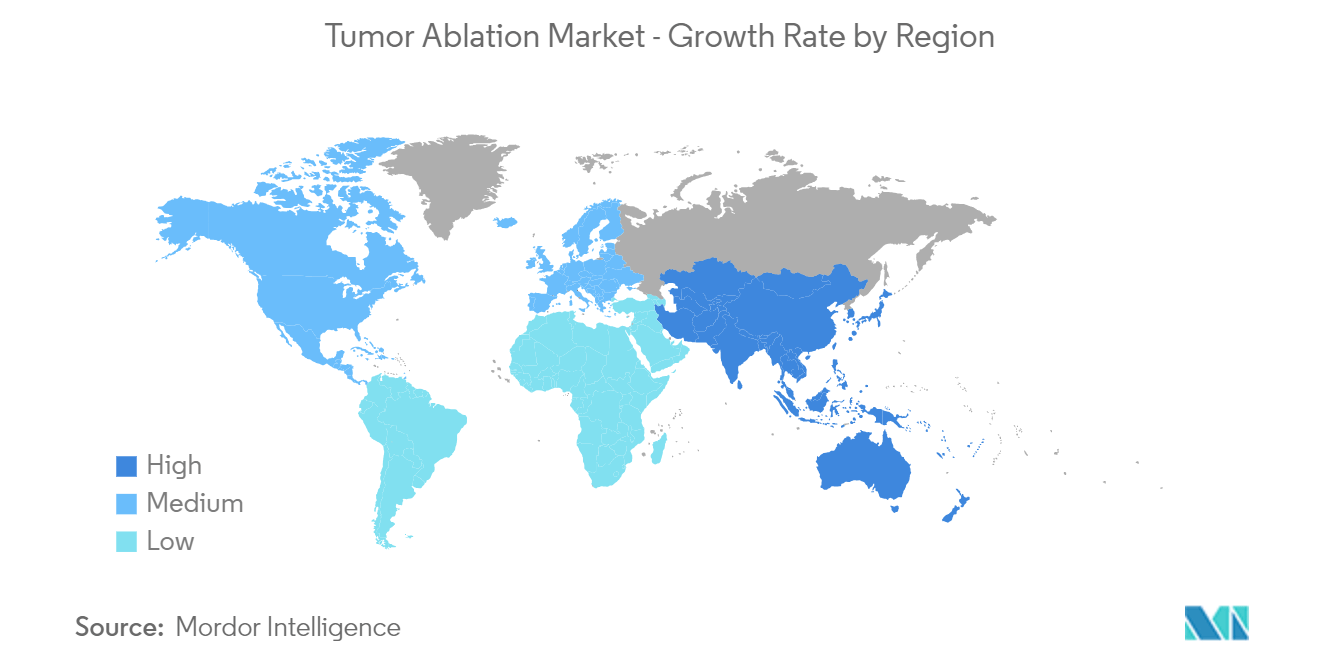
North America is Expected to Hold a Significant Market Share Over the Forecast Period
The North American region is expected to contribute significantly to the market growth during the study period owing to factors such as the growing cancer burden, increasing research and development procedures, and increasing product launches in the Tumor Ablation Market.
For instance, according to the American Cancer Society 2023 Cancer Statistics, new cancer cases are estimated to be 1.9 million in the United States in 2023. This estimation includes 1.01 million cases of males and 0.94 million cases of females. Similarly, as per the Statistics Canada 2022 update, 30,000 Canadians were diagnosed with lung and bronchus cancer in 2022. The high burden of cancer is expected to augment the demand for Tumor Ablation devices, thereby boosting market growth over the forecast period. Furthermore, the launch and approval of new products in the market are expected to impact the country's market growth positively. For instance, in March 2022, Quantum Surgical, a developer of ablation devices and other medical devices, received FDA approval for Epione, a new category of Interventional Oncology robotics dedicated to minimally invasive liver cancer treatment. The company's Epione system helps plan and perform minimally invasive ablation surgery as an outpatient procedure by deploying computer-guided needles through the skin to single out and destroy tumors.
The United States is expected to witness considerable growth in the North American region over the forecast period. The product launches and approvals by key players are the major drivers for the country. For instance, in September 2022, Stryker received 510(k) clearance from the Food and Drug Administration (FDA) for its OptaBlate bone Tumor Ablation System Market (OptaBlate). Similarly, in June 2022, Medtronic introduced a minimally invasive lung ablation procedure with the ILLUMISITE fluoroscopic navigation platform, which helps healthcare professionals to identify the distant areas of the lung accurately and can sample multi-directionally for a thorough biopsy.
Thus, due to the increase in cancer cases and the rise in product launches and approvals, the studied region is expected to hold a significant market share over the forecast period.
Tumor Ablation Industry Overview
The Tumor Ablation Market is consolidated and consists of a few major players. With technological advancements, it is believed that new companies will enter the market in the near future with their novel techniques. The major market players include Medtronic Plc, Boston Scientific Corporation, AngioDynamics Inc., Stryker, Merit Medical Systems, and Olympus Corporation, among others.
Tumor Ablation Market Leaders
-
Merit Medical Systems
-
Boston Scientific Corporation
-
Medtronic
-
Stryker
-
Johnson & Johnson Services, Inc.
- *Disclaimer: Major Players sorted in no particular order
Tumor Ablation Market News
- In January 2024, Techsomed Medical Technologies LTD received De Novo clearance from the Food and Drug Administration for the next-generation ultrasound-based liver ablation software BioTraceIO, developed for tissue response prediction. This software is integrated with a computational algorithm and will help healthcare professionals conduct liver tumor ablations with enhanced visualization of the ablated region with real-time ultrasound imaging. This de-novo clearance will help the company commercialize BioTraceIO in the United States market.
- In September 2023, Techsomed Medical Technologies LTD received 510(k) clearance from the United States Food and Drug Administration (FDA) for its Visible.IO software, which is designed to assist healthcare professionals in liver ablation procedures and improves the accuracy of the treatment.
Tumor Ablation Industry Segmentation
Tumor ablation is an image-guided technique in which the cancer cells in the tumor are killed through the delivery of concentrated energy or chemicals; the energy or chemicals are delivered through probes/needles.The tumor ablation market is segmented by technology (radiofrequency ablation, microwave ablation, cryoablation, and other technologies), mode of treatment (surgical ablation, laparoscopic ablation, and percutaneous ablation), application (liver cancer, lung cancer, kidney cancer, bone metastasis, and other applications), and geography (North America, Europe, Asia-Pacific, Middle East and Africa, and South America). The market report also covers the estimated market sizes and trends for 17 countries across major regions globally.
| By Technology | Radiofrequency (Rf) Ablation | ||
| Microwave Ablation | |||
| Cryoablation | |||
| Other Technologies | |||
| By Mode of Treatment | Surgical Ablation | ||
| Laparoscopic Ablation | |||
| Percutaneous Ablation | |||
| By Application | Liver Cancer | ||
| Lung Cancer | |||
| Kidney Cancer | |||
| Bone Metastasis | |||
| Other Applications | |||
| Geography | North America | United States | |
| Canada | |||
| Mexico | |||
| Europe | Germany | ||
| United Kingdom | |||
| France | |||
| Italy | |||
| Spain | |||
| Rest of Europe | |||
| Asia-Pacific | China | ||
| Japan | |||
| India | |||
| Australia | |||
| South Korea | |||
| Rest of Asia-Pacific | |||
| Middle East and Africa | GCC | ||
| South Africa | |||
| Rest of Middle East and Africa | |||
| South America | Brazil | ||
| Argentina | |||
| Rest of South America | |||
| Radiofrequency (Rf) Ablation |
| Microwave Ablation |
| Cryoablation |
| Other Technologies |
| Surgical Ablation |
| Laparoscopic Ablation |
| Percutaneous Ablation |
| Liver Cancer |
| Lung Cancer |
| Kidney Cancer |
| Bone Metastasis |
| Other Applications |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
Tumor Ablation Market Research FAQs
How big is the Tumor Ablation Market?
The Tumor Ablation Market size is expected to reach USD 1.76 billion in 2025 and grow at a CAGR of 10.10% to reach USD 2.85 billion by 2030.
What is the current Tumor Ablation Market size?
In 2025, the Tumor Ablation Market size is expected to reach USD 1.76 billion.
Who are the key players in Tumor Ablation Market?
Merit Medical Systems, Boston Scientific Corporation, Medtronic, Stryker and Johnson & Johnson Services, Inc. are the major companies operating in the Tumor Ablation Market.
Which is the fastest growing region in Tumor Ablation Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Tumor Ablation Market?
In 2025, the North America accounts for the largest market share in Tumor Ablation Market.
What years does this Tumor Ablation Market cover, and what was the market size in 2024?
In 2024, the Tumor Ablation Market size was estimated at USD 1.58 billion. The report covers the Tumor Ablation Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Tumor Ablation Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on: June 16, 2024
Our Best Selling Reports
Tumor Ablation Industry Report
The Tumor Ablation Market is segmented by technology, mode of treatment, application, and geography. The market size and market forecast are crucial aspects of this industry, providing insights into the market trends and market share across different regions. The global market for tumor ablation includes various technologies such as radiofrequency ablation, microwave ablation, cryoablation, and other technologies. The market forecast indicates a positive growth rate, with significant contributions from North America, Europe, Asia-Pacific, the Middle East and Africa, and South America.
Industry reports and industry analysis highlight the importance of understanding the market segmentation and market value. The market growth is driven by advancements in technology and increasing demand for minimally invasive treatments. Industry information and industry research provide a comprehensive market overview, detailing the market data and market predictions for the coming years.
Market leaders in the tumor ablation industry are focusing on expanding their market share through strategic partnerships and innovative product launches. The industry outlook and market outlook suggest a promising future for the tumor ablation market, with continuous improvements in industry statistics and industry trends.
Research companies play a vital role in providing accurate market analysis and market review, contributing to the overall industry size and industry sales. The report example and report PDF offer valuable insights into the market segmentation, including applications such as liver cancer, lung cancer, kidney cancer, bone metastasis, and other applications.
In summary, the tumor ablation market is poised for significant growth, supported by detailed industry analysis and market research. The market forecast and market growth indicate a strong demand for advanced ablation technologies, with a focus on improving patient outcomes and expanding the global market presence.