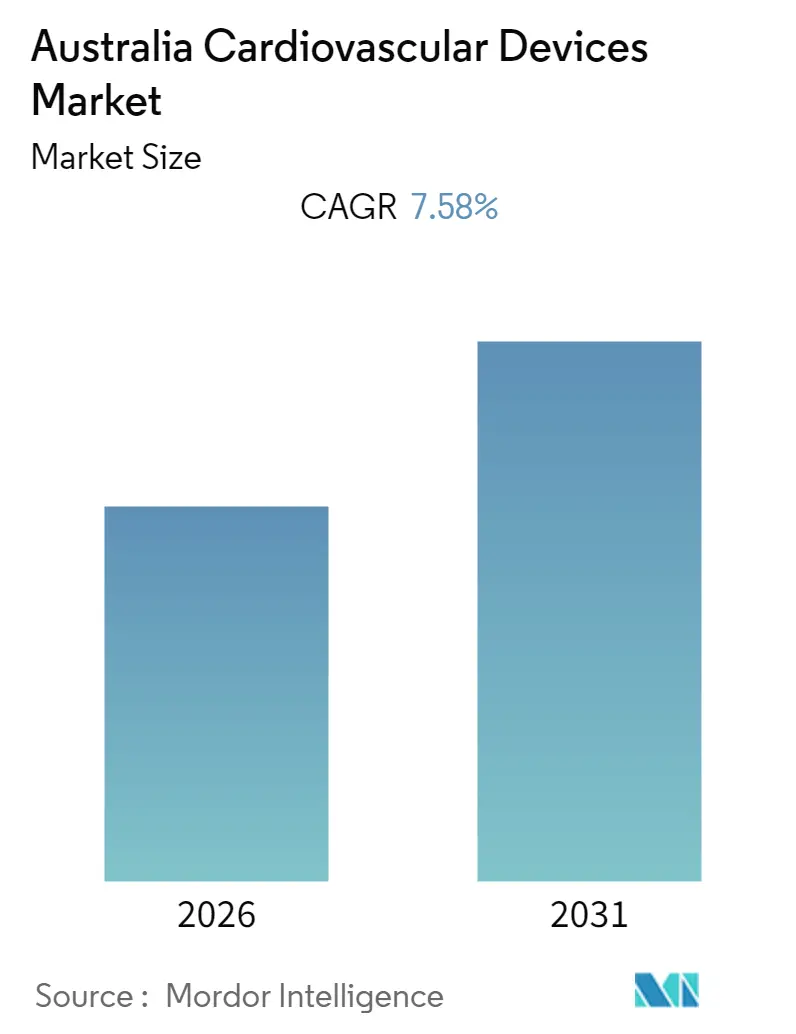
Australia Cardiovascular Devices Market Analysis by Mordor Intelligence
The Australia Cardiovascular Devices Market size is expected to register a CAGR of 7.58% during the forecast period (2026-2031).
- The major factors driving the market are rapid technological advancements, the rising burden of various cardiovascular diseases, and increased preference for minimally invasive procedures. Heart-related mortality is caused by the rising prevalence of heart disorders such as cardiomyopathy and stroke. The most common diseases in the world are those related to the cardiovascular system.
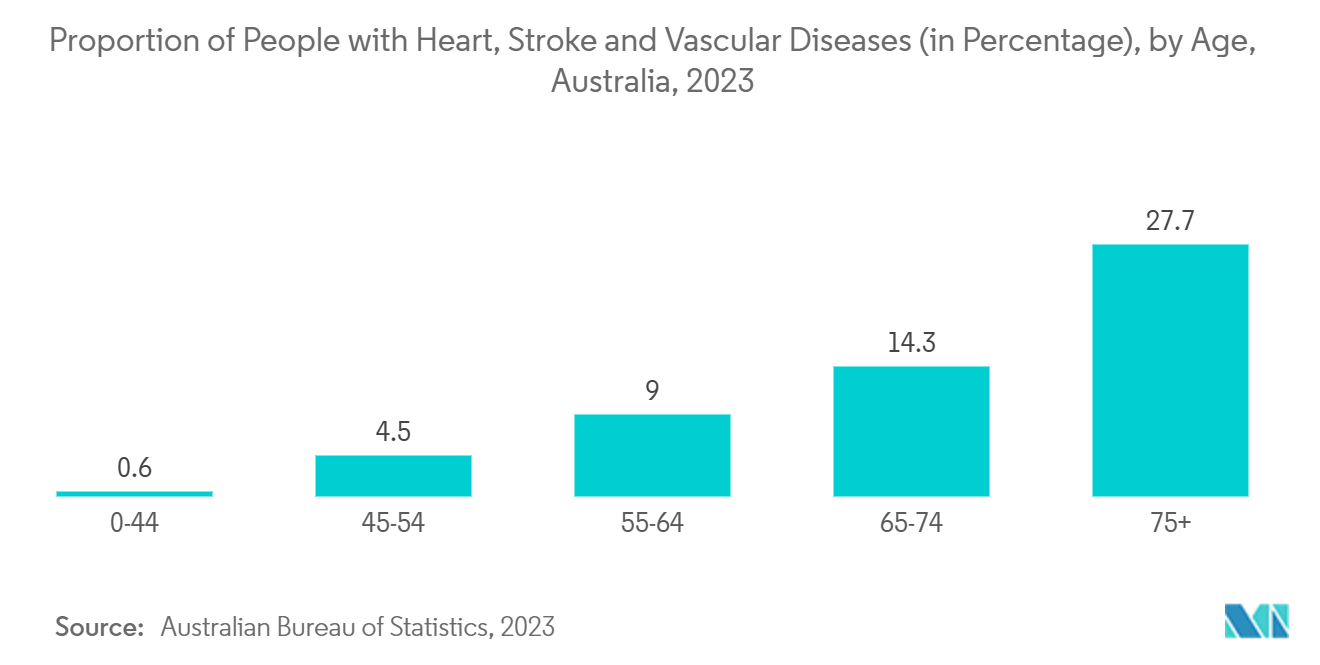
- According to the June 2024 updates from the Australian Institutes of Health and Welfare report, in 2023, Australians lost an estimated 666,000 years of healthy life (DALY) due to all forms of cardiovascular diseases (CVD), equivalent to 25.1 per 1,000 population. In addition, the same source stated that 146,000 coronary angiography procedures were reported for patients admitted to the hospital – 97,200 (67%) for males and 48,900 (33%) for females. Thus, the high burden of cardiovascular diseases is expected to contribute to market growth over the forecast period.
- Heart disease increases with age, which of all ages is expected to drive the demand for cardiovascular devices for the diagnosis and management of these diseases, thereby augmenting market growth. For instance, according to October 2023 data from the University of Sydney, by 2026, more than 22% of Australians will be aged over 65 years. Thus, the growing aging population in the country, which is prone to cardiovascular diseases, is expected to contribute to market growth over the forecast period.
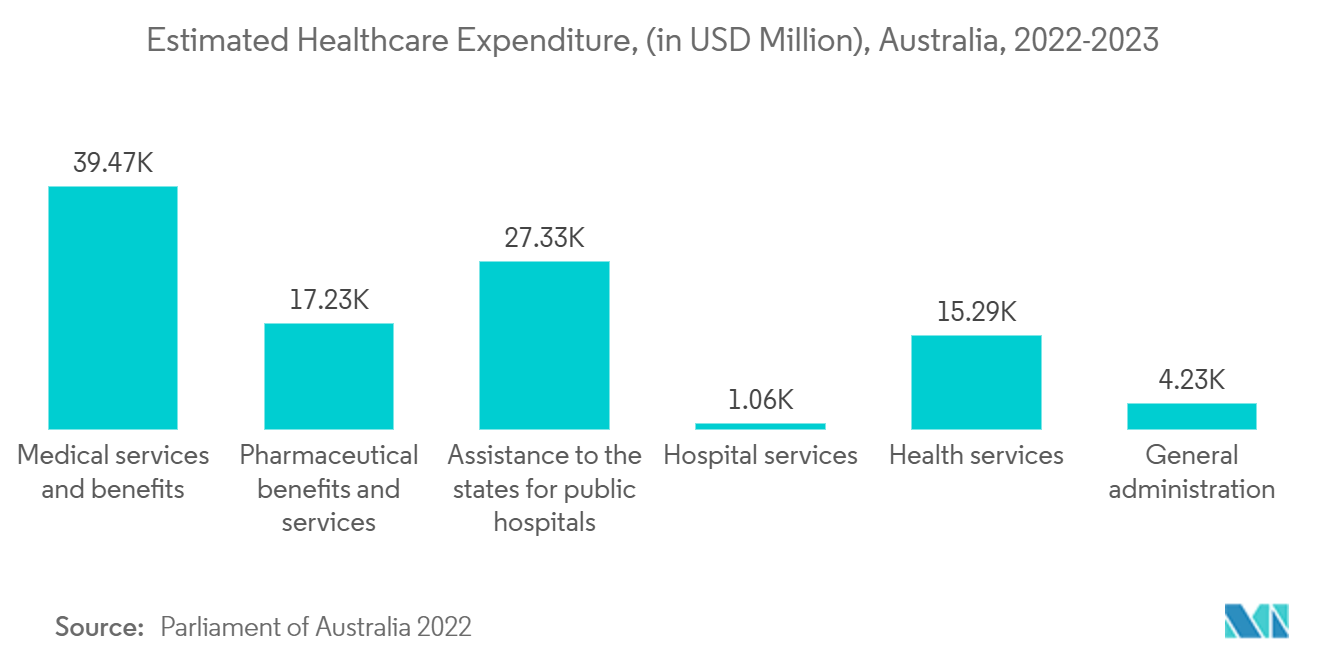
- Moreover, government support, increasing product approvals, and research and development activities undertaken by key market players contribute to the market's growth. For instance, in March 2022, the Regional Ministerial Budget Statement 2022-23 indicated that the Australian government allocated USD 17.2 million over four years to operate five mobile health clinics that will provide consultations and diagnosis in various medical specialties, including cardiology in Queensland.
- Therefore, owing to the rising prevalence of cardiac diseases and technological advancements in cardiovascular diseases, the market studied is expected to witness growth during the forecast period. However, stringent regulatory policies and product recalls are potential impediments to market growth.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Australia Cardiovascular Devices Market Trends and Insights
Electrocardiogram (ECG) Segment is Expected to Witness Growth Over the Forecast Period
- The electrocardiogram (ECG) segment is expected to experience substantial growth in the market studied, primarily due to the rising incidence of cardiac complications, the surge in product launches, and the increasing technological advancements in ECGs. ECGs, also known as electrocardiographs, record the heart's electrical signals and are commonly used equipment in operating rooms and ambulances. Personal devices such as smartwatches also offer ECG monitoring. ECGs can detect irregular heart rhythms, blocked or narrowed arteries in the heart, previous heart attacks, and the functioning of certain heart disease treatments such as pacemakers.
- The growth of the segment is being further boosted by increasing product launches, partnerships, and acquisitions in the country. For instance, in October 2022, AccurKardia, a software company that provides clinical-grade, device-agnostic, automated electrocardiogram (ECG) analytics, announced it would collaborate with Mawi, a MedTech provider of medical-grade wearables in the cardiac space, to integrate its proprietary ECG analytics into Mawi’s cardiac monitoring watch.
- The Victor Chang Cardiac Research Institute and the Charles Perkins Centre at the University of Sydney are two industrial sponsors of the consortium conducting clinical trials for cardiovascular devices. Such collaborative initiatives are expected to encourage medical device research and development in Australia and accelerate the commercialization of cardiovascular devices developed through this R&D, ultimately boosting the segment's growth during the forecast period.
- Therefore, due to the increasing technological advancements in ECGs and the rise in product launches, the segment is anticipated to register significant growth over the forecast period.
Catheters Segment is Anticipated to Witness Growth in the Australian Cardiovascular Devices Market Over the Forecast Period
- The catheter segment is expected to hold a significant share of the market during the forecast period, driven by several factors, including the rising incidence of cardiac diseases, government initiatives, and technological advancements in cardiac catheters. For example, in February 2024, BiVACOR, a clinical-stage medical device company, announced that USD 13 million had been awarded from the Australian government’s Medical Research Future Fund (MRFF) grant through the Artificial Heart Frontiers Program (AHFP) to support BiVACOR’s total artificial heart program and future product enhancements. Government support for cardiovascular services is expected to increase demand for catheters and drive market growth.
- The growth of the catheter segment in Australia is also being fueled by increasing product launches, partnerships, acquisitions, and collaborations. For instance, in February 2022, Vantari VR, an Australian medical virtual reality company, partnered with an internationally renowned cardiologist to develop a Virtual Right Heart Catheter Course to revolutionize clinician training. Such developments are expected to further boost the growth of the country's catheter segment during the forecast period.
- Moreover, approvals and product launches of new cardiac catheters are expected to drive market growth. In August 2022, Millar Inc., a medical device manufacturer and OEM solutions partner, received approval from the Therapeutic Goods Administration (TGA) in Australia for its Mikro-Cath Pressure Catheter, a single-use pressure catheter designed for medical research and diagnostic purposes. These types of developments in cardiac catheters in Australia are expected to contribute to market growth during the forecast period.
- Therefore, with the increasing number of catheter product launches and the rising incidence of cardiac complications, the catheter segment in Australia is expected to witness significant growth over the forecast period.
Competitive Landscape
The cardiovascular devices market in Australia is fragmented due to the presence of multiple companies operating in the region. The competitive landscape includes an analysis of several international and local companies that hold significant market share and are well-known in the industry. Key players in the market include Abbott Laboratories, Boston Scientific Corporation, Cardinal Health Inc., Edwards Lifesciences, General Electric Company (GE Healthcare), W. L. Gore & Associates Inc., Medtronic PLC, Biotronik, Siemens Healthineers AG, Canon Medical Systems Corporation, and B. Braun SE.
Australia Cardiovascular Devices Industry Leaders
Abbott Laboratories
Boston Scientific Corporation
Medtronic PLC
B. Braun SE
Cardinal Health Inc.
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2024: Monash University led a transdisciplinary consortium to develop and commercialize a suite of revolutionary and life-changing implantable cardiac devices that, for the first time, would offer longer-term solutions for all types of debilitating heart failure.
- June 2022: Teleflex Incorporated, a leading global provider of medical technologies, launched its Arrow Pressure Injectable Midline Catheter in Australia. The addition of the pressure injectable catheter further enhances the Midline portfolio to meet the expanded needs of clinicians and is designed to improve patient safety. The new 20-cm Arrow Pressure Injectable Midline with brightly colored yellow hubs and labeling will help clinicians overcome catheter identification confusion, which can lead to infusion mistakes that can harm patients.
Australia Cardiovascular Devices Market Report Scope
As per the scope of the report, cardiovascular devices are used to diagnose and treat heart disease and related cardiovascular problems. Cardiac devices offer monitoring services to hospitals and physicians to take care of the patients with the help of the data collected by cardiac monitors.
The Australian cardiovascular devices market is segmented by device type. By type, the market is segmented as diagnostic and monitoring devices and therapeutic and surgical devices.
The report offers the value (USD) for the above segments.
| Diagnostic and Monitoring Devices | Electrocardiogram (ECG) |
| Remote Cardiac Monitoring | |
| Other Diagnostic and Monitoring Devices | |
| Therapeutic and Surgical Devices | Cardiac Assist Devices |
| Cardiac Rhythm Management Devices | |
| Catheters | |
| Grafts | |
| Heart Valves | |
| Stents | |
| Other Therapeutic and Surgical Devices |
| By Device Type | Diagnostic and Monitoring Devices | Electrocardiogram (ECG) |
| Remote Cardiac Monitoring | ||
| Other Diagnostic and Monitoring Devices | ||
| Therapeutic and Surgical Devices | Cardiac Assist Devices | |
| Cardiac Rhythm Management Devices | ||
| Catheters | ||
| Grafts | ||
| Heart Valves | ||
| Stents | ||
| Other Therapeutic and Surgical Devices | ||


Key Questions Answered in the Report
What is the current Australia Cardiovascular Devices Market size?
The Australia Cardiovascular Devices Market is projected to register a CAGR of 7.58% during the forecast period (2026-2031)
Who are the key players in Australia Cardiovascular Devices Market?
Abbott Laboratories, Boston Scientific Corporation, Medtronic PLC, B. Braun SE and Cardinal Health Inc. are the major companies operating in the Australia Cardiovascular Devices Market.
What years does this Australia Cardiovascular Devices Market cover?
The report covers the Australia Cardiovascular Devices Market historical market size for years: 2019, 2020, 2021, 2022, 2023, 2024 and 2025. The report also forecasts the Australia Cardiovascular Devices Market size for years: 2026, 2027, 2028, 2029, 2030 and 2031.
Page last updated on:








