Atherectomy Devices Market Size and Share
Atherectomy Devices Market Analysis by Mordor Intelligence
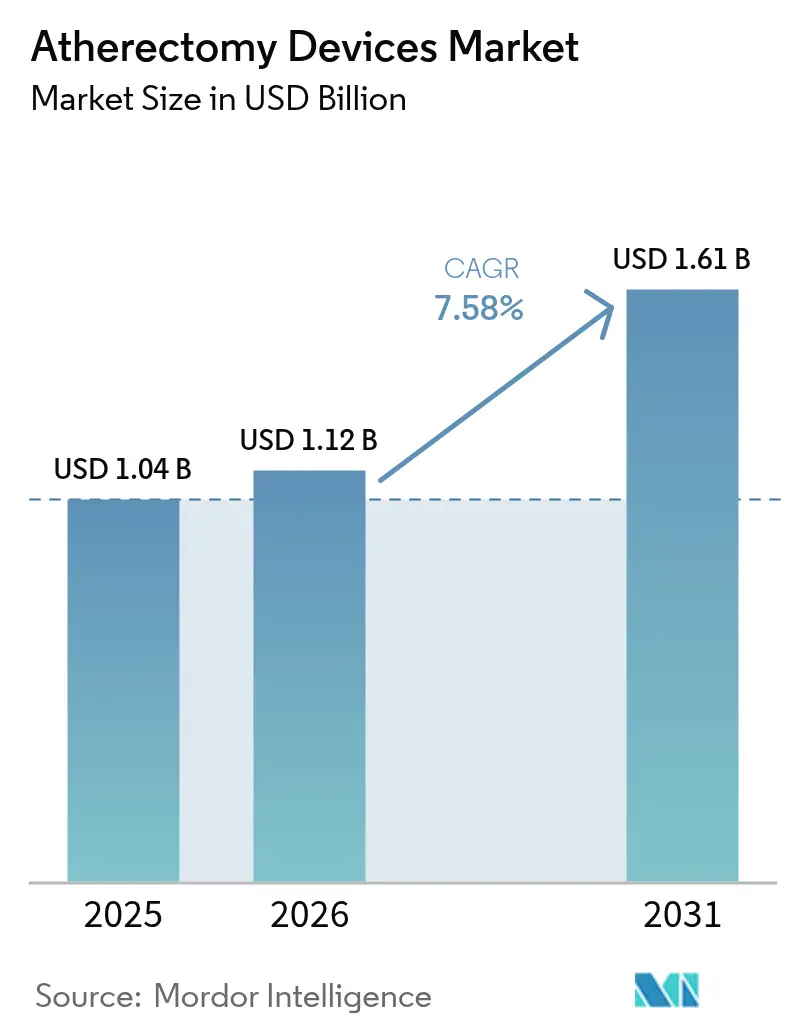
The atherectomy devices market size was valued at USD 1.04 billion in 2025 and estimated to grow from USD 1.12 billion in 2026 to reach USD 1.61 billion by 2031, at a CAGR of 7.58% during the forecast period (2026-2031). Growing acceptance of minimally invasive plaque-modification techniques, broader clinical use in complex calcified lesions, and favorable reimbursement for office-based laboratory (OBL) procedures in the United States underpin near-term demand. Device makers continue to pair atherectomy systems with drug-coated balloons to improve long-term patency, strengthening the therapy’s value proposition in peripheral vascular disease. Strategic acquisitions by multinational suppliers are consolidating intellectual property and global distribution, while Asia-Pacific localization programs are driving cost efficiencies that could accelerate regional adoption. Nonetheless, mixed evidence on long-term outcome superiority over plain balloon angioplasty, coupled with supply-chain tightness for diamond-coated burrs, tempers the five-year outlook.
Key Report Takeaways
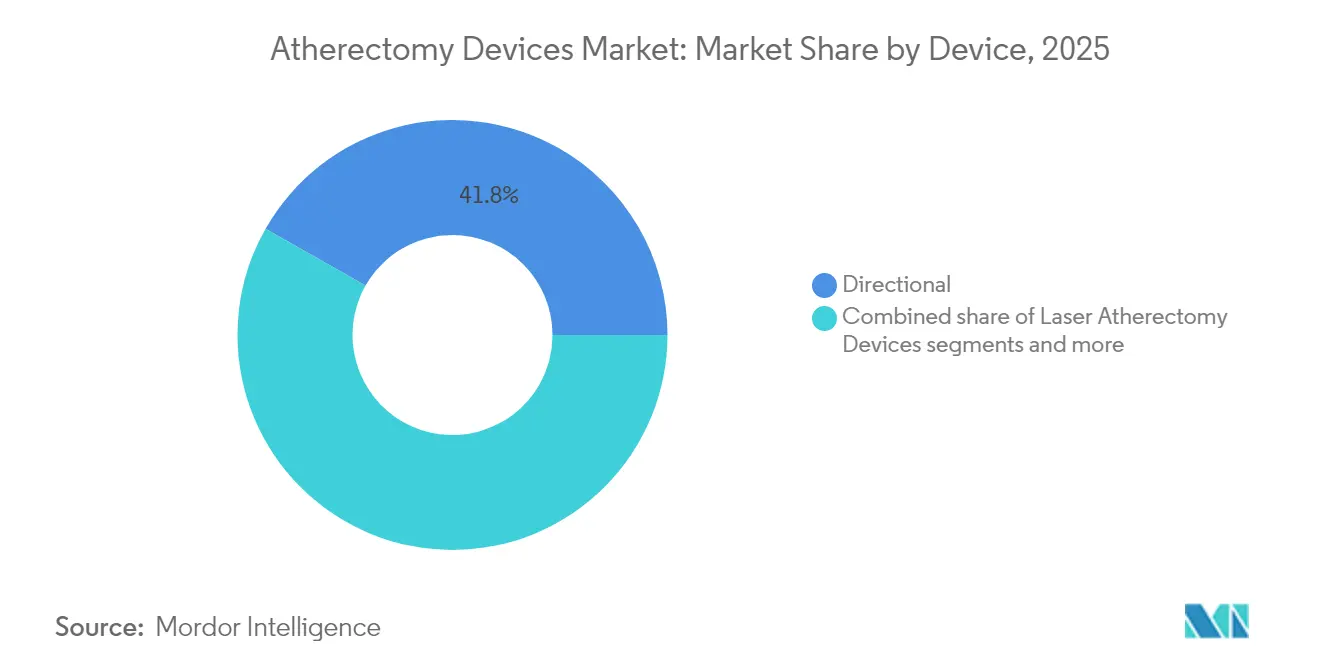
- By device type, directional systems led with 41.78% revenue share of the atherectomy devices market in 2025.
- By application, peripheral vascular disease accounted for 57.45% share of the atherectomy devices market size in 2025, whereas neurovascular procedures are projected to advance at a 7.86% CAGR through 2031.
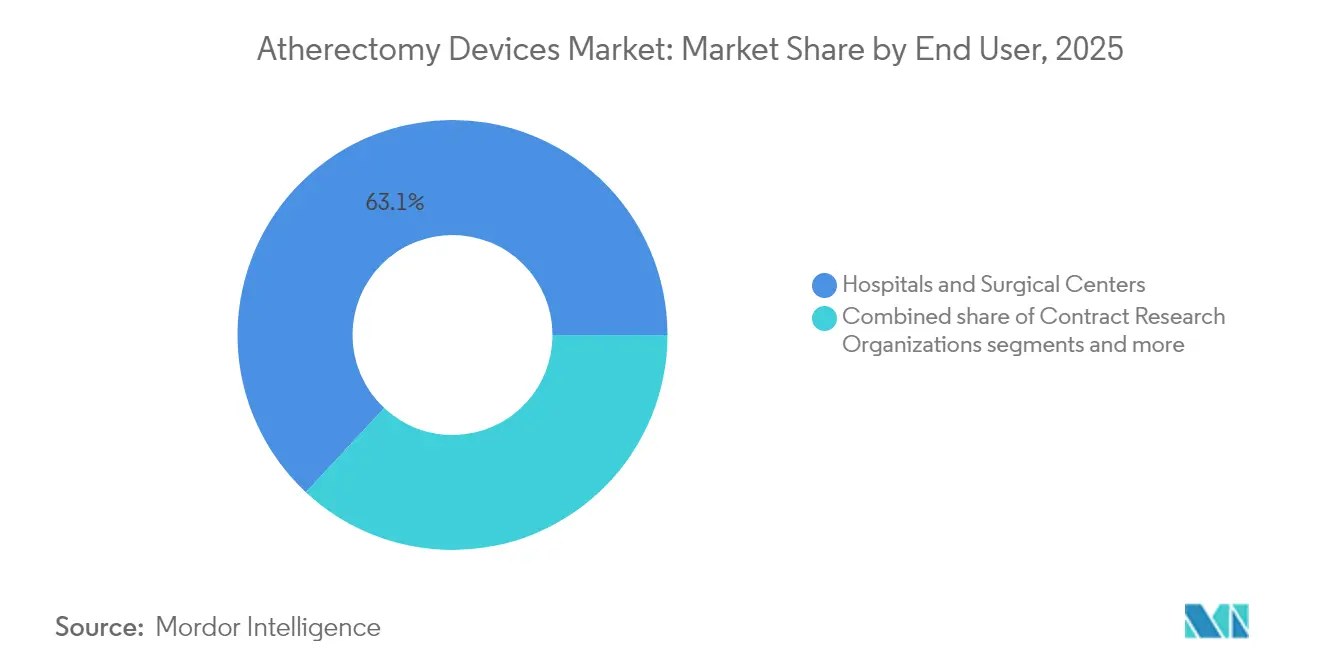
- By end user, hospitals and surgical centers held 63.05% of the atherectomy devices market share in 2025; ambulatory surgical centers are the fastest-growing setting at an 7.93% CAGR to 2031.
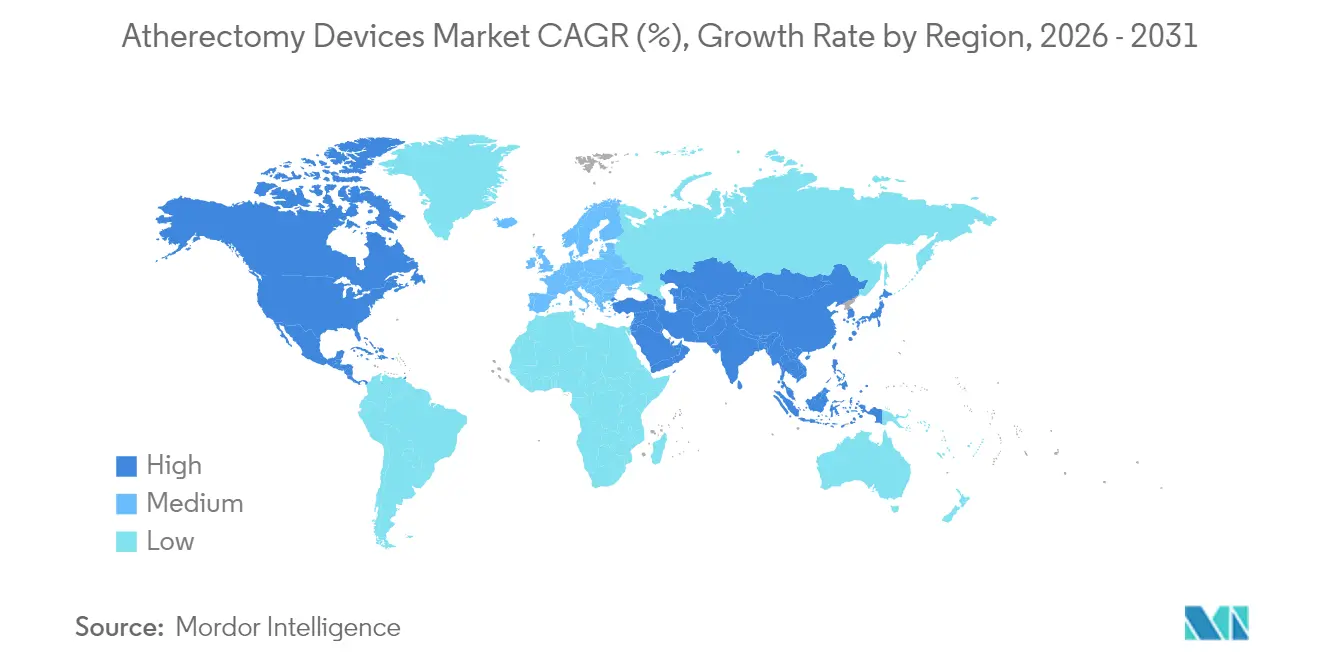
- By geography, North America dominated with 38.74% share in 2025, while Asia-Pacific is forecast to expand at an 8.08% CAGR between 2026 and 2031
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Atherectomy Devices Market Trends and Insights
Driver Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising prevalence of peripheral & coronary artery disease | +2.1% | North America, Europe, global spill-over | Long term (≥ 4 years) |
| Growing adoption of minimally invasive endovascular procedures | +1.8% | Global, strongest in developed economies | Medium term (2-4 years) |
| Favorable reimbursement incentives for U.S. office-based labs | +1.2% | United States | Short term (≤ 2 years) |
| Laser systems with active aspiration lower distal-embolization risk | +0.9% | North America, Europe | Medium term (2-4 years) |
| Domestic manufacturing push in Asia-Pacific | +0.8% | Asia-Pacific | Long term (≥ 4 years) |
| Synergistic outcomes of atherectomy plus drug-coated balloons | +0.7% | Global | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Rising Prevalence of Peripheral & Coronary Artery Disease
Peripheral artery disease affects more than 200 million people worldwide, most acutely in aging populations with diabetes and metabolic syndrome. The expanding burden raises procedural complexity because heavily calcified lesions respond poorly to plain balloon angioplasty. Current U.S. and European guidelines now recommend endovascular interventions for infra-inguinal lesions under 25 cm, opening a larger pool of candidates for atherectomy. Hospitals report that older patients present with longer, more complex occlusions that require vessel-preparation techniques to achieve acceptable luminal gain. Consequently, device makers emphasize systems able to debulk eccentric calcium while minimizing embolic events. The long-term demographic tailwind supports double-digit growth in procedure volumes for at least the next decade.
Growing Adoption of Minimally Invasive Endovascular Procedures
Hospitals and outpatient centers prefer shorter stays and quicker ambulation, pushing physicians to select techniques that reduce anesthesia time and wound complications. Medicare reimbursements for OBL interventions reached USD 6.8 billion across 6,308 facilities in 2023. Atherectomy fits this shift because most systems are compatible with 6Fr sheaths, allowing same-day discharge. Ambulatory surgical centers have posted an 8.06% CAGR in atherectomy procedure counts as vascular surgeons and advanced practice providers gain proficiency. Rising comfort among payors with OBL safety metrics further lifts utilization, reinforcing a feedback loop that sustains capital purchases of new systems. Comparable trends in Europe and advanced Asia-Pacific economies are visible albeit at lower absolute volumes.
Favorable Reimbursement Incentives for Office-Based Labs (U.S.)
Historically, CMS set higher relative value units for peripheral atherectomy in OBLs than in hospital outpatient departments, creating a financial tailwind for community-based vascular practices. While the 2024 Physician Fee Schedule reduced payment for over 300 OBL codes, atherectomy still commands sufficient margin for practices operating at moderate volume. Recent code assignments for intravascular lithotripsy, which often complements atherectomy, boosted average payments by USD 8,000, signaling that innovative vessel-preparation modalities can secure premium coverage when backed by robust evidence. The interplay between reimbursement revisions and clinical guidelines will determine whether OBLs maintain their current share of the atherectomy devices market.
Laser Systems with Active Aspiration Lower Distal-Embolization Risk
Second-generation laser catheters integrate aspiration ports proximal to the ablation window, removing particulate debris before it migrates downstream. In a 402-patient registry, the Phoenix system achieved procedural success exceeding 99%, with embolization rates of only 2%. These safety gains are critical as operators tackle longer chronic total occlusions and severely calcified in-stent restenosis that previously required open bypass. Reduced complication risk expands eligibility among frail patients and those with limited runoff vessels, thereby widening the addressable base for laser-enabled atherectomy.
Restraint Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Limited long-term evidence of superiority versus PTA/stenting | −1.4% | United States, Europe | Medium term (2-4 years) |
| High device and capital costs | −1.1% | Global | Short term (≤ 2 years) |
| Imminent reimbursement cuts tied to utilization reviews | −0.8% | United States, developed markets | Short term (≤ 2 years) |
| Supply-chain risk for diamond-coated burr materials | −0.6% | Global | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Limited Long-Term Evidence of Superiority vs. PTA/Stenting
The 2,005-patient ECLIPSE trial reported no statistical advantage for orbital atherectomy over plain balloon angioplasty in preventing target vessel failure before drug-eluting stent implantation00450-7/abstract). This neutral outcome echoes prior peripheral registries in which atherectomy reduced amputation risk yet increased repeat intervention rates. Evidence-based payors may therefore restrict reimbursement to subgroups with demonstrated benefit, constraining volume growth in regions that prioritize randomized data over operator preference.
High Device & Capital Costs for Providers
Rotational atherectomy consoles can exceed USD 150,000 and require single-use burrs priced above USD 2,000. OBL operators already face CMS payment that covers barely one-fifth of thrombectomy procedural costs, forcing careful case selection. Capital intensity favors integrated health systems capable of amortizing equipment across multiple sites, potentially squeezing smaller vascular practices out of the atherectomy devices market.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Device Type: Directional Systems Anchor Market Leadership
Directional systems generated 41.78% of atherectomy devices market revenue in 2025, reflecting surgeon preference for precise plaque excision and compatibility with post-dilation balloons. The atherectomy devices market size for directional platforms is projected to grow at a steady 7.08% CAGR to 2031 as new cutter-head geometries improve crossing profile without sacrificing debulking capacity. Laser catheters represent the fastest-rising modality at 7.74% CAGR because aspiration-assisted designs significantly lower embolic risk. Rotational and orbital devices remain essential for densely calcified coronary and iliac lesions yet face scrutiny following neutral ECLIPSE outcomes.
Competitive positioning hinges on differentiated burr coatings, real-time feedback algorithms, and bundled service contracts. Abbott’s Diamondback 360 leverages a diamond-embedded crown that orbits eccentrically to sand down calcium, whereas Boston Scientific’s Jetstream incorporates front-cutting blades plus aspiration for in-stent restenosis. University-industry projects such as milli-scale robotics foreshadow next-generation systems capable of remote navigation through tortuous neurovascular beds. FDA guidance on Predetermined Change Control Plans allows manufacturers to pre-specify software updates, shortening iteration cycles and accelerating innovation cadence.

By Application: Peripheral Vascular Disease Dominates, Neurovascular Surges
Peripheral vascular disease interventions captured 57.45% of atherectomy devices market share in 2025 as femoropopliteal and below-the-knee lesions make up the bulk of global endovascular workload. The segment benefits from rising diabetes incidence and wider screening programs that detect occlusive disease earlier. Neurovascular procedures, however, will post the fastest expansion at 7.86% CAGR, propelled by growth in middle meningeal artery embolization for chronic subdural hematomas. Combination therapy strategies that unite clot retrieval, mechanical debulking, and pharmacologic lysis promise to broaden atherectomy’s role in stroke-adjacent care pathways. Coronary artery disease remains a steady but slower-growing application, with adoption tempered by neutral trial data and abundant stent options.
By End User: Outpatient Migration Reshapes Demand
Hospitals and tertiary surgical centers accounted for 63.05% of atherectomy devices market revenue in 2025 thanks to comprehensive imaging and standby surgical backup. Yet ambulatory surgical centers are scaling faster—an 7.93% CAGR—because payors reward same-day discharge and lower facility fees. The atherectomy devices market size tied to ambulatory volumes could surpass USD 668.3 million by 2031 if reimbursement remains favorable. OBLs occupy a middle ground; they thrived on 2018-2023 payment levels but now face margin compression. Quality-focused OBL operators respond by integrating outcomes registries and strict appropriateness criteria to safeguard payer confidence.

Geography Analysis
North America generated 38.74% of atherectomy devices market revenue in 2025 on the back of high procedural density, entrenched reimbursement, and robust clinical research infrastructure. The region’s growth moderates to mid-single-digit rates as volume shifts from hospital inpatient to outpatient venues. The atherectomy devices market size in Asia-Pacific will expand at an 8.08% CAGR through 2031, underpinned by domestic manufacturing incentives and accelerating cardiovascular disease prevalence. China’s Fourteenth Five-Year Plan calls for local production of high-end interventional devices, positioning regional firms to undercut imports without compromising quality. Japan and South Korea, meanwhile, leverage mature reimbursement systems that now recognize vessel-preparation codes, stimulating early adoption.
Europe remains the third-largest regional segment, characterized by evidence-oriented purchasing committees influenced by the Medical Device Regulation. Hospitals emphasize long-term safety registries; therefore, suppliers that document reduced reintervention rates achieve preferred-vendor status. Innovation uptake is fastest in Germany and the Netherlands where OBL equivalents—practice-based cath labs—operate under bundled diagnosis-related group payments. Latin America and Middle East & Africa collectively account for less than 10% of global sales but show high-single-digit growth as private insurers expand coverage for peripheral vascular procedures.
Competitive Landscape
The atherectomy devices market is moderately consolidated, with the top five suppliers controlling significant global revenue. Boston Scientific, Medtronic, Abbott, Teleflex, and Stryker employ mergers to fill technology gaps and extend channel reach. In January 2025, Stryker closed a USD 4.9 billion purchase of Inari Medical, securing thrombectomy know-how that complements its existing Jetstream franchise. Teleflex followed by acquiring Biotronik’s vascular intervention unit for €760 million in July 2025, securing a European manufacturing foothold.
Product differentiation has shifted from raw cutting efficiency to integrated therapy platforms combining vessel-preparation, antiproliferative drug delivery, and intelligent data capture. Vendors roll out cloud-connected consoles that log run time, cutter torque, and aspiration flow, feeding predictive maintenance algorithms that lower downtime. Smaller innovators target niche neurovascular or robotic niches, betting that faster FDA software iteration pathways will level the playing field.
Third-party service organizations offering per-click pricing on capital equipment are emerging, especially in low-volume community hospitals. Such models lower upfront cost but lock customers into proprietary consumables, reinforcing brand stickiness. Competitive intensity is expected to rise as Asian OEMs secure CE and FDA clearances by 2027, introducing price competition in value-sensitive markets.
Atherectomy Devices Industry Leaders
Abbott Laboratories
Boston Scientific Corporation
Medtronic
Becton, Dickinson and Company
Koninklijke Philips N.V.
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- July 2025: Teleflex completed acquisition of Biotronik’s Vascular Intervention business for €760 million, broadening its peripheral debulking portfolio and boosting European sales reach
- May 2025: FDA granted 510(k) clearance to Crossroads Neurovascular’s PATH BGC balloon guide catheter, expanding atherectomy-adjacent options for cerebrovascular interventions
Global Atherectomy Devices Market Report Scope
As per the scope of the report, atherectomy is a minimally invasive endovascular procedure generally performed to remove atherosclerosis from blood vessels in the body. It is generally used to treat arteries. It can be used in veins and vascular bypass grafts as well. The atherectomy devices market is segmented by product (directional atherectomy systems, orbital atherectomy systems, photo-ablative (laser) atherectomy systems, and rotational atherectomy systems), application (peripheral vascular applications, cardiovascular applications, and neurovascular applications), end user (hospitals and surgical centers, ambulatory care centers, and other end users), and geography (North America, Europe, Asia-Pacific, Middle East and Africa, and South America). The report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD million) for the above segments.
| Directional Atherectomy Devices |
| Rotational Atherectomy Devices |
| Orbital Atherectomy Devices |
| Laser Atherectomy Devices |
| Peripheral Vascular Disease |
| Coronary Artery Disease |
| Neurovascular Disease |
| Hospitals & Surgical Centers |
| Ambulatory Surgical Centers |
| Office-Based Labs |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| South Korea | |
| Australia | |
| Rest of Asia-Pacific | |
| South America | Brazil |
| Argentina | |
| Rest of South America | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa |
| By Device Type (Value) | Directional Atherectomy Devices | |
| Rotational Atherectomy Devices | ||
| Orbital Atherectomy Devices | ||
| Laser Atherectomy Devices | ||
| By Application (Value) | Peripheral Vascular Disease | |
| Coronary Artery Disease | ||
| Neurovascular Disease | ||
| By End User (Value) | Hospitals & Surgical Centers | |
| Ambulatory Surgical Centers | ||
| Office-Based Labs | ||
| by Geography (Value) | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| South Korea | ||
| Australia | ||
| Rest of Asia-Pacific | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||


Key Questions Answered in the Report
What is the projected value of the atherectomy devices market in 2031?
The market is expected to reach USD 1.61 billion by 2031 at a 7.58% CAGR.
Which device category currently holds the largest share?
Directional systems command 41.78% of 2025 revenue due to precise plaque excision capabilities.
Which regional segment will grow fastest through 2031?
Asia-Pacific is forecast to expand at an 8.08% CAGR, supported by domestic manufacturing initiatives.
How is reimbursement influencing outpatient utilization?
Despite recent CMS cuts, atherectomy still yields positive margins in many OBLs, sustaining outpatient migration.
What evidence challenges routine use in coronary lesions?
The ECLIPSE trial found no target vessel failure advantage for orbital atherectomy over balloon-first strategies, questioning its routine application.
Page last updated on:









