Acute Respiratory Distress Syndrome Treatment Market Size
Acute Respiratory Distress Syndrome Treatment Market Analysis
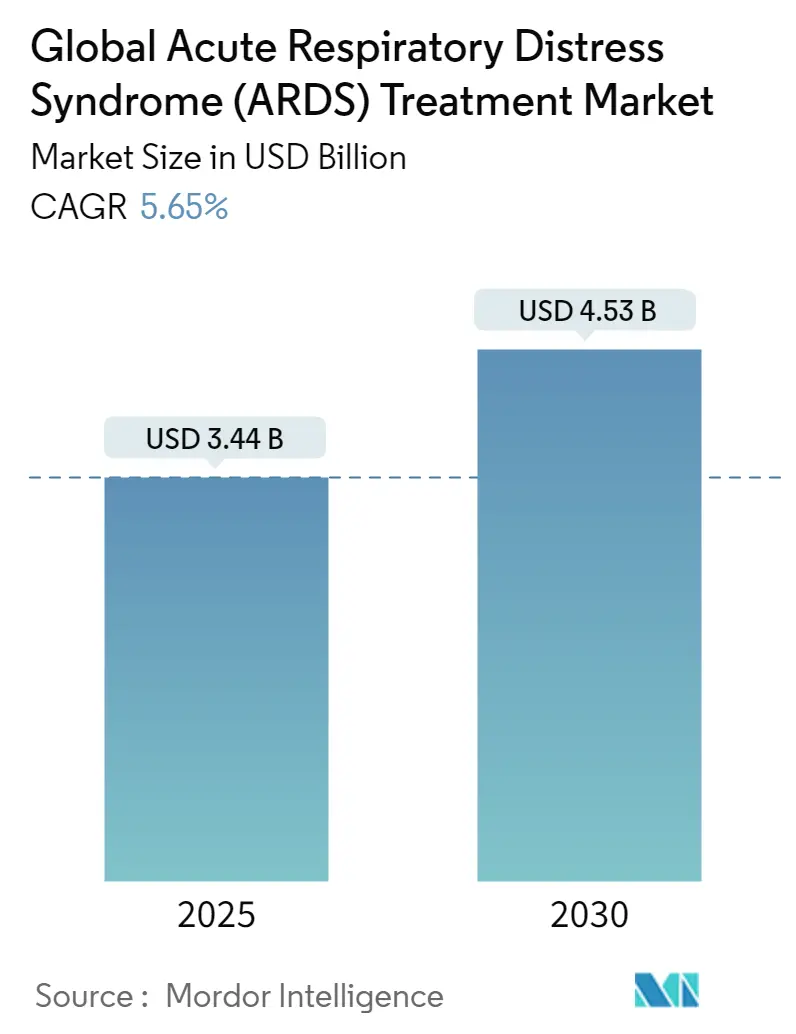
The Global Acute Respiratory Distress Syndrome Treatment Market size is estimated at USD 3.44 billion in 2025, and is expected to reach USD 4.53 billion by 2030, at a CAGR of 5.65% during the forecast period (2025-2030).
The acute respiratory distress syndrome (ARDS) treatment landscape has evolved significantly with advanced therapeutic approaches and innovative technologies. According to recent epidemiological data from 2022, approximately 190,000 ARDS cases are reported annually in the United States, with incidence rates varying dramatically by age group, from 16 per 100,000 person-years in young adults to 306 per 100,000 person-years in elderly patients. The treatment paradigm has shifted towards more personalized approaches, with healthcare providers increasingly adopting combination therapies that include both pharmacological interventions and advanced respiratory support systems devices. This evolution has led to improved patient outcomes and has created opportunities for innovative treatment modalities in the market.
The research and development landscape has witnessed substantial advancement in biomarker-based approaches and novel therapeutic interventions. In March 2023, Tetra Bio-Pharma received significant funding of USD 150,000 from the National Research Council of Canada Industrial Research Assistance Program for developing novel oral therapeutics targeting systemic hyperinflammation. This development represents a broader trend of increasing investment in innovative respiratory therapeutic approaches, with several pharmaceutical companies focusing on developing targeted therapies that address the underlying pathophysiology of ARDS rather than just managing symptoms.
The regulatory environment continues to evolve favorably for ARDS treatments, with authorities demonstrating increased flexibility in their approval processes for innovative therapies. In April 2022, the FDA granted Direct Biologics approval to proceed with a landmark Phase 3 clinical trial for their investigational EV Drug ExoFlow, marking a significant milestone in ARDS treatment development. The regulatory landscape has become more accommodating to novel therapeutic approaches, particularly those demonstrating promising results in early-stage trials, which has accelerated the pathway from development to market approval.
Strategic collaborations and partnerships have emerged as key drivers of innovation in the ARDS treatment market. Companies are increasingly forming alliances to combine their technological expertise and research capabilities, leading to more effective treatment solutions. For instance, in January 2023, Telesair, Inc. secured USD 22 million in Series A funding for commercializing their Bonhawa Respiratory Humidifier, demonstrating the market's strong interest in advanced respiratory drug delivery solutions. These partnerships have not only accelerated product development but have also improved access to advanced treatments through enhanced distribution networks and shared resources.
Acute Respiratory Distress Syndrome Treatment Market Trends
Increasing Prevalence of Acute Respiratory Distress Syndrome
The rising prevalence of Acute Respiratory Distress Syndrome (ARDS) globally has become a significant concern for healthcare systems, driving the demand for advanced treatment options and specialized care facilities. According to studies published in 2022, approximately 190,000 ARDS cases are reported annually in the United States alone, with the incidence rising significantly with patient age, from 16 per 100,000 person-years for those aged 15-19 to 306 per 100,000 person-years for those aged 75-84. The severity distribution of ARDS cases further emphasizes the critical nature of the condition, with 25% initially classified as mild and 75% as moderate or severe, though notably, a third of mild cases progress to moderate or severe disease. Additionally, approximately 10-15% of patients admitted to intensive care units and up to 23% of mechanically ventilated patients meet the criteria for ARDS, highlighting the substantial burden on healthcare resources.
The chronic nature of ARDS and its impact on patient outcomes has led to increased focus on early intervention and specialized treatment protocols. Recent studies indicate that the ongoing burden of healthcare utilization for ARDS patients remains high, with 40-52% of patients requiring re-hospitalization within one year. This high rate of hospital readmission, coupled with mortality rates ranging from 38% up to 50% in severe cases, has prompted healthcare providers to invest in advanced treatment modalities and specialized care units. The increasing recognition of ARDS as a significant health concern has also led to enhanced research efforts and development of innovative therapeutic approaches, driving market growth for ARDS treatments and management solutions, including mechanical ventilation and oxygen therapy.
High Prevalence of Tobacco Smoking, Urbanization, and Growing Levels of Pollution
The escalating rates of tobacco consumption and environmental pollution have emerged as critical factors contributing to the rising incidence of ARDS globally. According to the Centers for Disease Control and Prevention's 2022 update, nearly 40 million United States adults continue to smoke cigarettes, while 3.08 million middle and high school students use at least one tobacco product, including e-cigarettes. The persistence of high smoking rates, with approximately 1,600 youth younger than 18 years smoking their first cigarette every day, creates a substantial population at risk for developing ARDS. This risk is further compounded by the fact that cigarette smoke exposure is associated with an increased likelihood of developing ARDS in cases of trauma, transfusion, and non-pulmonary sepsis.
The impact of environmental pollution on respiratory health has reached alarming levels, with the World Health Organization reporting that almost all of the global population (99%) is exposed to air pollution levels that put them at increased risk for diseases, including ARDS. The United States Environmental Protection Agency data from 2022 revealed that approximately 67 million tons of pollution were emitted into the atmosphere in the United States alone, with about 102 million people nationwide living in counties with pollution levels above the primary National Ambient Air Quality Standards. The National Institute of Environmental Health Sciences has confirmed that air pollution can significantly affect lung development and is implicated in the development of various respiratory diseases, creating a larger pool of individuals susceptible to ARDS. The use of respiratory monitoring devices is becoming increasingly important in managing these conditions.
Growing Geriatric Population
The global demographic shift towards an aging population has emerged as a significant driver for the ARDS treatment market, as elderly individuals are more susceptible to developing severe respiratory conditions. According to the World Population Prospects 2022 report, there were 771 million people aged 65 years or over globally, representing a threefold increase from the 258 million recorded in 1980. This population is projected to reach 994 million by 2030 and surge to 1.6 billion by 2050, with 80% of older people expected to be living in low- and middle-income countries. The rapid aging of the global population is particularly significant as older individuals are more vulnerable to infections and acquired diseases such as ARDS, creating an expanding patient pool requiring specialized respiratory care.
The acceleration of population aging is creating unprecedented demands on healthcare systems worldwide, with projections indicating that by 2030, one out of every six people will be 60 years of age or older. This demographic transition is particularly noteworthy as it extends beyond traditional high-income countries, with two-thirds of the world's population over 60 expected to live in low- and middle-income countries by 2050. The World Economic Forum's 2022 data further emphasizes this trend, indicating that the share of the elderly population will rise from one billion currently to 1.4 billion by 2030. This significant increase in the elderly population, combined with their higher susceptibility to respiratory conditions, is driving the demand for advanced ARDS treatments and specialized care facilities, including the use of ventilators and pulmonary surfactant therapies.
Segment Analysis: By Treatment
Devices Segment in Acute Respiratory Distress Syndrome Treatment Market
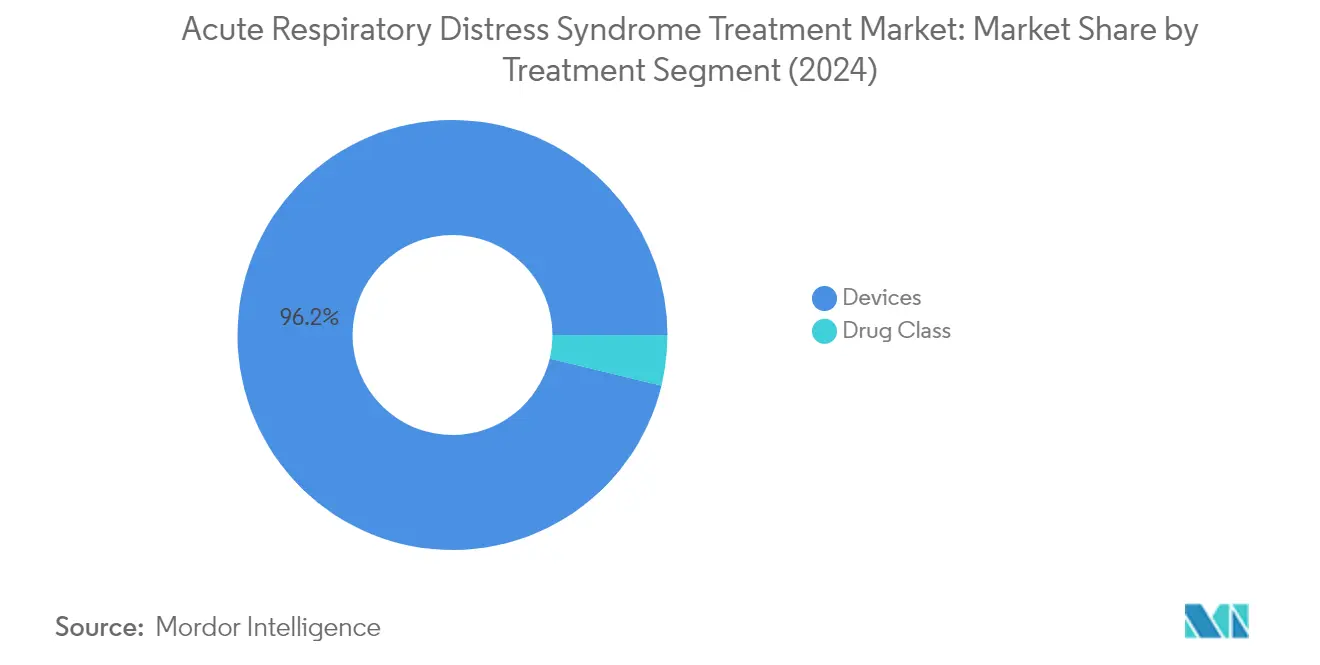
The devices segment dominates the acute respiratory distress syndrome treatment market, commanding approximately 96% of the total market share in 2024. This substantial market share is attributed to the critical role of devices such as ventilators, humidifiers, nebulizers, and oxygen concentrators in managing ARDS patients. The high utility of advanced ventilators, particularly in intensive care units, continues to drive this segment's dominance. The segment's growth is further supported by continuous technological advancements in respiratory care devices, increasing adoption of portable oxygen concentrators, and the rising demand for home care ventilation solutions. Major market players like Hamilton Medical AG, GE Healthcare, and Fisher & Paykel Healthcare Limited are actively contributing to this segment through innovative product launches and strategic expansions in their device portfolios.
Drug Class Segment in Acute Respiratory Distress Syndrome Treatment Market
The drug class segment is emerging as the fastest-growing segment in the ARDS treatment market, projected to grow at approximately 8% during the forecast period 2024-2029. This accelerated growth is driven by increasing research and development activities in novel therapeutic approaches, including vasodilators, bronchodilators, steroids, antibiotics, and surfactants. The segment's growth is further bolstered by rising investments in clinical trials for new drug development, particularly in areas such as extracellular vesicles and cell therapies. Recent developments, such as Direct Biologics' ExoFlo receiving FDA approval for phase III clinical trials and Windtree Therapeutics' positive results for lucinactant, demonstrate the segment's dynamic evolution. The increasing focus on personalized medicine approaches and the development of targeted therapies for ARDS are expected to maintain this segment's robust growth trajectory.
Segment Analysis: By End User
Hospitals and Clinics Segment in Acute Respiratory Distress Syndrome Treatment Market
The hospitals and clinics segment dominates the acute respiratory distress syndrome (ARDS) treatment market, commanding approximately 86% market share in 2024. This substantial market share is attributed to the critical nature of ARDS treatment, which typically requires intensive care unit (ICU) facilities and advanced medical equipment available primarily in hospital settings. The segment's dominance is further strengthened by the increasing adoption of sophisticated ventilation technologies, the presence of skilled healthcare professionals, and the rising number of ICU beds in hospitals worldwide. Additionally, the growing preference for hospital-based treatment for severe respiratory conditions, coupled with the availability of reimbursement policies for hospital-based treatments, continues to drive this segment's growth. The segment's strong position is also supported by hospitals' ability to invest in advanced critical care equipment and their capacity to handle complex ARDS cases requiring multidisciplinary medical intervention.
Other End Users Segment in Acute Respiratory Distress Syndrome Treatment Market
The other end users segment, which includes ambulatory surgical centers, home care settings, and long-term care centers, plays a crucial role in providing continued care and support for ARDS patients. This segment is experiencing steady growth as healthcare systems increasingly emphasize post-hospital care and rehabilitation services for ARDS survivors. The segment's development is driven by the rising demand for long-term respiratory care facilities, the growing trend of home-based medical care, and the increasing availability of portable respiratory care devices suitable for use in non-hospital settings. Furthermore, the expansion of ambulatory care facilities and their improving capabilities in handling respiratory care cases contributes to the segment's evolution. The integration of telemedicine services and remote patient monitoring systems has also enhanced the ability of these facilities to provide effective care while maintaining close coordination with primary healthcare providers.
Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market Geography Segment Analysis
Acute Respiratory Distress Syndrome Treatment Market in North America
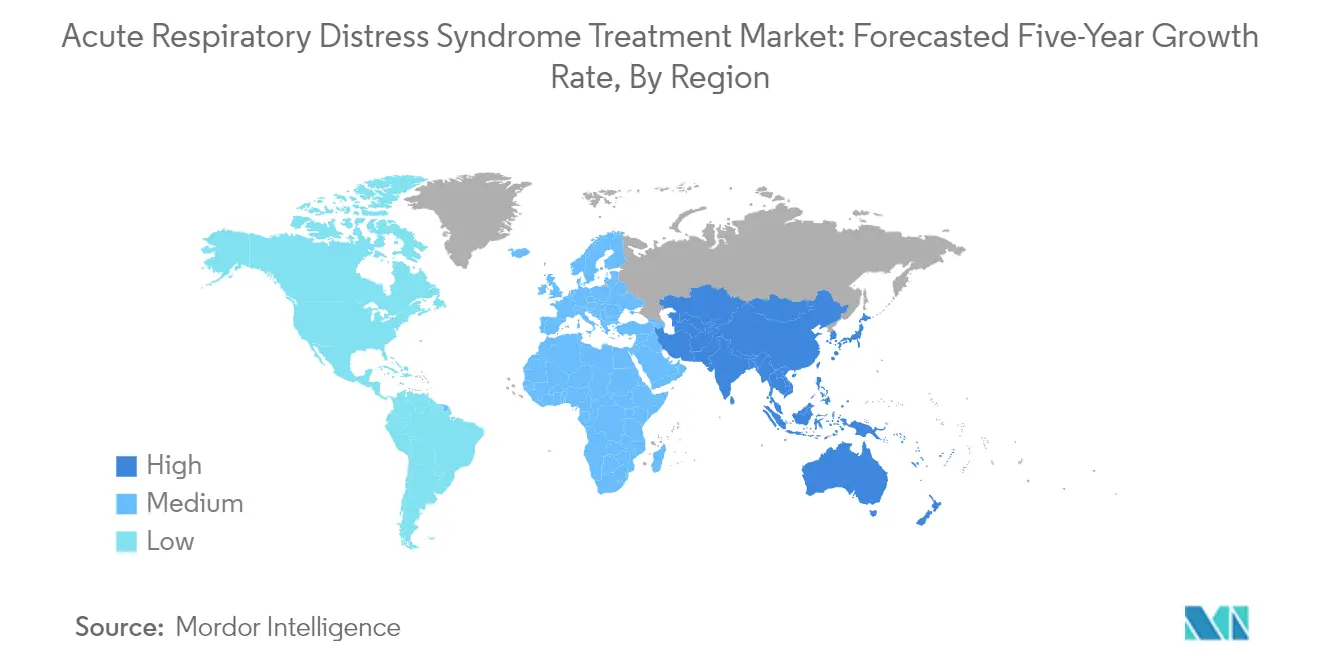
The North American acute respiratory distress syndrome treatment market demonstrates robust growth driven by advanced healthcare infrastructure and significant research initiatives. The region benefits from the presence of major market players, established healthcare systems, and substantial investment in medical research and development. The United States, Canada, and Mexico form the key markets in this region, with each country contributing uniquely to the market's dynamics through different healthcare policies and treatment approaches. Additionally, the region's ventilator market plays a crucial role in supporting ARDS treatment advancements.
Acute Respiratory Distress Syndrome Treatment Market in United States
The United States dominates the North American ARDS treatment market, holding approximately 92% of the regional market share. This leadership position is attributed to the country's advanced healthcare infrastructure, high healthcare spending, and presence of major medical device manufacturers. The nation's robust research and development facilities have led to the development of innovative treatments and devices. The high burden of ARDS, coupled with the presence of well-equipped intensive care units and specialized medical professionals, further strengthens the market position of the United States in the region. The mechanical ventilation market is integral to the treatment strategies employed across the country.
Acute Respiratory Distress Syndrome Treatment Market in Canada
Canada emerges as the fastest-growing market in North America, with a projected growth rate of approximately 7% during 2024-2029. The country's growth is driven by increasing research initiatives, rising healthcare expenditure, and growing awareness about ARDS treatment options. The Canadian healthcare system's focus on improving patient outcomes through advanced medical technologies and treatments has created a favorable environment for market expansion. The country's strategic initiatives in healthcare innovation and commitment to providing accessible medical care continue to drive market growth. The development of the oxygen therapy market is pivotal in enhancing treatment outcomes in Canada.
Acute Respiratory Distress Syndrome Treatment Market in Europe
The European ARDS treatment market showcases significant development across its diverse healthcare landscapes. The region's market is characterized by strong healthcare infrastructure, substantial research activities, and innovative medical technologies. Germany, the United Kingdom, France, Italy, and Spain represent the key markets, each contributing distinctively to the regional market dynamics through their unique healthcare systems and treatment approaches. The critical care equipment market in Europe is a significant contributor to these advancements.
Acute Respiratory Distress Syndrome Treatment Market in Germany
Germany maintains its position as the largest market for ARDS treatment in Europe, commanding approximately 23% of the regional market share. The country's leadership is supported by its advanced medical technology sector, robust healthcare infrastructure, and significant presence of medical device manufacturers. Germany's commitment to healthcare innovation and substantial investment in medical research continues to strengthen its position in the European market. The respiratory care device market in Germany is instrumental in supporting these developments.
Acute Respiratory Distress Syndrome Treatment Market in France
France demonstrates remarkable growth potential in the European market, with an expected growth rate of approximately 8% during 2024-2029. The country's progressive healthcare policies, increasing research initiatives, and growing focus on innovative treatment solutions drive this growth. France's commitment to advancing medical technologies and improving patient care through innovative treatments has established it as a key growth market in the region. The oxygen therapy market is a vital component of France's healthcare advancements.
Acute Respiratory Distress Syndrome Treatment Market in Asia-Pacific
The Asia-Pacific ARDS treatment market exhibits dynamic growth patterns influenced by diverse healthcare systems and increasing healthcare investments. The region encompasses major markets including China, Japan, India, Australia, and South Korea, each contributing to the market's expansion through different healthcare approaches and treatment methodologies. The market benefits from rapid healthcare infrastructure development and increasing awareness about advanced treatment options. The ventilator market is a key area of focus in the region's healthcare advancements.
Acute Respiratory Distress Syndrome Treatment Market in Japan
Japan leads the Asia-Pacific ARDS treatment market, supported by its advanced healthcare infrastructure and significant technological innovations in medical devices. The country's leadership position is strengthened by its aging population, which drives the demand for advanced respiratory care solutions. Japan's focus on healthcare technology development and strong presence of medical device manufacturers continues to reinforce its market position. The mechanical ventilation market is crucial in addressing the needs of Japan's healthcare system.
Acute Respiratory Distress Syndrome Treatment Market in India
India emerges as the fastest-growing market in the Asia-Pacific region, driven by improving healthcare infrastructure and increasing accessibility to advanced medical treatments. The country's healthcare sector demonstrates significant potential for growth through expanding hospital infrastructure, rising healthcare spending, and growing awareness about ARDS treatment options. India's focus on developing affordable healthcare solutions and increasing investment in medical technology contributes to its rapid market expansion. The critical care equipment market is pivotal in supporting India's healthcare advancements.
Acute Respiratory Distress Syndrome Treatment Market in Middle East & Africa
The Middle East & Africa ARDS treatment market demonstrates evolving growth patterns influenced by improving healthcare infrastructure and increasing investment in medical technologies. The region's market dynamics are shaped by varying healthcare systems across different countries, with GCC countries leading in terms of market size and South Africa showing the fastest growth. The market benefits from increasing healthcare spending, growing awareness about respiratory diseases, and improving access to advanced medical treatments across the region.
Acute Respiratory Distress Syndrome Treatment Market in South America
The South American ARDS treatment market shows promising development driven by improving healthcare infrastructure and increasing adoption of advanced medical technologies. Brazil emerges as the largest market in the region, while Argentina demonstrates the fastest growth rate. The regional market is characterized by growing healthcare expenditure, increasing awareness about respiratory diseases, and rising investment in medical technology. The market continues to evolve with improving access to healthcare services and growing adoption of advanced treatment options.
Acute Respiratory Distress Syndrome Treatment Industry Overview
Top Companies in Acute Respiratory Distress Syndrome Treatment Market
The competitive landscape of the ARDS treatment market is characterized by the presence of established medical device manufacturers and pharmaceutical companies pursuing innovative therapeutic solutions. Companies are heavily investing in research and development to advance their product pipelines, particularly focusing on novel drug development and enhanced respiratory care devices systems. Strategic collaborations between pharmaceutical companies and research institutions have become increasingly common to accelerate drug development and clinical trials. Market leaders are expanding their geographical presence through distribution partnerships and regional manufacturing facilities, while simultaneously focusing on technological advancements in ventilation systems and respiratory monitoring devices technologies. The industry has witnessed a significant surge in patent applications and fast-track designations for ARDS treatments, indicating intense competition in therapeutic innovation.
Diverse Players Shape Dynamic Market Structure
The ARDS treatment market exhibits a balanced mix of global medical device conglomerates and specialized pharmaceutical companies, creating a moderately consolidated competitive environment. Large multinational corporations like GE Healthcare, Abbott, and Drägerwerk dominate the medical devices segment with their comprehensive product portfolios and established distribution networks. These companies leverage their extensive research capabilities and global presence to maintain market leadership. Meanwhile, specialized pharmaceutical companies and biotechnology firms focus on developing targeted respiratory therapeutic solutions, creating a dynamic competitive landscape.
The market has witnessed strategic consolidation through mergers and acquisitions, particularly in the medical devices segment, as companies seek to enhance their technological capabilities and expand their geographic reach. Partnerships between device manufacturers and healthcare providers have become increasingly important for product development and market penetration. The emergence of innovative startups and research-focused companies has introduced new competitive dynamics, particularly in the development of novel therapeutic approaches and advanced ventilator industry technologies.
Innovation and Adaptability Drive Market Success
Success in the ARDS treatment market increasingly depends on companies' ability to develop innovative solutions while maintaining operational efficiency. Incumbent players must focus on continuous product innovation, particularly in developing more efficient and user-friendly ventilator market devices. Companies need to establish strong relationships with healthcare providers and research institutions to ensure product adoption and gather clinical evidence. Additionally, maintaining regulatory compliance while accelerating product development timelines has become crucial for maintaining market position.
For emerging players and contenders, differentiation through specialized therapeutic approaches and targeted solutions offers opportunities for market entry. Companies must consider the concentrated nature of hospital procurement systems and develop strategies to overcome high entry barriers in the medical device segment. The moderate threat of substitution necessitates continuous innovation in treatment approaches and device functionality. Regulatory requirements, particularly for novel therapeutic solutions, significantly influence market dynamics and require companies to maintain robust compliance frameworks while pursuing innovation.
Acute Respiratory Distress Syndrome Treatment Market Leaders
-
HEALIOS K.K
-
Faron Pharmaceuticals
-
BioMarck Pharmaceuticals
-
Athersys
-
Apeptico Forschung
- *Disclaimer: Major Players sorted in no particular order
Acute Respiratory Distress Syndrome Treatment Market News
- In April 2022, the Food and Drug Administration (FDA) approved Direct Biologics to proceed with a Landmark Phase 3 clinical trial using its investigation EV Drug ExoFlow to treat Acute Respiratory Distress Syndrome (ARDS).
- In January 2022, Veru declared that Sabizabulin has been given a fast-track designation by the FDA for the treatment of hospitalized COVID-19 patients who are at high risk of developing acute respiratory distress syndrome.
Acute Respiratory Distress Syndrome Treatment Industry Segmentation
Acute respiratory distress syndrome (ARDS) is a rapidly progressive disease occurring in critically ill patients. The main complication in ARDS is that fluid leaks into the lungs, making breathing difficult or impossible. The causes of ARDS are divided into two categories: direct or indirect injuries to the lung. Some of the direct injuries to the lung include pneumonia, aspiration, trauma, and others whereas the indirect injuries to the lung include inflammation of the pancreas, severe infection (also known as sepsis), blood transfusions, burns, and medication reactions. Acute Respiratory Distress Syndrome (ARDS) Treatment Market is Segmented by Treatment (Drug Class, Devices), End User (Hospitals/Clinics, Other End Users), and Geography (North America, Europe, Asia-Pacific, Middle East, and Africa, South America). The market report also covers the estimated market sizes and trends for 17 different countries across major regions globally.The report offers the value (in USD million) for the above segments.
| By Treatment | By Drug Class | Vasoconstrictor | |
| Bronchodilators | |||
| Streoid and Antibiotics | |||
| Sedative and Paralytic | |||
| Surfactant | |||
| Other | |||
| Devices | |||
| By End User | Hospitals/Clinics | ||
| Other End Users | |||
| Geography | North America | United States | |
| Canada | |||
| Mexico | |||
| Europe | Germany | ||
| United Kingdom | |||
| France | |||
| Italy | |||
| Spain | |||
| Rest of Europe | |||
| Asia-Pacific | China | ||
| Japan | |||
| India | |||
| Australia | |||
| South Korea | |||
| Rest of Asia-Pacific | |||
| Middle East and Africa | GCC | ||
| South Africa | |||
| Rest of Middle East and Africa | |||
| South America | Brazil | ||
| Argentina | |||
| Rest of South America | |||
| By Drug Class | Vasoconstrictor |
| Bronchodilators | |
| Streoid and Antibiotics | |
| Sedative and Paralytic | |
| Surfactant | |
| Other | |
| Devices |
| Hospitals/Clinics |
| Other End Users |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
Acute Respiratory Distress Syndrome Treatment Market Research FAQs
How big is the Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market?
The Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market size is expected to reach USD 3.44 billion in 2025 and grow at a CAGR of 5.65% to reach USD 4.53 billion by 2030.
What is the current Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market size?
In 2025, the Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market size is expected to reach USD 3.44 billion.
Who are the key players in Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market?
HEALIOS K.K, Faron Pharmaceuticals, BioMarck Pharmaceuticals, Athersys and Apeptico Forschung are the major companies operating in the Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market.
Which is the fastest growing region in Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market?
North America is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market?
In 2025, the Asia-Pacific accounts for the largest market share in Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market.
What years does this Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market cover, and what was the market size in 2024?
In 2024, the Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market size was estimated at USD 3.25 billion. The report covers the Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Global Acute Respiratory Distress Syndrome (ARDS) Treatment Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on: June 16, 2024