Upper Limb Prosthetics Market Size and Share
Upper Limb Prosthetics Market Analysis by Mordor Intelligence
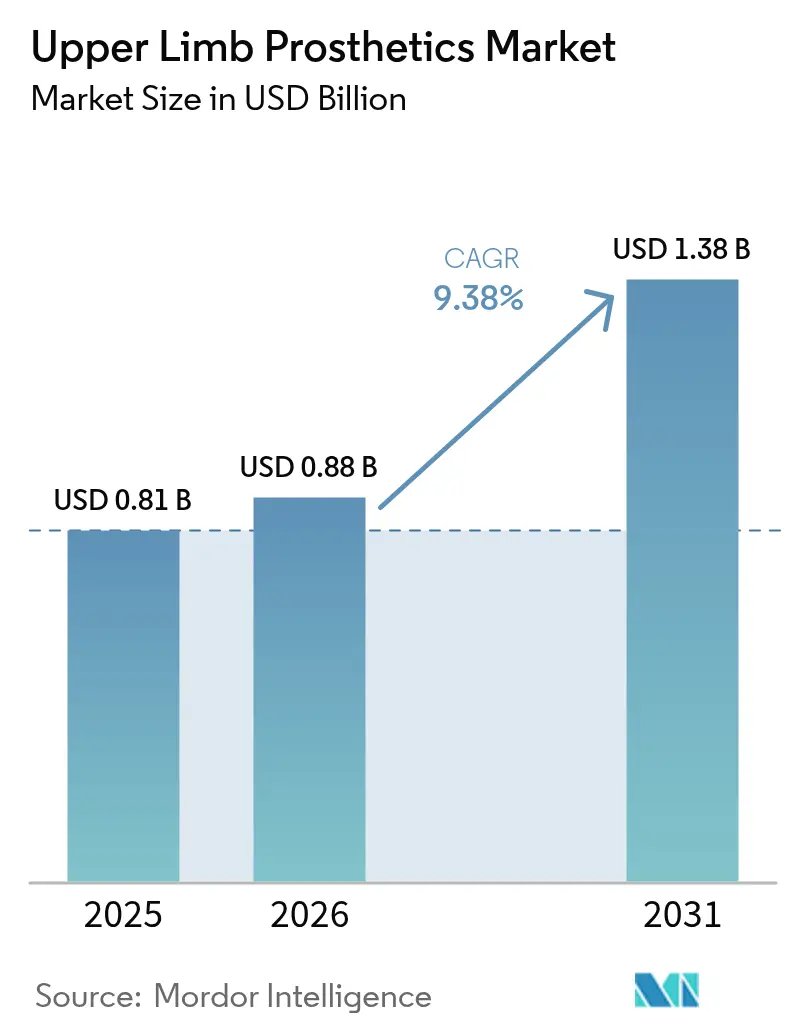
The Upper Limb Prosthetics Market size was valued at USD 0.81 billion in 2025 and is estimated to grow from USD 0.88 billion in 2026 to reach USD 1.38 billion by 2031, at a CAGR of 9.38% during the forecast period (2026-2031).
Reimbursement reform in the United States, Europe, and Japan is widening access to powered devices, while defence-funded research in osseointegration and neural interfaces is migrating into civilian clinics. Multi-articulated hands equipped with pattern-recognition control and vibrotactile feedback are replacing passive cosmetic limbs, reducing training time and improving task accuracy. Ageing populations in OECD countries continue to push vascular and diabetic amputation volumes upward, amplifying long-term demand. At the same time, additive manufacturing is compressing production lead times for paediatric bionic arms from eight weeks to ten days, bringing entry-level myoelectric solutions below the USD 10,000 price threshold.
Key Report Takeaways
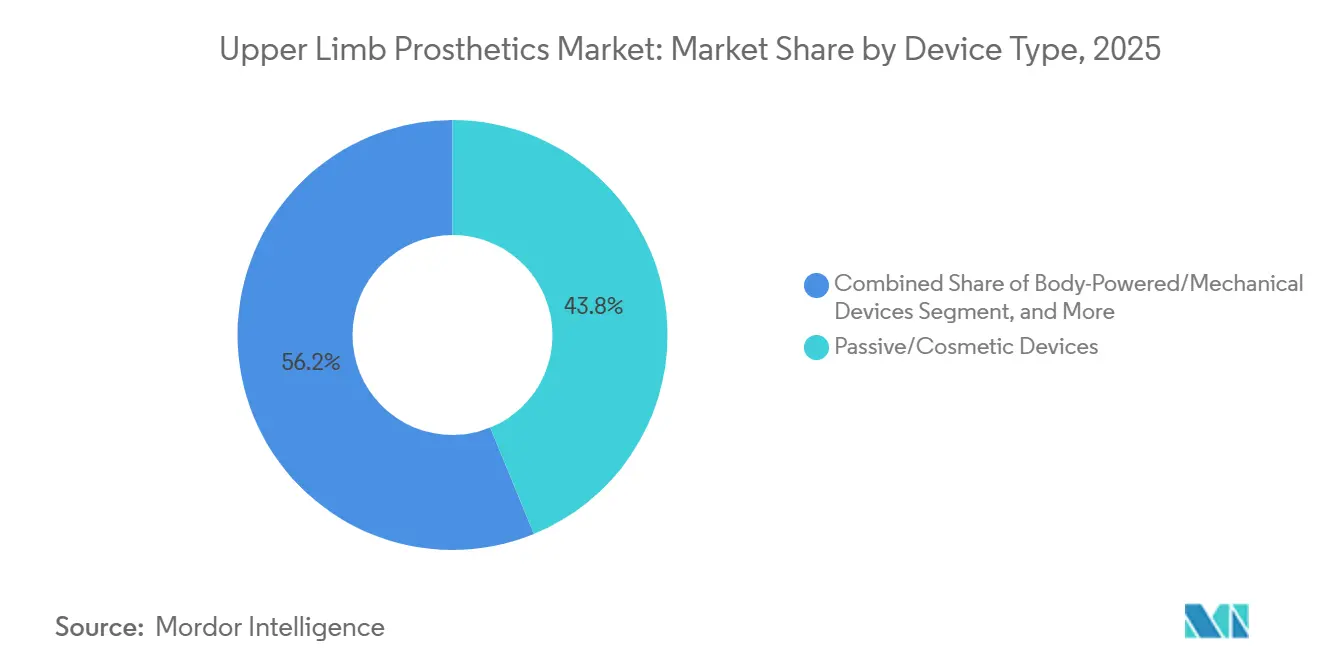
- By product type, passive and cosmetic devices accounted for 43.82% of the upper-limb prosthetics market share in 2025. Myoelectric and powered devices are projected to advance at a 10.06% CAGR between 2026 and 2031.
- By component, hand and other terminal devices accounted for 34.27% of the upper limb prosthetics market in 2025. Prosthetic elbows are set to register the fastest growth, expanding at an 11.63% CAGR through 2031.
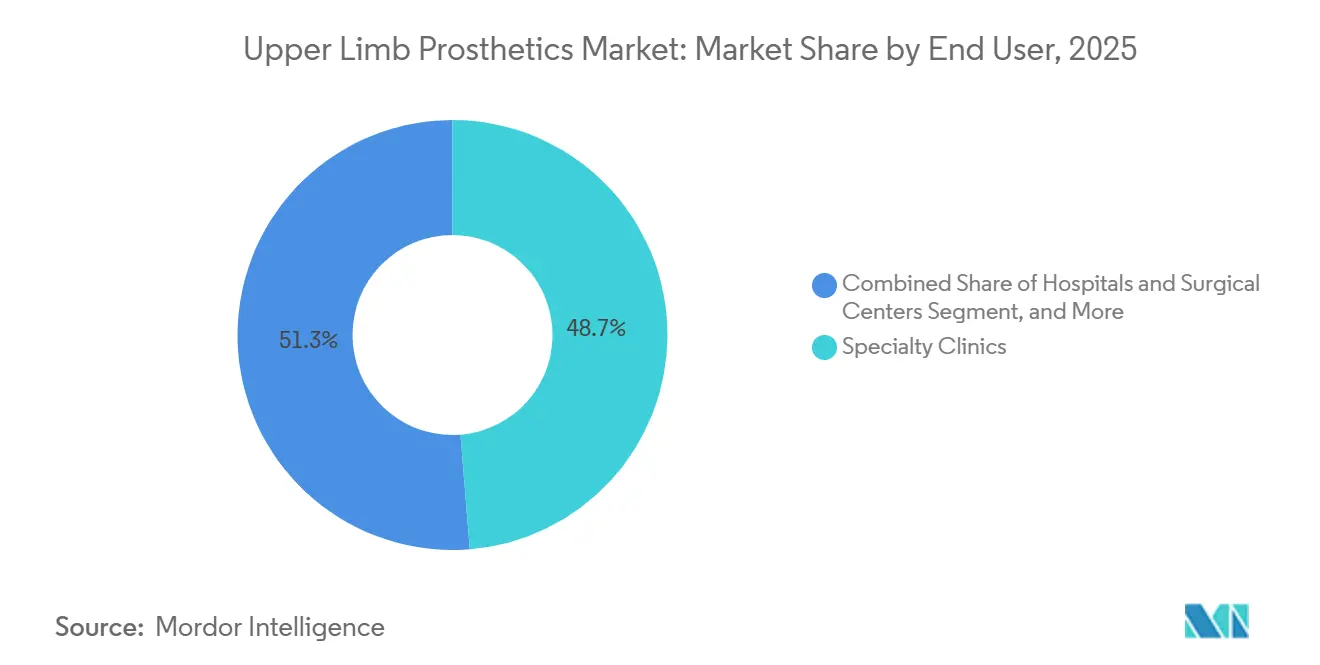
- By end user, specialty clinics accounted for 48.72% of end-user revenue in 2025. Rehabilitation centers are expected to grow at a 9.92% CAGR over 2026-2031.
- By geography, North America led with 43.18% of global revenue in 2025. Asia-Pacific is forecast to post the fastest regional expansion, rising at a 12.71% CAGR to 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Upper Limb Prosthetics Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Ageing-population-driven vascular & diabetic amputations surge | +2.1% | Global, concentrated in North America, Europe, Japan | Long term (≥ 4 years) |
| Emergence of AI-based sensory feedback systems enhancing user acceptance | +1.8% | North America, Europe, Asia-Pacific urban centers | Medium term (2-4 years) |
| Rapid advances in myoelectric control algorithms & multi-articulated hands | +1.5% | Global, led by North America & Europe R&D hubs | Medium term (2-4 years) |
| Wider reimbursement expansion in veteran & worker-comp programs | +1.3% | North America, Europe, Australia | Short term (≤ 2 years) |
| 3D-printed, low-cost paediatric bionic arms addressing unmet needs | +0.9% | Global, with early adoption in UK, Middle East, Southeast Asia | Medium term (2-4 years) |
| Defence-funded osseointegration R&D crossing over to civilian clinics | +0.7% | North America, Europe, Australia | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Ageing-Population-Driven Vascular & Diabetic Amputations Surge
The global population aged 65 and older reached 761 million in 2024 and continues to expand at 3.1% annually, pushing demand for upper-limb devices in regions where chronic disease intersects with advanced surgical capacity.[1]United Nations Department of Economic and Social Affairs, “World Population Ageing 2024,” UN.ORG Vascular complications from uncontrolled diabetes account for roughly 54% of non-traumatic upper-limb amputations across OECD economies, and diabetic patients face a 15-fold higher amputation risk than non-diabetic cohorts. Japan now reimburses myoelectric prostheses for individuals aged 70 or older, reflecting a policy shift that took effect in April 2025. A longer post-amputation life expectancy means patients routinely require multiple socket replacements, battery upgrades, and software updates over 15-20 years of device use. Consequently, the driver represents a structural, not cyclical, reallocation of healthcare budgets toward durable medical equipment that safeguards independence and lowers long-term care spending.
Emergence of AI-Based Sensory Feedback Systems Enhancing User Acceptance
High-density electromyography paired with machine-learning algorithms now enables prosthetic hands to recognize eight or more grip patterns from only two electrode sites, cutting cognitive load and shortening training periods from 18 weeks to four.[2]Coapt LLC, “Complete Control System Technical Documentation,” COAPTENGINEERING.COMSensory feedback delivered via vibrotactile actuators reduces object-drop rates by 41% during daily living tasks. Commercial roll-outs such as the TASKA Hand integrate fingertip force sensors that dynamically modulate motor torque, allowing delicate operations like food preparation without manual mode changes. By converting a traditionally open-loop device into a bidirectional human-machine interface, AI-enabled systems are accelerating user adoption and decreasing abandonment rates.
Rapid Advances in Myoelectric Control Algorithms & Multi-Articulated Hands
Commercial hands now deliver 14 selectable grip patterns with proportional speed control, while targeted muscle reinnervation surgery creates new EMG sites that support simultaneous shoulder, elbow, and hand movement. The LUKE Arm, cleared for expanded Medicare coverage in 2024, offers ten powered joints and foot-pedal adjuncts for bilateral amputees. Although lithium-polymer packs limit continuous use to 8-12 hours, next-generation solid-state batteries promise 30% higher energy density by 2028, extending charge cycles closer to full-day operation. Hardware miniaturization, surgical innovation, and algorithm refinement are jointly shrinking the performance gap between biological and prosthetic limbs.
Wider Reimbursement Expansion in Veteran & Worker-Comp Programs
The U.S. Department of Veterans Affairs removed prior authorization for myoelectric and osseointegrated systems in January 2025, cutting lead times by up to six months. TRICARE lifted its lifetime cap for upper-limb prosthetics to USD 125,000, while Medicare introduced HCPCS code L6026 with an allowable charge of USD 18,500 for pattern-recognition controllers.[3]Centers for Medicare & Medicaid Services, “HCPCS Code Updates and Prosthetic Coverage,” CMS.GOV Early provision of advanced devices has been shown to reduce long-term disability payments by 23% through faster return-to-work timelines. Similar expansions in California, Texas, and New York workers’ compensation schemes reinforce the view that high-function systems yield downstream economic benefits.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High acquisition & lifetime maintenance cost of powered prostheses | -1.4% | Global, acute in emerging markets and rural areas | Medium term (2-4 years) |
| Shortage of skilled prosthetists for complex upper-limb fittings | -0.9% | Global, severe in Asia-Pacific, Middle East, Africa | Long term (≥ 4 years) |
| Battery-life & durability gaps in multi-DOF devices | -0.7% | Global, impacting industrial and agricultural users | Short term (≤ 2 years) |
| Fragmented national regulatory pathways slowing cross-border launches | -0.6% | Global, particularly EU-Asia regulatory divergence | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
High Acquisition & Lifetime Maintenance Cost of Powered Prostheses
Entry-level myoelectric hands start at USD 20,000, while multi-articulated systems with closed-loop feedback exceed USD 120,000, pricing out 68% of global amputees who live in low- or middle-income countries where annual per-capita health spending falls below USD 500. Ownership costs escalate with socket replacements every 3-5 years, battery swaps every two years, and software updates that command USD 500-1,000 annually. Even in the United States, average Medicare beneficiaries face USD 6,200 in out-of-pocket expenses for an advanced device despite coverage. Although modular 3D-printed designs reduce acquisition costs for paediatric users, progress remains incremental, constraining penetration in price-sensitive markets.
Shortage of Skilled Prosthetists for Complex Upper-Limb Fittings
Only 4,200 certified prosthetists practice in the United States, equating to one per 79,000 residents, and fewer than one-third are trained in advanced myoelectric fittings. Rural patients often travel 500 miles for device tuning, delaying functional rehabilitation by three to six months. Internationally, the gap widens: sub-Saharan Africa averages one prosthetist per two million people, and Southeast Asia reports one per 800,000 [WHO.INT]. Tele-fitting pilots show promise but face licensure barriers that limit interstate or cross-border practice. Until education pipelines and regulatory frameworks evolve, the clinician shortfall will continue to curb device adoption, especially for transhumoral and shoulder-disarticulation cases that demand high-skill installations.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Product Type: Passive Devices Anchor Share, Powered Systems Drive Growth
Passive and cosmetic limbs dominated the upper-limb prosthetics market, accounting for 43.82% in 2025, supported by price points of USD 3,000-8,000 that meet the needs of appearance-focused users. Body-powered solutions held roughly 28% of unit volume, appealing to industrial laborers who value mechanical endurance in harsh settings. Powered myoelectric devices are projected to post a 10.06% CAGR over 2026-2031, fueled by reimbursement gains and AI-enabled control that lowers training barriers. Hybrid TMR-enabled systems remain niche but deliver simultaneous multi-joint control, redefining standard-of-care protocols in academic centers.
The bifurcated landscape sends high-volume, low-margin demand toward passive devices in emerging economies, while high-value growth concentrates in powered systems across insured markets. Paediatric programs increasingly adopt low-cost printed bionics until skeletal maturity, after which users upgrade to multi-articulated hands. Meanwhile, the durability of body-powered gear is securing a loyal base among agriculture and construction workers, even as sensor-equipped hands begin to encroach on that space.

By Component: Terminal Devices Lead, Elbows Accelerate
Hand and other terminal units captured 34.27% of 2025 revenue, reflecting universal need across amputation levels and concentrated R&D in grip diversity and cosmetic realism. Elbow mechanisms, once limited to single-axis hinges, are forecast to climb at an 11.63% CAGR thanks to multi-DOF joints that enable overhead reach and reduced compensatory shoulder motion.
Investment is tilting toward elbows because functional outcomes for transhumeral amputees historically lag behind those of transradial cases. Devices such as the DynamicArm introduce proportional speed control and automatic damping that cut contralateral shoulder pain by one-third. Simultaneously, miniaturized gearboxes have shrunk wrist diameters to 50 mm, making powered rotation viable for small adults and older children. Realistic silicone covers for terminal devices continue to address psychosocial factors that influence overall device acceptance.
By End User: Specialty Clinics Dominate, Rehabilitation Centers Gain Ground
Specialty clinics accounted for 48.72% of 2025 revenue, leveraging deep expertise in socket fabrication and EMG electrode placement to successfully fit complex cases. Rehabilitation centers are on track for a 9.92% CAGR as payers adopt bundled-care models that tie reimbursement to functional milestones rather than device delivery volume. Hospitals contribute about 28% of revenue, mostly in immediate post-amputation stabilization before referring patients to outpatient facilities.
The trend signals convergence: specialty clinics are embedding physical therapists, and rehabilitation centers are hiring prosthetists to deliver turnkey care models. Bundled payments encourage multidisciplinary coordination, rewarding providers who can demonstrate improved return-to-work rates and reduced secondary injuries. Over time, this integration is expected to shift share gradually toward comprehensive platforms capable of managing the entire patient journey.

Geography Analysis
North America led the upper-limb prosthetics market, accounting for 43.18% of global revenue in 2025. Canada’s single-payer provinces reimburse myoelectric solutions for traumatic amputees but maintain stricter functional criteria for vascular cases, creating regional disparities. Mexico relies on small workshops producing passive limbs at USD 500-1,200, though a federal program launched in 2024 aims to open 12 myoelectric centers by 2028.
Germany, the United Kingdom, and France accounted for 68% of that figure, aided by statutory insurance that reimburses up to EUR 80,000 (USD 87,000) per device. The region’s transition to the Medical Device Regulation initially slowed launches but is now smoothing cross-border approvals, lowering compliance costs for multinationals.
Asia-Pacific is expected to deliver the fastest growth, expanding at a 12.71% CAGR through 2031. Japan’s super-aged society has prompted insurance to cover powered devices for seniors over 70. China’s local manufacturers are scaling up output with facilities capable of producing 8,000 units per year, targeting price points 40% below those of Western analogues. India faces affordability constraints, yet it distributed 1,200 printed limbs in 2025 under a national disabilities program. Australia’s NDIS funds up to AUD 150,000 (USD 98,000) per prosthesis, making the country a per-capita leader in powered adoption.
Markets in the Middle East and Africa remain underpenetrated; GCC nations import high-end devices for their citizens, while migrant laborers rely on charity-funded prosthetics. South America is concentrated in Brazil and Argentina, where partial reimbursement leaves out-of-pocket gaps of USD 8,000-15,000 for powered systems, limiting uptake to higher-income groups.

Competitive Landscape
The upper-limb prosthetics industry is characterized by moderate concentration. Incumbents leverage vertical integration and deep payer relationships, while newcomers such as Open Bionics, COVVI, and TASKA differentiate through modular 3D-printed designs that drop paediatric device costs below USD 10,000.
Technology forms the chief battleground. USPTO filings for 2024-2025 include 47 patents on osseointegrated electrode arrays and 62 on vibrotactile feedback, underscoring industry-wide migration toward closed-loop control. AI-driven algorithms like Coapt’s Complete Control raise task-completion accuracy to 92% within four weeks of training, a leap that shortens rehabilitation cycles. Direct-to-consumer subscription models are emerging: Open Bionics offers USD 150 monthly service that covers socket changes and software updates, unbundling acquisition from lifetime upkeep.
Private equity is consolidating specialty clinics to build national networks capable of negotiating volume rebates and winning bundled-care contracts. Hanger Clinic’s 2025 acquisition of 14 practices raised its footprint to 850 U.S. locations, positioning it for multi-year Veterans Affairs tenders. Automotive and electronics suppliers are eyeing entry via partnerships, drawn by the cross-applicability of their battery and sensor expertise. As cost discipline improves, margins on entry-level powered systems are likely to compress, intensifying competition.
Upper Limb Prosthetics Industry Leaders
Össur
Fillauer LLC.
Steeper Inc.
Ottobock SE & Co. KgaA
Ortho Europe
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- April 2025: Phantom Neuro secured USD 19 million Series A led by Ottobock to advance neural interfaces for upper limb control.
- April 2025: CMS introduced HCPCS codes L6700 and L6032, widening Medicare coverage for pattern-recognition modules and ultralight materials.
- March 2025: Johns Hopkins University revealed a hybrid bionic hand combining soft and rigid structures with real-time sensory feedback.
- April 2024: Ottobock received grant funding to develop rotation hydraulic joints.
Global Upper Limb Prosthetics Market Report Scope
As defined in the scope of the report, a prosthesis is an artificial device that substitutes for a missing body part. Upper limb prosthetics can be applied from the shoulder joint to the fingers, including the hand, wrist, forearm, elbow, upper arm, and shoulder.
The Upper Limb Prosthetics Market Report is Segmented by Product Type (Passive/Cosmetic, Body-Powered/Mechanical, Myoelectric/Powered, Hybrid & TMR-Enabled), Component (Hand/Terminal Device, Wrist, Elbow, Shoulder & Upper Arm), End User (Specialty Clinics, Hospitals & Surgical Centers, Rehabilitation Centers), and Geography (North America, Europe, Asia-Pacific, Middle East & Africa, South America). The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD million) for the above segments.
| Passive / Cosmetic Devices |
| Body-Powered / Mechanical Devices |
| Myoelectric / Powered Devices |
| Hybrid & TMR-Enabled Devices |
| Prosthetic Hand / Terminal Device |
| Prosthetic Wrist |
| Prosthetic Elbow |
| Prosthetic Shoulder & Upper Arm |
| Specialty Clinics |
| Hospitals & Surgical Centers |
| Rehabilitation Centers |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East & Africa | GCC |
| South Africa | |
| Rest of Middle East & Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product Type | Passive / Cosmetic Devices | |
| Body-Powered / Mechanical Devices | ||
| Myoelectric / Powered Devices | ||
| Hybrid & TMR-Enabled Devices | ||
| By Component | Prosthetic Hand / Terminal Device | |
| Prosthetic Wrist | ||
| Prosthetic Elbow | ||
| Prosthetic Shoulder & Upper Arm | ||
| By End User | Specialty Clinics | |
| Hospitals & Surgical Centers | ||
| Rehabilitation Centers | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East & Africa | GCC | |
| South Africa | ||
| Rest of Middle East & Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
How large will the upper limb prosthetics market be by 2031?
It is projected to reach USD 1.38 billion by 2031, reflecting a 9.38% CAGR over 2026-2031.
Which device type is expanding fastest?
Powered myoelectric systems are forecast to grow at 10.06% CAGR, the highest among all product types.
What region shows the strongest growth outlook?
Asia-Pacific is expected to post the quickest expansion at a 12.71% CAGR through 2031.
Which component segment is gaining traction most rapidly?
Prosthetic elbows, driven by multi-DOF mechanisms, are projected to advance at an 11.63% CAGR.
Why are specialty clinics so dominant in device fittings?
They hold 48.72% of 2025 revenue due to their specialized expertise in socket design and EMG calibration, which are essential for advanced myoelectric fittings.
Page last updated on:









