Non-Invasive Prenatal Testing Market Size and Share
Non-Invasive Prenatal Testing Market Analysis by Mordor Intelligence
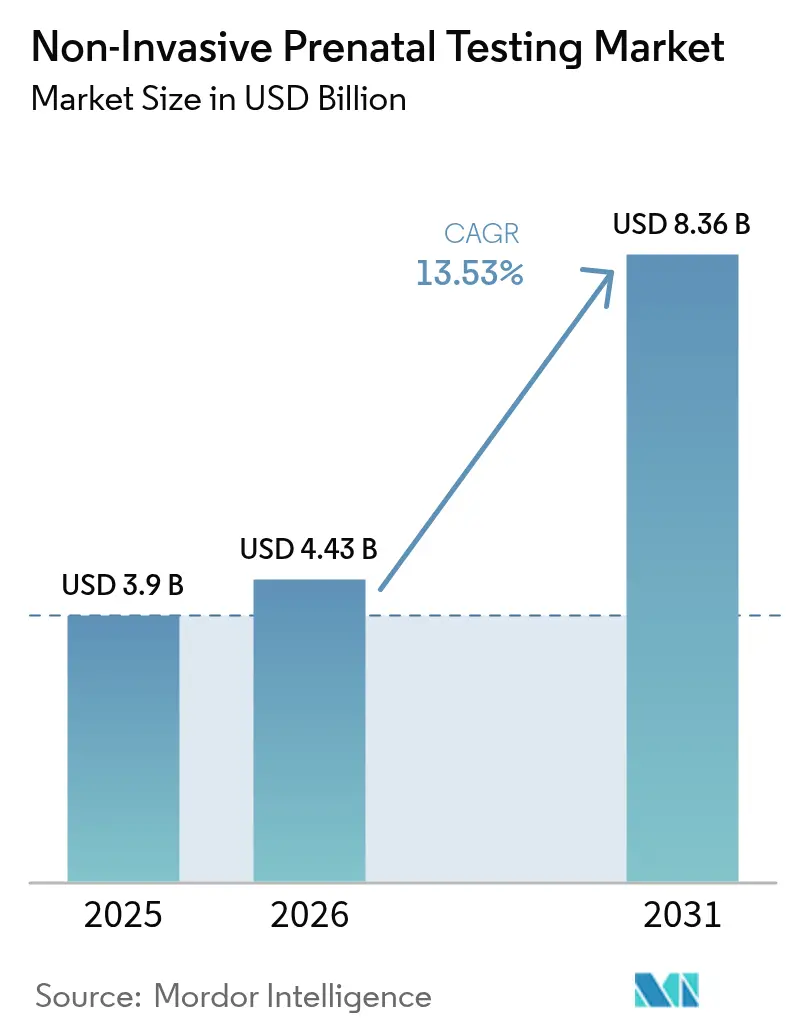
The non-invasive prenatal testing market size is projected to expand from USD 3.9 billion in 2025 and USD 4.43 billion in 2026 to USD 8.36 billion by 2031, registering a 13.53% CAGR over 2026-2031. Sequencing costs falling below USD 200 per sample, universal guideline support, and payer policies that now cover average-risk pregnancies are converting NIPT from a specialist screen into a first-line test in obstetric care. Rising maternal age—births to women aged 35-44 climbed 3.2% in 2024—elevates aneuploidy risk and broadens the eligible population. Technology is also shifting: isothermal rolling-circle amplification (RCA) workflows that avoid thermal cyclers are growing rapidly, lowering capital barriers for smaller laboratories. Competitive dynamics are intensifying as BGI Genomics undercuts Western pricing in Asia-Pacific while Labcorp’s 2024 acquisition of Invitae consolidates U.S. capacity.
Key Report Takeaways
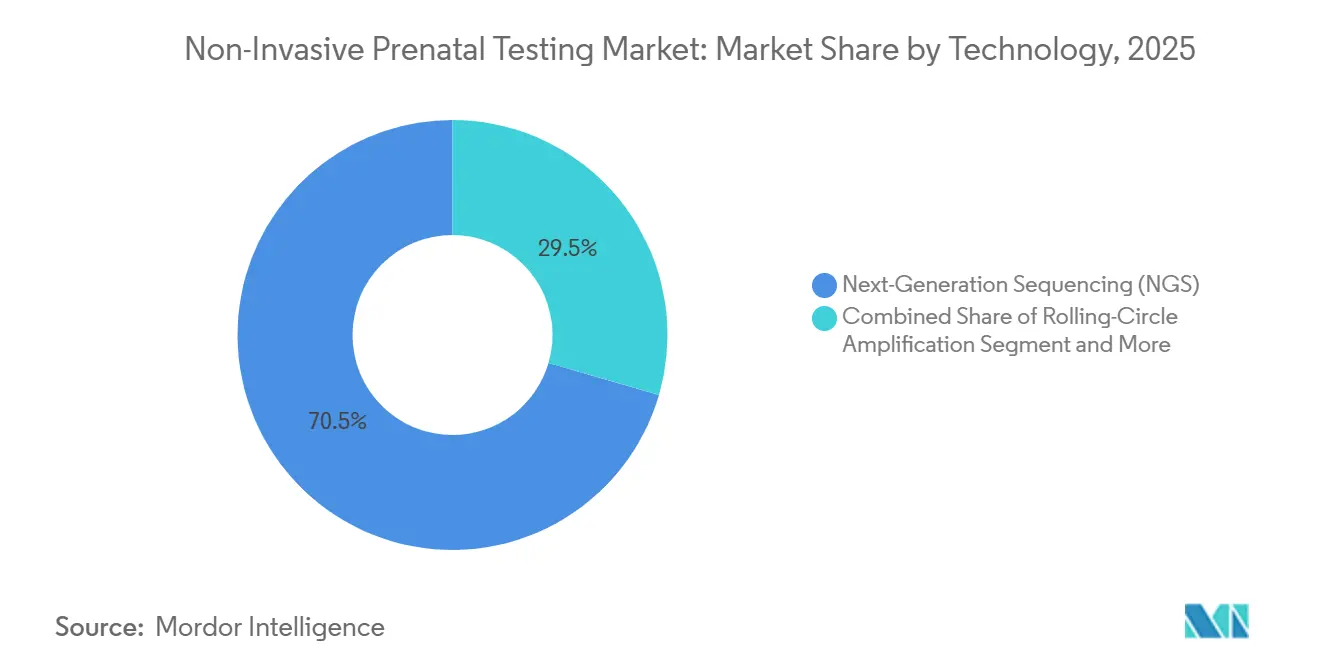
- By technology, next-generation sequencing led with 70.55% of the non-invasive prenatal testing market share in 2025; RCA is projected to expand at an 18.25% CAGR to 2031.
- By test type, aneuploidy screening accounted for 85.53% of the non-invasive prenatal testing market size in 2025, while whole-genome cfDNA screening is advancing at a 19.75% CAGR through 2031.
- By gestation window, the 13-24-week segment captured 50.15% share in 2025; testing at 10-12 weeks is growing at 14.82% CAGR on algorithmic fetal-fraction gains.
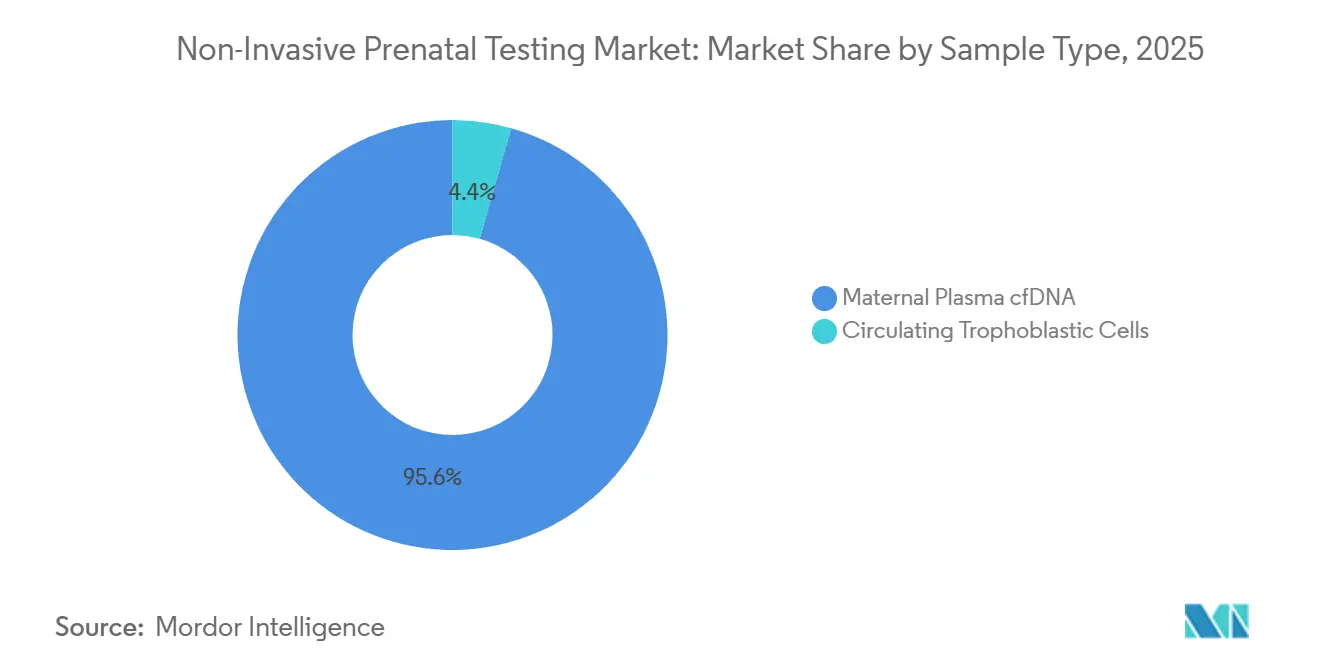
- By sample type, maternal plasma cfDNA dominated with 95.65% share in 2025; circulating trophoblastic cells are forecast to increase at 20.32% CAGR as microfluidics mature.
- By component, services held 62.23% revenue share in 2025, whereas kits & reagents are rising at 16.21% CAGR as hospitals decentralize testing.
- By end user, diagnostic laboratories controlled 58.5% in 2025; IVF & fertility clinics show the fastest 17.42% CAGR due to bundled PGT-A workflows.
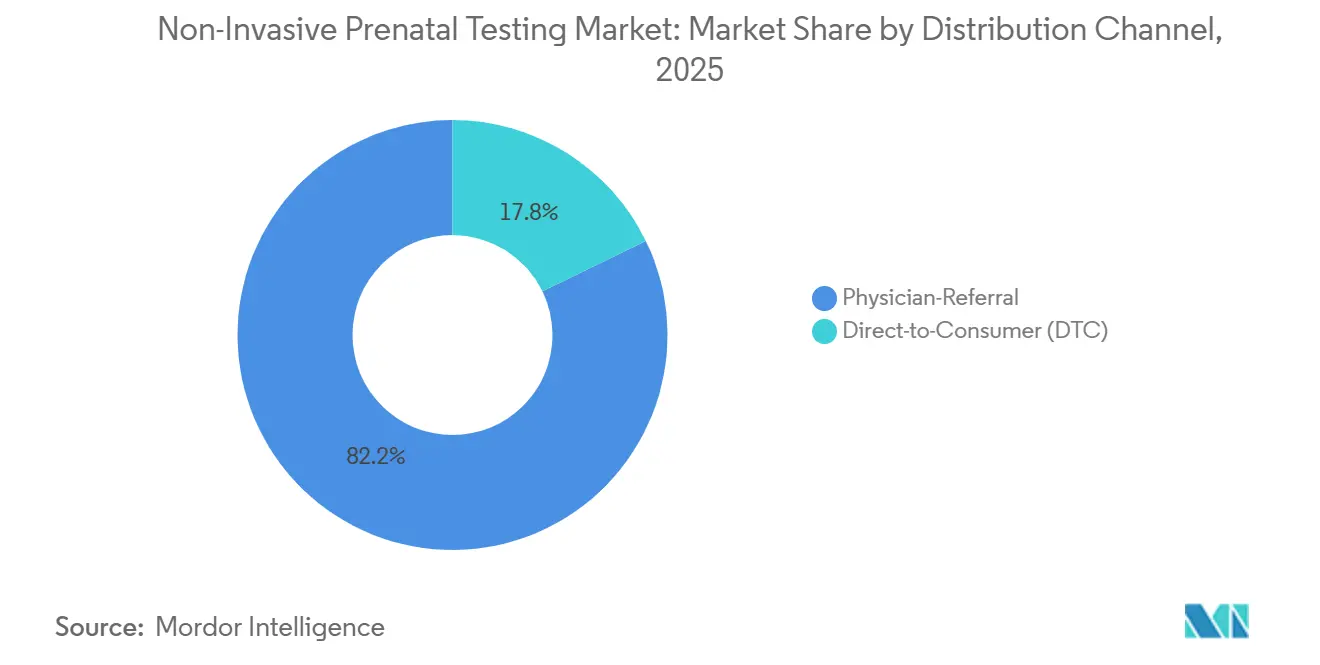
- By distribution channel, physician-referral paths held 82.23% in 2025, yet direct-to-consumer services are expanding at 21.02% CAGR despite stricter FDA oversight.
- By application, Down syndrome detection represented 72.52% revenue in 2025, while Turner syndrome screening is increasing at 15.12% CAGR on improved sex-chromosome calling.
- By geography, North America led with 45.23% share in 2025; Asia-Pacific is the fastest region, growing at 16.42% CAGR on domestic platform approvals and payer adoption.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Non-Invasive Prenatal Testing Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising global maternal age elevating aneuploidy risk | +2.8% | Global, with acute concentration in North America, Europe, Japan, South Korea | Long term (≥ 4 years) |
| Shift from invasive karyotyping to cfDNA screening | +3.1% | Global, led by North America & EU, accelerating in APAC urban centers | Medium term (2-4 years) |
| Sequencing cost curve less than USD 200/sample enabling mass adoption | +2.5% | Global, most pronounced in price-sensitive APAC and Latin America markets | Short term (≤ 2 years) |
| Payer coverage expansion to average-risk pregnancies | +2.2% | North America & EU core, selective adoption in GCC and urban China | Medium term (2-4 years) |
| AI-assisted fetal fraction calling improving first-trimester accuracy | +1.6% | Global, early gains in North America, EU, Australia, South Korea | Medium term (2-4 years) |
| Bundled reproductive-genetic panels (carrier + NIPT) gaining traction | +1.3% | North America, EU, Israel, Singapore; emerging in India, Brazil | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Rising Global Maternal Age Elevating Aneuploidy Risk
Women are delaying childbirth for economic and career reasons, pushing the median maternal age higher in every OECD country. In the United States, the mean age at first birth reached 27.8 years in 2024. Age-linked trisomy incidence drives obstetricians to recommend cfDNA screening universally, especially where public programs subsidize testing for women ≥ 35 years. National registries in Denmark, Japan, and South Korea record parallel shifts, converting high-risk screening into population-level demand. As older mothers often pursue prenatal care earlier and more frequently, laboratories can capture samples sooner, lifting overall test volumes. This demographic trend feeds long-term growth for the non-invasive prenatal testing market by expanding both the eligible base and the clinical urgency for accurate, low-risk diagnostics.
Shift from Invasive Karyotyping to cfDNA Screening
Professional societies now endorse cfDNA as first-line screening, eliminating legacy risk-stratification hurdles. ACOG’s 2024 bulletin recommends cfDNA for all pregnancies, while ACMG’s 2025 statement highlights > 99% detection for trisomy 21 versus 85% for biochemical methods. Payers have followed suit; UnitedHealthcare, Centene, and Medica stopped requiring high-risk justification in 2024-2025, transforming care pathways. Amniocentesis, though still definitive, carries a 0.1-0.3% loss risk that many patients now avoid, further propelling cfDNA volumes. As laboratories retire serum screening platforms, sequencing capacity is redeployed to support expanded panels, reinforcing the migration away from invasive diagnostics and boosting the non-invasive prenatal testing market.
Sequencing Cost Curve Less Than USD 200/Sample Enabling Mass Adoption
High-throughput instruments such as Illumina’s NovaSeq X deliver whole-genome reads at USD 200, down 40% from 2022 levels[1]Illumina Inc., “NovaSeq X Platform Economics,” Illumina, ILLUMINA.COM. Complete Genomics’ DNBSEQ-T7 claims sub-USD 150 costs in 2025, pressuring incumbents on price. Public programs respond: California’s Prenatal Screening Program reimburses NIPT at USD 344, reflecting falling lab expense. Lower input costs let providers offer tests nearer biochemical-screening prices, unlocking adoption in middle-income brackets across China, India, and Brazil. As volume scales, reagent suppliers shift to subscription models, making per-test economics even more favorable and sustaining growth for the non-invasive prenatal testing market.
Payer Coverage Expansion to Average-Risk Pregnancies
Coverage has widened dramatically. UnitedHealthcare’s 2024 policy opened benefits to 2.8 million additional pregnancies each year. Centene mirrored this in 2025, while Medicare’s draft NCD proposes funding for women ≥ 35 years or abnormal ultrasound findings. These decisions erase cost barriers and integrate NIPT into routine prenatal panels. Even where microdeletion panels remain excluded, core aneuploidy screening volumes spike. Commercial labs that once relied on cash-pay patients now secure predictable reimbursement, improving margin stability and reinforcing the expansion trajectory of the non-invasive prenatal testing market.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Persisting ethical debate on secondary findings & sex selection | -1.2% | Global, most acute in India, China, Middle East; regulatory friction in EU | Long term (≥ 4 years) |
| Bioinformatics talent shortage in emerging markets | -0.9% | APAC (ex-Japan, Australia), Latin America, Middle East & Africa | Medium term (2-4 years) |
| Discordant results in multifetal / IVF pregnancies dampen clinician confidence | -0.7% | Global, concentrated in markets with high IVF utilization (Israel, Spain, Denmark, Japan) | Medium term (2-4 years) |
| Reimbursement push-back on microdeletion add-ons | -1.1% | North America & EU core, selective coverage in private-pay Asia markets | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Persisting Ethical Debate on Secondary Findings & Sex Selection
Incidental maternal findings—such as malignancy signals in cfDNA—raise disclosure dilemmas that require extra counseling time and consent forms. In India, audits uncovered clinics reporting fetal sex in violation of the PCPNDT Act, prompting license suspensions[2]Ministry of Health and Family Welfare India, “PCPNDT Audit 2024,” MOHFW.GOV.IN. China similarly bans non-medical sex reporting. The European Society of Human Genetics now urges explicit pre-test consent for secondary findings. Added administrative overhead can deter providers and delay testing, moderating growth in the non-invasive prenatal testing market.
Bioinformatics Talent Shortage in Emerging Markets
ISCB tallied fewer than 800 clinical-genomics bioinformaticians across India, Brazil, and Southeast Asia in 2024. Limited local expertise forces outsourcing of data analysis, adding up to five days of turnaround and eroding cost advantages. South Africa’s pilot NIPT service experienced 12-week backlogs over software staffing gaps. Although BGI’s training academy graduated 240 analysts in 2024, demand still outstrips supply, capping throughput expansion in several high-growth regions of the non-invasive prenatal testing market.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Technology: Isothermal Methods Challenge Sequencing Dominance
The non-invasive prenatal testing market size for technology reached USD 3.9 billion in 2025, with next-generation sequencing accounting for 70.55%. RCA platforms, however, are accelerating at an 18.25% CAGR on simpler, benchtop hardware that slashes capital costs. Clinical Chemistry reported 98.7% concordance between RCA and NGS for trisomy 21 detection, cutting library prep time to 90 minutes. NGS remains entrenched thanks to FDA grandfathering and CE-IVD validation pathways, but small hospitals favor RCA kits that match daily throughput needs.
NGS players continue to enhance throughput; Illumina’s NextSeq 2000 multiple-flow-cell architecture allows labs to batch low-volume runs flexibly, while Thermo Fisher’s Genexus integrates sample-to-report automation. Even so, isothermal vendors appeal to decentralized networks in Latin America and Southeast Asia where power stability and HVAC constraints limit traditional sequencers. Patent disputes between Illumina and Natera over SNP methodologies inject uncertainty, prompting some providers to trial open-filed RCA alternatives. Overall, technology diversification broadens supplier competition and underpins sustained growth for the non-invasive prenatal testing market.

By Test Type: Genome-Wide Panels Gain Ground
In 2025, aneuploidy panels held 85.53% of the non-invasive prenatal testing market share. Whole-genome cfDNA screening, meanwhile, is growing at 19.75% CAGR as clinicians seek copy-number variant (CNV) insights beyond common trisomies. The Lancet study of 90,000 pregnancies identified clinically significant CNVs in 1.7% of cases missed by standard panels. Despite payer hesitancy, private patients opt for broader coverage, especially in China where domestic kits bundle genome-wide analysis at sub-USD 450 price points.
Microdeletion screening faces reimbursement roadblocks in the United States but gains traction in Germany and Israel for high-risk cohorts. Rh-D genotyping remains a niche, stabilizing revenues in Rhesus-negative populations. Monogenic disorder cfDNA panels are emerging; Natera’s 21-gene Fetal Focus launch in 2026 illustrates a pivot toward single-gene detection without extra blood draws. As validation datasets expand, broader panels could shift payer perspectives, accelerating revenue diversification inside the non-invasive prenatal testing market.
By Gestation Window: Earlier Testing Reshapes Workflows
The non-invasive prenatal testing market size tied to the 13-24-week capturing 50.15%. Algorithms that refine fetal-fraction estimates now support reliable calls as early as 10 weeks, propelling the 10-12-week cohort at a 14.82% CAGR. Obstetrics & Gynecology data show patients tested before 13 weeks are 40% likelier to act on abnormal results within legal termination timelines.
Earlier draws appeal to telehealth prenatal programs, permitting same-day phlebotomy at primary-care clinics. However, strict ultrasound dating requirements in Japan and parts of Europe slow first-trimester uptake. Laboratories mitigate redraw risk through predictive models that flag low expected fetal fraction, scheduling later collections when necessary. Wider first-trimester adoption accelerates turnover and elevates total addressable volume across the non-invasive prenatal testing market.
By Sample Type: Fetal Cell Isolation Emerges
Maternal cfDNA retained 95.65% share in 2025. Yet rare fetal cells, captured via microfluidics, are forecast to rise at 20.32% CAGR. Science Translational Medicine demonstrated 96% karyotype concordance to amniocentesis using DEPArray-isolated cells. Single-cell analysis may solve confined placental mosaicism false positives but currently costs USD 200-300 above cfDNA workflows and lacks FDA clearance.
Vendors target high-risk patients refusing invasive tests; Menarini won CE-IVD in 2024, and Rarecells plans a 2027 launch. Automation and economies of scale are critical to mainstream adoption. If per-test costs fall below USD 400, fetal-cell NIPT could cannibalize cfDNA share, introducing fresh competition inside the non-invasive prenatal testing market.
By Component: Kits & Reagents Capture Decentralization Momentum
Services dominated revenue at 62.23% in 2025, reflecting centralized labs processing > 10,000 monthly samples. Kits & reagents, however, are growing at 16.21% CAGR as hospital-based genomics labs bring testing in-house to cut 3-5-day courier delays. Illumina’s VeriSeq v2 kit lists at USD 350 per sample with embedded DRAGEN analytics, letting mid-volume centers break even at 400 tests per year.
Instrument makers downsize footprints; Thermo Fisher’s Genexus occupies < 2 m², ideal for suburban hospitals. Europe’s IVDR accelerates kit demand by mandating CE-IVD products, disadvantaging home-brew assays. As kit penetration rises, reagent suppliers diversify distribution, bolstering recurring revenue streams for stakeholders in the non-invasive prenatal testing market.
By End User: IVF Clinics Integrate Genetics Across the Care Continuum
Diagnostic laboratories controlled 58.5% share in 2025, serving obstetric offices through established courier loops. IVF & fertility clinics will outpace at 17.42% CAGR, pairing PGT-A embryo screens with pregnancy NIPT follow-up. A Fertility & Sterility survey showed 62% of IVF patients choose NIPT compared with 34% in natural conceptions. Clinics appreciate integrated results spanning carrier status, embryo genetics, and pregnancy health, reinforcing their role as premium genetic services hubs.
Hospitals retain relevance in markets with bundled maternity packages, yet facility fees make them costlier than freestanding labs. Research institutes focus on expanding test scope to preeclampsia risk and single-gene disorders. The IVF channel’s growth increases sample complexity—multiple gestations, mosaicism—spurring algorithm refinements that benefit the wider non-invasive prenatal testing market.
By Distribution Channel: Direct-to-Consumer Models Test Regulatory Boundaries
Physician-referral pathways generated 82.23% of 2025 revenue, underlining clinician gatekeeping in most jurisdictions. Direct-to-consumer platforms are set to grow at a 21.02% CAGR. FDA draft guidance requires 510(k) review for at-home kits, pushing players like Everly Health to pause offerings, yet providers that embed telehealth consults continue operating.
Transparent USD 299 price points attract younger pregnant individuals lacking established obstetric care. NSGC’s 2025 poll found 18% of orders initiated without onsite physician visits. Europe bans pure DTC marketing in key countries, while Australia permits it with mandatory counseling. Regulatory clarity will decide whether DTC models remain a niche convenience or evolve into a mainstream conduit for the non-invasive prenatal testing market.
By Application: Sex-Chromosome Panels Narrow False Positives
Down syndrome commands 72.52% of 2025 revenue thanks to prevalence and strong clinical consensus. Turner syndrome, however, is the fastest-rising application at 15.12% CAGR after algorithmic threshold adjustments halved false positives. UnitedHealthcare now reimburses sex-chromosome aneuploidy panels, broadening payer acceptance beyond trisomies.
Clinicians value comprehensive risk insights but caution that variable phenotypes complicate counseling. Combining cfDNA with first-trimester ultrasound boosts positive predictive value to 78% for monosomy X. As machine-learning-derived risk scores mature, sex-chromosome panels should gather incremental share, diversifying application revenues within the non-invasive prenatal testing market.
Geography Analysis
North America led the non-invasive prenatal testing market in 2025 with a 45.23% share, supported by guideline-driven universal screening and payers that removed risk prerequisites. Average negotiated test prices remain high at USD 800-1,200, sustaining healthy margins for U.S. labs. Canada’s funding varies provincially; Ontario covers women ≥ 40 years, while British Columbia relies on private pay, capping national penetration at 35%. Mexico’s private hospitals offer USD 600-800 tests, but public institutions still default to biochemical screening.
Asia-Pacific is the fastest-growing region at 16.42% CAGR. China’s NMPA approvals for BGI and Berry Genomics platforms lifted national test volumes 40% year-over-year. Domestic sequencing economics enable USD 150-300 pricing, expanding access beyond top-tier cities. Japan’s certified facility network increased to 142 in 2025, though cautious counseling slows mass adoption. India’s urban middle class drives volumes at INR 18,000 (USD 215) per test, yet rural cold-chain gaps limit nationwide reach.
Europe shows mid-single-digit growth influenced by IVDR compliance costs. Germany funds only high-risk cases but covers microdeletions for select patients. The U.K.’s NHS restricts NIPT to women flagged high-risk by first-trimester screens, a targeted policy that still cut amniocentesis 60%. Middle East adoption is led by the UAE, where expatriate populations demand premium prenatal care; Dubai labs report 25% annual growth. Latin America is nascent beyond Brazil, where Dasa launched USD 500 NIPT in 2024.
Competitive Landscape
Market concentration is moderate: the top five suppliers hold a significant share of global revenue, leaving room for regional challengers. Labcorp’s 2024 Invitae acquisition merged 1.2 million annual tests into a 2,000-site network, consolidating U.S. capacity. BGI Genomics enjoys 40% gross margins by controlling sequencers, reagents, and software, undercutting Western pricing across APAC[3]BGI Genomics, “Annual Report 2024,” BGI.COM. Natera differentiates through SNP-based analytics claiming 99.9% trisomy 21 sensitivity, though patent litigation with Illumina continues to shape technology choice.
Emerging entrants pursue fetal-cell isolation to solve placental mosaicism false positives; Menarini’s DEPArray earned CE-IVD in 2024. Oxford Nanopore pilots same-day NIPT on portable sequencers, courting point-of-care scenarios. Strategic moves include Cooper Surgical’s EMR integration with Illumina, streamlining ordering in IVF clinics. As payer scrutiny intensifies, labs emphasize reporting quality and genetic-counselor access to defend premium pricing in the non-invasive prenatal testing market.
Non-Invasive Prenatal Testing Industry Leaders
Centogene NV
F. Hoffmann-La Roche Ltd.
Eurofins Scientific SE
Revvity, Inc.
BGI Genomics Co. Ltd
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2026: BillionToOne launched UNITY’s expanded Red Blood Cell Fetal Antigen NIPT and the first platelet antigen cfDNA assay.
- January 2026: Natera unveiled a 21-gene Fetal Focus single-gene NIPT, backed by blinded EXPAND trial results.
Global Non-Invasive Prenatal Testing Market Report Scope
As per the scope of the report, non-invasive prenatal testing (NIPT), also called non-invasive prenatal screening (NIPS), is a method for assessing the risk that a fetus will be born with certain genetic abnormalities. It is based on analysis of cell-free DNA (cfDNA) in maternal blood. This testing analyzes small DNA fragments circulating in a pregnant woman's blood. NIPT analyzes the genetic information in this DNA to screen for several abnormalities.
The segmentation of the Non-Invasive Prenatal Testing (NIPT) market is categorized by technology, test type, gestation window, sample type, component, end user, distribution channel, application, and geography. By technology, the market includes next-generation sequencing (NGS), rolling-circle amplification, microarray, real-time PCR, and other technologies. By test type, it is segmented into aneuploidy screening, microdeletion/microduplication screening, whole-genome cfDNA screening, Rh-D genotyping, and monogenic disease testing. By gestation window, the segmentation covers 10–12 weeks, 13–24 weeks, and more than 24 weeks. By sample type, it includes maternal plasma cfDNA and circulating trophoblastic cells. By component, the market is divided into instruments, kits and reagents, and services. By end user, the segmentation includes hospitals and birthing centers, diagnostic laboratories, IVF and fertility clinics, and research institutes. By distribution channel, it is categorized into physician-referral and direct-to-consumer (DTC). By application, the market covers Down syndrome (trisomy 21), Edwards syndrome (trisomy 18), Patau syndrome (trisomy 13), Turner syndrome, and other chromosomal abnormalities. By geography, the segmentation includes North America, Europe, Asia-Pacific, the Middle East and Africa, and South America. The report offers the value (in USD) for the above segments.
| Next-Generation Sequencing (NGS) |
| Rolling-Circle Amplification |
| Microarray |
| Real-Time PCR |
| Other Technologies |
| Aneuploidy Screening |
| Microdeletion / Microduplication Screening |
| Whole-Genome cfDNA Screening |
| Rh-D Genotyping |
| Monogenic Disease Testing |
| 10 - 12 Weeks |
| 13 - 24 Weeks |
| > 24 Weeks |
| Maternal Plasma cfDNA |
| Circulating Trophoblastic Cells |
| Instruments |
| Kits & Reagents |
| Services |
| Hospitals & Birthing Centers |
| Diagnostic Laboratories |
| IVF & Fertility Clinics |
| Research Institutes |
| Physician-Referral |
| Direct-to-Consumer (DTC) |
| Down Syndrome (Trisomy 21) |
| Edwards Syndrome (Trisomy 18) |
| Patau Syndrome (Trisomy 13) |
| Turner Syndrome |
| Other Chromosomal Abnormalities |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Technology | Next-Generation Sequencing (NGS) | |
| Rolling-Circle Amplification | ||
| Microarray | ||
| Real-Time PCR | ||
| Other Technologies | ||
| By Test Type | Aneuploidy Screening | |
| Microdeletion / Microduplication Screening | ||
| Whole-Genome cfDNA Screening | ||
| Rh-D Genotyping | ||
| Monogenic Disease Testing | ||
| By Gestation Window | 10 - 12 Weeks | |
| 13 - 24 Weeks | ||
| > 24 Weeks | ||
| By Sample Type | Maternal Plasma cfDNA | |
| Circulating Trophoblastic Cells | ||
| By Component | Instruments | |
| Kits & Reagents | ||
| Services | ||
| By End User | Hospitals & Birthing Centers | |
| Diagnostic Laboratories | ||
| IVF & Fertility Clinics | ||
| Research Institutes | ||
| By Distribution Channel | Physician-Referral | |
| Direct-to-Consumer (DTC) | ||
| By Application | Down Syndrome (Trisomy 21) | |
| Edwards Syndrome (Trisomy 18) | ||
| Patau Syndrome (Trisomy 13) | ||
| Turner Syndrome | ||
| Other Chromosomal Abnormalities | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
How quickly is the non-invasive prenatal testing market expected to grow through 2031?
Revenue is forecast to rise from USD 4.43 billion in 2026 to USD 8.36 billion by 2031, advancing at a 13.53% CAGR.
Which technology is gaining momentum against next-generation sequencing?
Rolling-circle amplification is the fastest-growing method, expanding at an 18.25% CAGR due to its isothermal workflow and lower capital costs.
Why is Asia-Pacific the fastest regional market?
Domestic platform approvals, sub-USD 300 pricing, and rising maternal age push Asia-Pacific volumes at a 16.42% CAGR.
What is driving early-gestation testing demand?
AI-enhanced fetal-fraction algorithms reduce first-trimester no-call rates, enabling accurate results from 10 weeks gestation.
How are IVF clinics influencing test adoption?
Bundled embryo screening and pregnancy NIPT packages boost uptake, making IVF centers the fastest-growing end-user category at 17.42% CAGR.
Page last updated on:









