Multiple Sclerosis Therapeutics Market Size and Share
Multiple Sclerosis Therapeutics Market Analysis by Mordor Intelligence
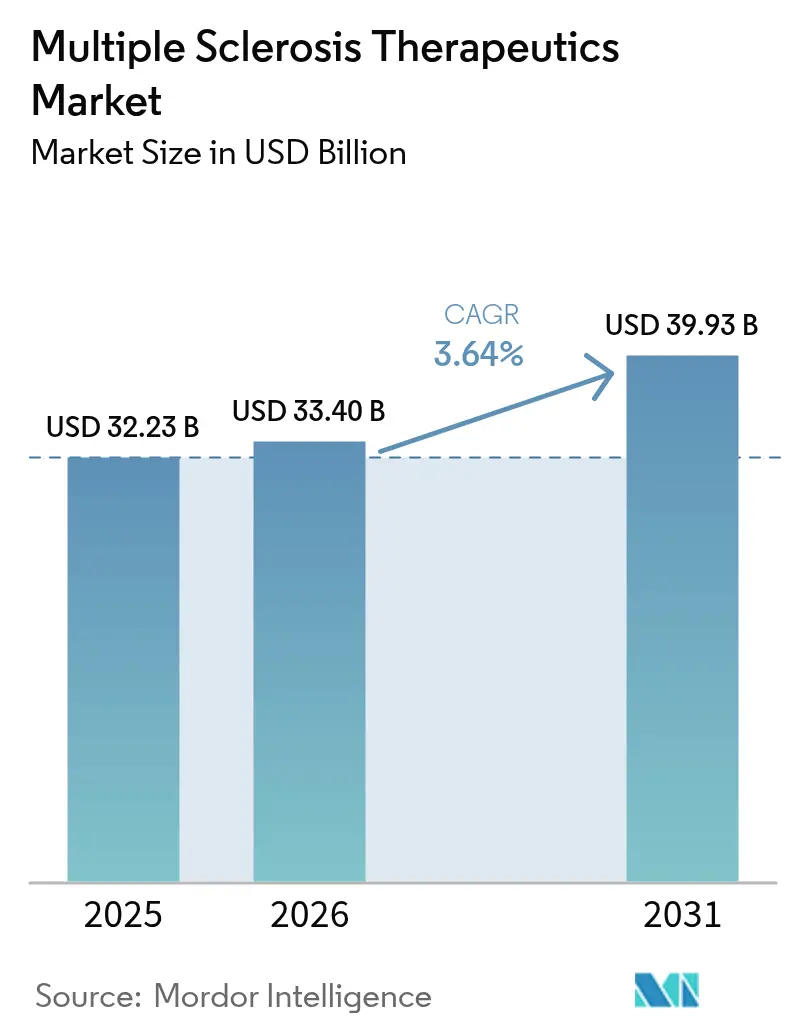
The Multiple Sclerosis Therapeutics Market size is projected to expand from USD 32.23 billion in 2025 and USD 33.40 billion in 2026 to USD 39.93 billion by 2031, registering a CAGR of 3.64% between 2026 to 2031.
Growth in the multiple sclerosis therapeutics market reflects a shift toward outcomes-based reimbursement in OECD health systems, where payers favor therapies that demonstrate strong real-world persistence and measurable reductions in relapse-related admissions, reshaping pricing power and reward structures for manufacturers. Large-molecule platforms continue to anchor revenue as subcutaneous monoclonal antibody options reduce reliance on infusion centers and align with adherence-focused care models that integrate smartphone monitoring and nurse support. Oral options are gaining popularity faster because once-daily S1P modulators minimize dosing friction for treatment-naive patients who prefer pills over injections when efficacy is similar.
Multiple Sclerosis (MS) affects cognitive, emotional, motor, sensory, and visual functions and is caused by the immune system attacking the brain and spinal cord. In recent years, the burden of MS has been observed to be on the rise, which is boosting the demand in the multiple sclerosis therapeutics market for proper disease management. For instance, according to the August 2024 update of the World Health Organization (WHO), more than 2.8 million people globally are now estimated to live with MS, including 1 million in the United States annually, highlighting the significant size of the MS in the United States. Thus, this escalating burden of MS will provide lucrative growth opportunities for the market during the forecast period.
Key Report Takeaways
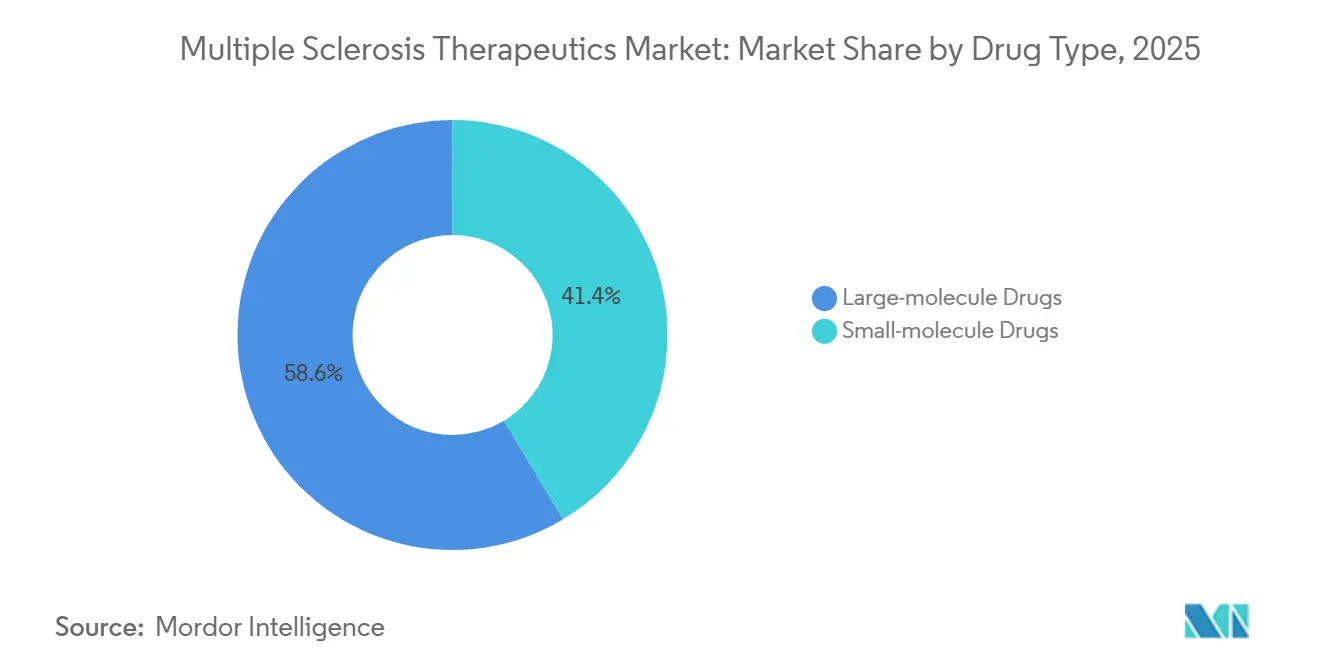
- By drug type, large-molecule drugs led with 58.60% revenue share in 2025, and this segment is projected to expand at a 7.50% CAGR through 2031.
- By route of administration, injectables accounted for 48.02% share in 2025, while oral therapies are forecast to grow at 8.50% through 2031.
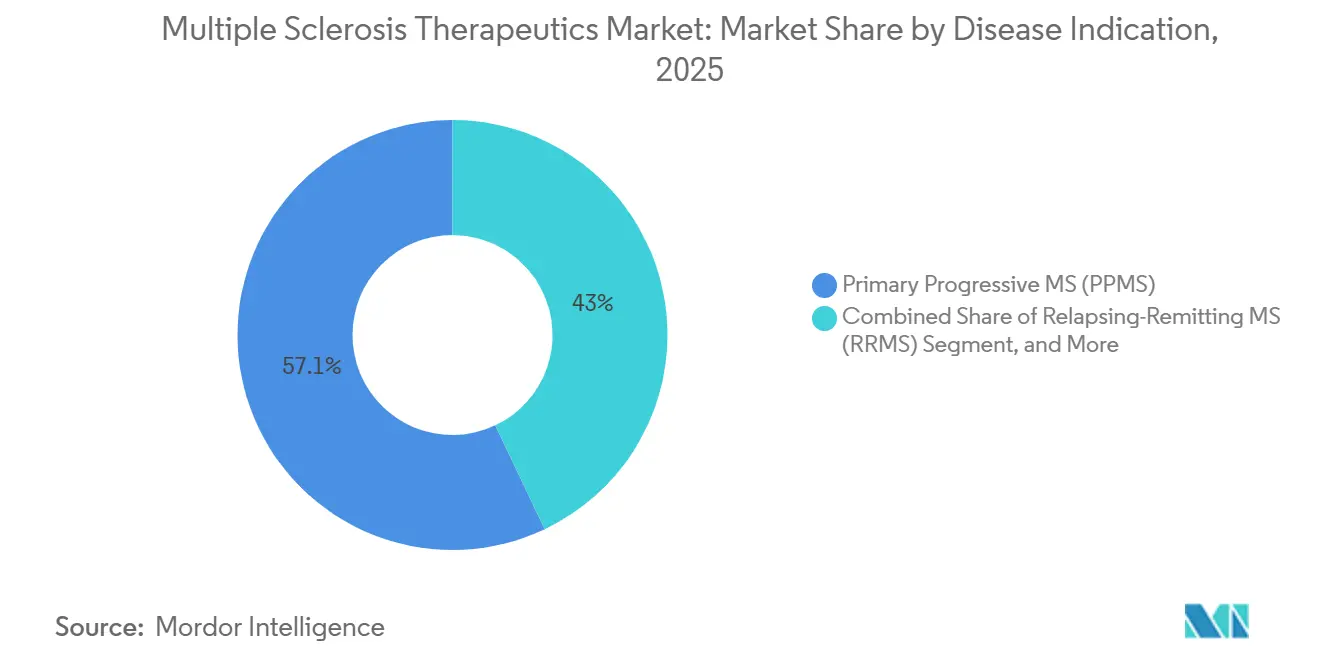
- By disease indication, primary progressive multiple sclerosis accounted for 57.05% in 2025, and secondary progressive multiple sclerosis is projected to grow at 7.02% through 2031.
- By drug class, immunomodulators accounted for 37.30% of revenue in 2025, while monoclonal antibodies are projected to grow at 7.11% through 2031.
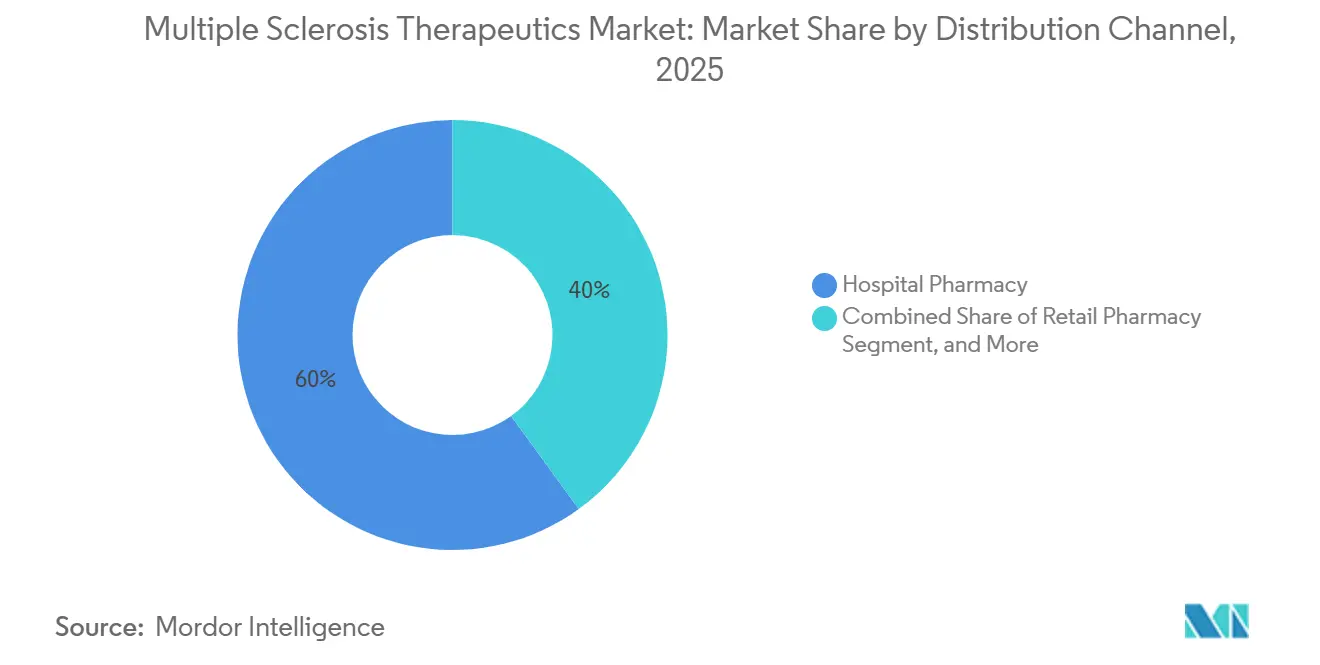
- By distribution channel, hospital pharmacies accounted for 60.00% of dispensing revenue in 2025, while online and specialty pharmacies are projected to grow at 8.02%.
- By geography, North America accounted for 41.76% of revenue in 2025, while Asia-Pacific is forecast to grow at 7.22% through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Multiple Sclerosis Therapeutics Market Trends and Insights
Drivers Impact Analysis*
| DRIVER | (~) % IMPACT ON CAGR FORECAST | GEOGRAPHIC RELEVANCE | IMPACT TIMELINE |
|---|---|---|---|
| Rising global prevalence of MS | +0.8% | Global, with concentration in North America and Northern Europe | Long term (≥ 4 years) |
| Shift toward high-efficacy monoclonal-antibody DMTs | +1.2% | North America, Western Europe, urban Asia-Pacific | Medium term (2-4 years) |
| Broader reimbursement & access programs in OECD markets | +0.9% | OECD nations, early adoption in Gulf Cooperation Council | Medium term (2-4 years) |
| Rapid uptake of once-daily oral DMTs | +1.0% | Global, accelerated in Asia-Pacific and Latin America | Short term (≤ 2 years) |
| Pipeline of CNS-penetrant BTK inhibitors | +0.7% | North America and Europe initially, spillover to Asia-Pacific | Long term (≥ 4 years) |
| AI-driven adherence platforms improving real-world persistence | +0.5% | North America, Western Europe, pilot programs in urban China | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Rising Global Prevalence of MS
Diagnosed multiple sclerosis cases surpassed 2.8 million globally in 2024, driven by broader MRI access and the wider use of updated diagnostic criteria that capture earlier disease activity.[1]World Health Organization, “Atlas of Multiple Sclerosis and Global Burden 2024,” WHO, who.int Prevalence remains highest at higher latitudes, while incidence is accelerating in fast-urbanizing regions that face overlapping risks from lower vitamin D exposure, pollution, and Epstein-Barr virus seroprevalence among susceptible populations.[2]Alastair Compston, “Epidemiology and Risk Factors in Multiple Sclerosis,” The Lancet Neurology, thelancet.com
Companies are staging launches to align with this distribution, prioritizing originator biologics where prevalence is dense and using biosimilars or generics where budget pressure is stronger, and diagnosis ramps faster. Partnerships with radiology and neurology networks are becoming a route to earlier patient capture, longer therapy duration, and greater lifetime value per patient in the studied market. The market also benefits from clinician education programs that standardize early workups around MRI and serum markers and prompt referral pathways, thereby hastening treatment initiation. These epidemiology-linked actions support steadier volumes, helping cushion pricing cycles over the forecast period.
Shift Toward High-Efficacy Monoclonal-Antibody DMTs
Regulators cleared multiple monoclonal antibodies for relapsing MS in 2024-2025, with approvals anchored in robust reductions in annualized relapse rates and strong MRI lesion suppression. Clinical practice shifted toward earlier use of high-efficacy therapies for patients with aggressive early disease, reducing the median time to initiation to 1.8 years in 2025. Subcutaneous formulations of ocrelizumab and ofatumumab, approved in 2024, enabled home administration under nurse supervision and removed infusion-center bottlenecks in thinly resourced settings. Payer coverage trends for these agents often track digital supports that verify dosing and persistence, which now factor into reimbursement discussions in the multiple sclerosis therapeutics market. As physicians increasingly match therapy to early disease severity, value propositions emphasize disability slowing and reduced acute-care events, which resonate in cost-effectiveness models. This use of high-efficacy treatment earlier in the course of care supports sustained adoption over the forecast period.
Rapid Uptake of Once-Daily Oral DMTs
Patient surveys conducted in 2024 showed a strong preference for oral dosing when efficacy and safety are similar, and this explains the rapid adoption of once-daily regimens in new starts. Real-world adherence data indicate higher 12-month persistence with once-daily dosing than with more frequent oral regimens or injectables, which supports better clinical stability over time. These findings led manufacturers to prioritize extended-release designs that sustain therapeutic exposure with simpler schedules.[3]Nature Research Editorial, “CNS Penetration of Small Molecules in MS,” Nature Medicine, nature.com Evidence from MS care settings links dosing simplicity to fewer missed doses, lower cognitive load, and higher satisfaction, all of which are rewarded in payer frameworks that tie reimbursement to persistence thresholds in the multiple sclerosis therapeutics market. The directional pull toward oral convenience is visible in formulary negotiations where medication possession ratios are scored alongside clinical endpoints. Launch strategies now embed patient onboarding and app-based reminders from the start to protect persistence through the first year of therapy.
Pipeline of CNS-Penetrant BTK Inhibitors
BTK inhibitors such as tolebrutinib, evobrutinib, and orelabrutinib advanced into Phase 3 programs focused on progressive forms of MS during 2024, targeting both B-cell signaling and microglial activation in the central nervous system. Small-molecule profiles allow for blood-brain barrier penetration, supporting inhibition of resident immune processes implicated in neurodegeneration. The FDA granted Breakthrough Therapy designation to tolebrutinib in June 2025 based on interim data showing a 31% reduction in 6-month confirmed disability progression in non-relapsing SPMS, signaling meaningful potential in a population with constrained options. If pivotal data sustain these effects, BTK inhibitors can support a differentiated value story around progressive disease mechanisms and oral convenience in the multiple sclerosis therapeutics market.
Restraint Impact Analysis*
| RESTRAINT | (~) % IMPACT ON CAGR FORECAST | GEOGRAPHIC RELEVANCE | IMPACT TIMELINE |
|---|---|---|---|
| Adverse-event profile of immunosuppressive DMTs | -0.6% | Global, heightened scrutiny in North America and Europe | Short term (≤ 2 years) |
| High treatment cost and payer pressure | -0.9% | North America, Western Europe, emerging in Asia-Pacific | Medium term (2-4 years) |
| Upcoming biosimilar wave eroding branded prices | -1.1% | Europe initially, North America from 2026 onward | Medium term (2-4 years) |
| Insufficient predictive biomarkers for therapy selection | -0.4% | Global | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Adverse-Event Profile of Immunosuppressive DMTs
CD20 and alpha 4 integrin monoclonal antibodies carry risks that require structured monitoring and risk-mitigation steps, including screening for JC virus and heightened imaging vigilance for progressive multifocal leukoencephalopathy. Regulators specify safety programs and label updates that guide vaccination scheduling and infection surveillance, thereby increasing the complexity of care coordination for prescribers. The need for frequent lab work and imaging adds cost that can deter selection for patients with known risk markers, even when efficacy is persuasive. Post-marketing pharmacovigilance records document serious infections associated with immunosuppression, which trigger periodic label refinements and practice advisories.
High Treatment Cost and Payer Pressure
High-efficacy agents face tightening payer controls across prior authorization, step therapy, and outcomes-based contracts that link net pricing to real-world disability trends. Coverage protocols often require trials of lower-cost options before approving premium therapies, which can delay access to the best medical fit for some patients. In major European markets, health technology assessors require evidence that aligns with cost-effectiveness thresholds, and positive listings often follow price concessions or managed access conditions. Manufacturers respond with adherence ecosystems that document persistence to meet payer audit requirements in the Multiple Sclerosis Therapeutics market. These dynamics compress margins on legacy platforms while channeling investment into solutions that deliver system-level savings and verified outcomes.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Drug Type: Biologics Anchor Revenue Despite Oral Gains
Large-molecule drugs accounted for 58.60% of revenue in 2025 and are projected to grow at 7.50% annually through 2031, driven by subcutaneous formats and care models that support home administration with nurse oversight. antibodies dominate the biologics mix, with twice-yearly or monthly dosing that reduces day-to-day decision-making load, helping persistence and reducing acute care utilization tied to relapses. The multiple sclerosis therapeutics market favors large-molecule platforms that align with payer requirements for real-world data capture through connected devices and patient-reported outcome tools. Small molecules remain strategically important where oral convenience and rapid initiation can open access for new starts that avoid injection hesitancy. As digital adherence moves into packaging and hub services, manufacturers emphasize integration that simplifies verification for coverage and reimbursement.
By Route of Administration: Oral Convenience Reshapes Prescribing
In 2025, injectables dominated the revenue landscape, capturing 48.02% of the market share. This highlights the established role of therapies like interferons, glatiramer acetate, and infusion-based antibodies, all of which require controlled administration settings. Meanwhile, oral therapies are projected to grow at a rate of 8.50%, more than double the overall CAGR. A significant factor driving this growth is the appeal of once-daily S1P modulators, particularly for patients who prefer simplified routines during their initial year of treatment. This trend is further supported by the alignment between patient-reported preferences and real-world adherence data, which strengthens the case for oral therapies, especially when their efficacy is comparable to alternatives. At the same time, subcutaneous monoclonal antibodies are simplifying the treatment process. By reducing the need for infusion visits while maintaining high efficacy, they provide a notable advantage, particularly in regions with limited infusion capacity. These evolving dynamics ensure that injectables remain essential for patients who prioritize less frequent administration along with hands-on clinical monitoring.
By Disease Indication: Progressive Phenotypes Drive Premium Pricing
In 2025, Primary Progressive Multiple Sclerosis accounted for 57.05% of the revenue, driven by elevated therapy costs per patient and a lack of approved alternatives, which limited discounting pressures. Meanwhile, Secondary Progressive Multiple Sclerosis is projected to grow at a rate of 7.02% through 2031, as BTK inhibitors target disability progression, addressing mechanisms beyond traditional inflammatory activities. Although Clinically Isolated Syndrome represents a smaller segment, it continues to grow due to earlier treatments informed by MRI indicators predicting a shift to a definitive disease. These evolving dynamics are concentrating value in progressive forms, where unmet need remains high. Additionally, payer willingness to invest is influenced by disability trajectories and associated system costs. Looking ahead, differentiation among progressive phenotypes is expected to focus on CNS penetration and neurodegeneration markers.

By Drug Class: Monoclonal Antibodies Outpace Legacy Platforms
In 2025, immunomodulators like interferons and glatiramer acetate accounted for 37.30% of the revenue. However, monoclonal antibodies, driven by their efficacy and increasing acceptance among payers, are projected to grow at a rate of 7.11%, capturing a larger market share as hospitalizations decline. CD20 and alpha‑4 integrin antibodies, offering superior relapse-reduction effects compared to older agents, result in fewer emergency department visits and imaging events in patient care. S1P modulators, representing 19% of the market value, continue to gain momentum as second-generation compounds address first-dose monitoring challenges and align with the preference for oral administration. These differences among classes enable clear positioning in care pathways, securing alignment from both physicians and payers. Furthermore, digitally enabled devices and home administration services strengthen the position of antibodies in high-efficacy segments.
By Distribution Channel: Specialty Pharmacy Gains as Hub Services Expand
In 2025, hospital pharmacies accounted for 60.00% of dispensed multiple sclerosis therapeutics, as the nature of infusion logistics and the use of high-risk agents necessitate controlled environments and immediate emergency support. Retail channels, on the other hand, cater to stable oral regimens, aligning with routine refill cycles and incurring lower monitoring costs for plans. Online and specialty pharmacies are set to witness an 8.02% growth, driven by hub services that integrate copay support, nurse case management, and adherence apps into a unified interface, enhancing patient persistence and retention. Both manufacturers and specialty pharmacies are establishing proprietary registries to monitor dosing, refills, and side effects, aiding in payer contracting and fulfilling post-marketing obligations. These data repositories not only serve their primary purpose but also generate feedback loops that enhance patient engagement and refine targeted outreach workflows.

Geography Analysis
North America accounted for 41.76% of revenue in 2025, driven by elevated per-patient spending, swift regulatory reviews, and timely payer access decisions. Regional policy discussions now emphasize outcome-linked contracts that partially refund drug costs if real-world disability trends differ from trial results. This focus strengthens the emphasis on verified persistence and functional outcomes. As biosimilars for high-efficacy agents approach their U.S. market debut, originators are shifting attention to device enhancements and supportive ecosystems, differentiating beyond molecule identity. Providers in integrated delivery networks are well-positioned to benefit from adherence-linked reimbursements, given their systems' ability to capture closed-loop data that meets payer audit requirements. These factors keep the Multiple Sclerosis Therapeutics market focused on real-world evidence demonstrating system-level value.
Europe captured a significant share of 2025 revenue, with Germany, the U.K., France, Italy, and Spain accounting for the majority of sales. Growth trends reflect a more substantial reliance on reference pricing and managed access to influence net costs. The U.K.'s National Institute for Health and Care Excellence has tightened cost-effectiveness thresholds, requiring evidence aligned with real-world outcomes and adherence documentation for positive guidance.
Asia-Pacific is projected to grow at 7.22% through 2031, driven by faster regulatory processes and provincial approvals increasingly aligned with national approvals. During 2024-2025, China approved several novel disease-modifying therapies and moved them into reimbursement pathways more quickly than European timelines, expanding eligible patient pools shortly after launch. Local producers in India have scaled older platforms for export, supporting access in regions with constrained branded pricing, while innovators defend their market share through device patents and support services. Urban centers across the Asia-Pacific are well-suited to pilot AI-enabled adherence platforms, given the growing interoperability of pharmacy claims and EHR data across major hospital networks. These trends indicate that the Multiple Sclerosis Therapeutics market in Asia-Pacific will continue to combine regulatory speed with digital adoption over the forecast period.
Competitive Landscape
Biogen, Roche, Novartis, Sanofi, and Bristol Myers Squibb are projected to command a combined 62% share of the multiple sclerosis therapeutics market, indicating a moderate concentration in the sector. As the EMA greenlights high-efficacy biosimilars, the competitive landscape intensifies. These approvals not only open up tenders but also compel original manufacturers to stand out, emphasizing digital adherence, home administration, and coordinated long-term care services. Roche and Novartis have broadened their offerings to include subcutaneous administration and at-home nurse support. This strategic move enhances flexibility, bolstering patient persistence and diminishing reliance on infusion centers. Biogen's investments in digital tools and patient services underscore this shift. Their initiatives, which ensure accurate dosing verification, consolidate patient-reported outcomes, and facilitate payer audits for outcome-based contracts, highlight a broader trend. The industry is transitioning from merely innovating molecules to cultivating comprehensive patient ecosystems, generating validated real-world data in the multiple sclerosis therapeutics market.
Multiple Sclerosis Therapeutics Industry Leaders
Biogen Inc.
Novartis AG
Merck KGaA
Sanofi S. A.
Acorda Therapeutics, Inc.
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2026: TG Therapeutics announced new data presentations for BRIUMVI (ublituximab xiiy) at the ACTRIMS annual forum in San Diego, California.
- November 2025: Sandoz reported the United States availability of TYRUKO (natalizumab sztn), the first and only FDA-approved natalizumab biosimilar for relapsing forms of MS.
- April 2025: Sanofi announced positive top-line Phase 3 HERCULES results for tolebrutinib in non-relapsing SPMS, demonstrating a 31% reduction in 6-month confirmed disability progression versus placebo, and the FDA granted Priority Review in March 2025, with a PDUFA date set for October 2026.
Global Multiple Sclerosis Therapeutics Market Report Scope
As per the scope of the report, multiple sclerosis (MS) involves an immune-mediated process in which an abnormal response of the body’s immune system is directed against the central nervous system (CNS).
The multiple sclerosis therapeutics market is segmented by drug type, route of administration, disease indication, drug class, distribution channel, and geography. By drug type, the market is segmented into small-molecule drugs and large-molecule drugs. By route of administration, the market is segmented into oral, injectable, and other routes. By drug indication, the market is segmented into relapsing-remitting MS (RRMS), secondary progressive MS (SPMS), primary progressive MS (PPMS), and clinically isolated syndrome (CIS). By drug class, the market is segmented into immunomodulators, monoclonal antibodies, S1p receptor modulators, and antineoplastics /others. By distribution channel, the market is segmented into hospital pharmacies, retail pharmacies, and online/specialty pharmacies. By geography, the market is segmented into North America, Europe, Asia-Pacific, Middle East and Africa, and South America. The market report also covers the estimated market sizes and trends for 17 different countries across major regions globally. The report offers market size and forecasts in value (USD) for the above segments.
| Large-molecule Drugs |
| Small-molecule Drugs |
| Oral |
| Injectable |
| Others |
| Relapsing-Remitting MS (RRMS) |
| Secondary Progressive MS (SPMS) |
| Primary Progressive MS (PPMS) |
| Clinically Isolated Syndrome (CIS) |
| Immunomodulators |
| Monoclonal Antibodies |
| S1P Receptor Modulators |
| Antineoplastics / Others |
| Hospital Pharmacy |
| Retail Pharmacy |
| Online/Specialty Pharmacy |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Drug Type | Large-molecule Drugs | |
| Small-molecule Drugs | ||
| By Route of Administration | Oral | |
| Injectable | ||
| Others | ||
| By Disease Indication | Relapsing-Remitting MS (RRMS) | |
| Secondary Progressive MS (SPMS) | ||
| Primary Progressive MS (PPMS) | ||
| Clinically Isolated Syndrome (CIS) | ||
| By Drug Class | Immunomodulators | |
| Monoclonal Antibodies | ||
| S1P Receptor Modulators | ||
| Antineoplastics / Others | ||
| By Distribution Channel | Hospital Pharmacy | |
| Retail Pharmacy | ||
| Online/Specialty Pharmacy | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the Multiple Sclerosis Therapeutics market growth outlook through 2031?
The multiple sclerosis therapeutics market size is projected to grow from USD 33.40 billion in 2026 to USD 39.93 billion by 2031 at a 3.64% CAGR.
Which treatment modalities are shaping near?term adoption in Multiple Sclerosis care?
Once?daily oral DMTs show higher 12?month persistence than more complex regimens, while subcutaneous monoclonal antibodies enable home administration and support earlier high?efficacy use.
Where is demand for Multiple Sclerosis therapies growing fastest regionally?
Asia?Pacific is forecast to grow at 7.22% through 2031, supported by faster national approvals and speedier access pathways in major markets.
Which mechanisms are most likely to expand options for progressive disease?
BTK inhibitors target B?cell and microglial pathways with CNS penetration and have shown promising disability outcomes in SPMS cohorts.
How are payers influencing therapy selection in the United States and Europe?
Payers increasingly tie coverage to persistence and functional outcomes, using step therapy, reference pricing, and outcomes?based contracts to align net costs with real?world value.
Which companies currently lead the global revenue mix in this space?
Biogen, Roche, Novartis, Sanofi, and Bristol Myers Squibb together held an estimated 62% share in 2025 within the multiple sclerosis therapeutics market.
Page last updated on:









