Deep Brain Stimulation Devices Market Size and Share
Deep Brain Stimulation Devices Market Analysis by Mordor Intelligence
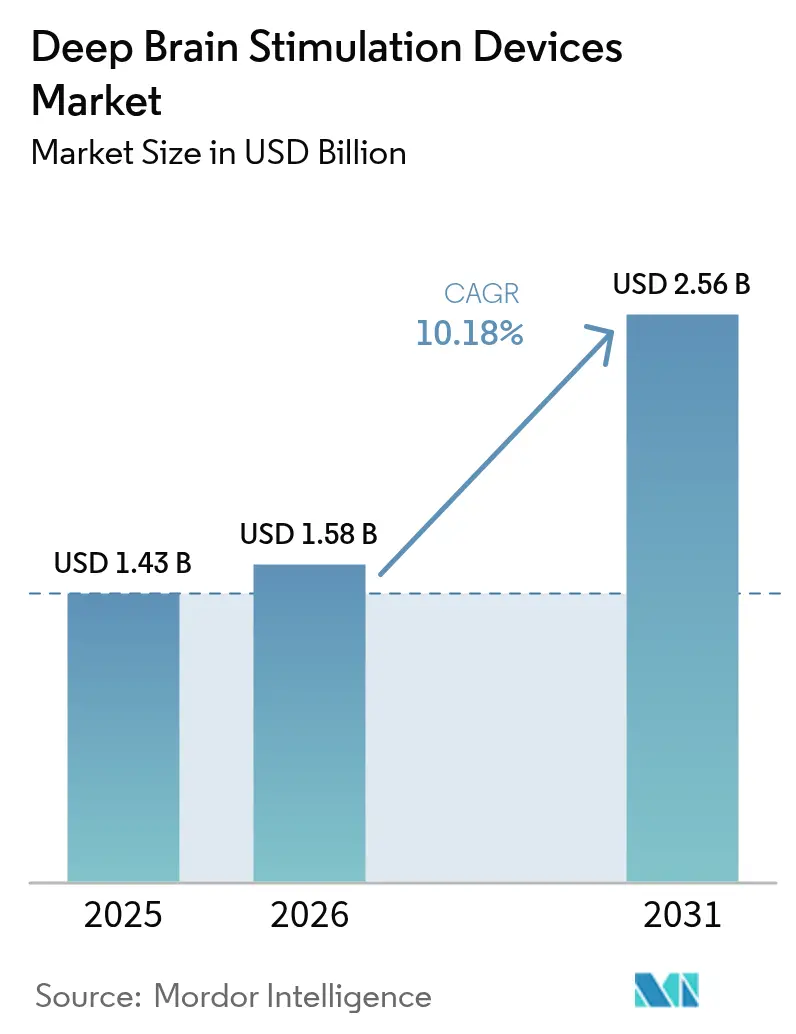
The deep brain stimulation devices market size in 2026 is estimated at USD 1.58 billion, growing from 2025 value of USD 1.43 billion with 2031 projections showing USD 2.56 billion, growing at 10.18% CAGR over 2026-2031. Rising adoption of sensing-enabled closed-loop generators, broader reimbursement for earlier-stage Parkinson’s patients, and a steady pipeline of adaptive algorithms are accelerating procedure volumes. Hospitals remain the primary implant setting, but migration toward ambulatory surgical centers is lifting capacity without comparable capital spend. Miniaturized rechargeable IPGs that last 9-15 years are lowering lifetime ownership costs, while AI-guided candidate selection tools improve responder rates and justify payor coverage. North America anchors global revenue, yet Asia-Pacific is closing the gap as surgeon-training initiatives and government neurotechnology programs scale access. Tight ^99Mo supply and post-implant infection risk temper momentum but also stimulate innovation in diagnostics and antimicrobial hardware.
Key Report Takeaways
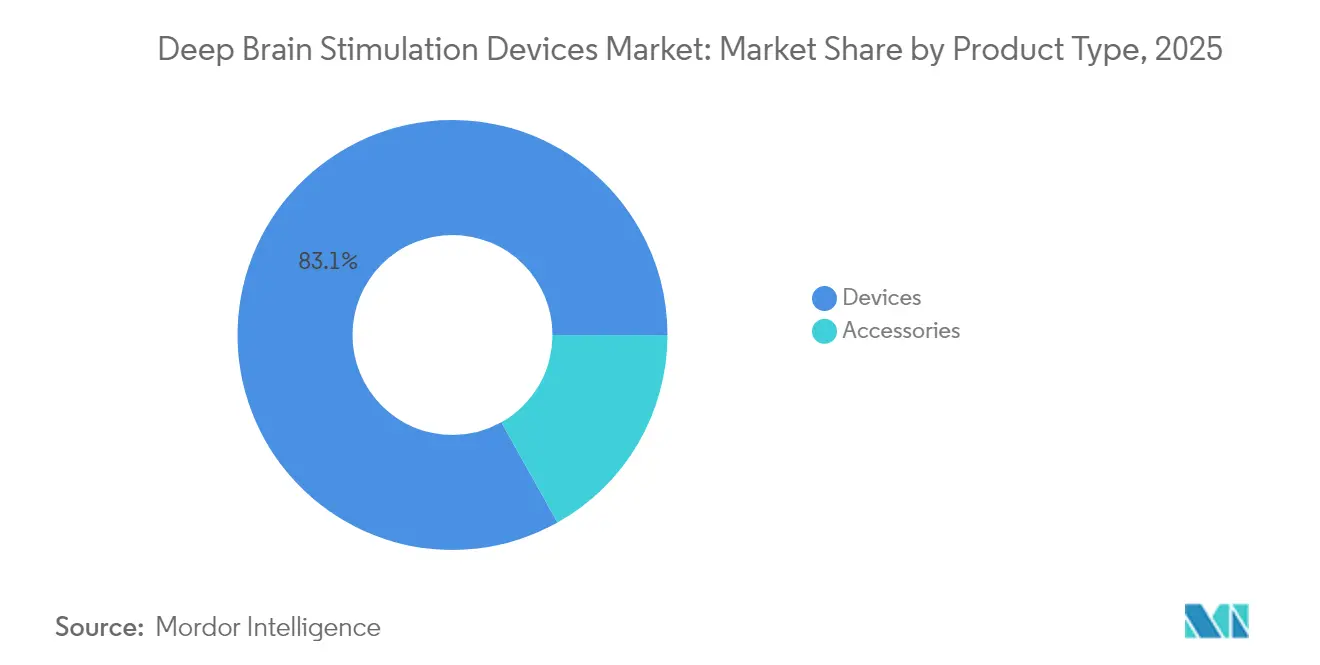
- By product type, Devices held 83.12% of deep brain stimulation devices market share in 2025, while Accessories are forecast to grow at a 10.47% CAGR to 2031.
- By application, Parkinson’s disease accounted for 61.21% share of the deep brain stimulation devices market size in 2025 and Depression is advancing at an 10.74% CAGR through 2031.
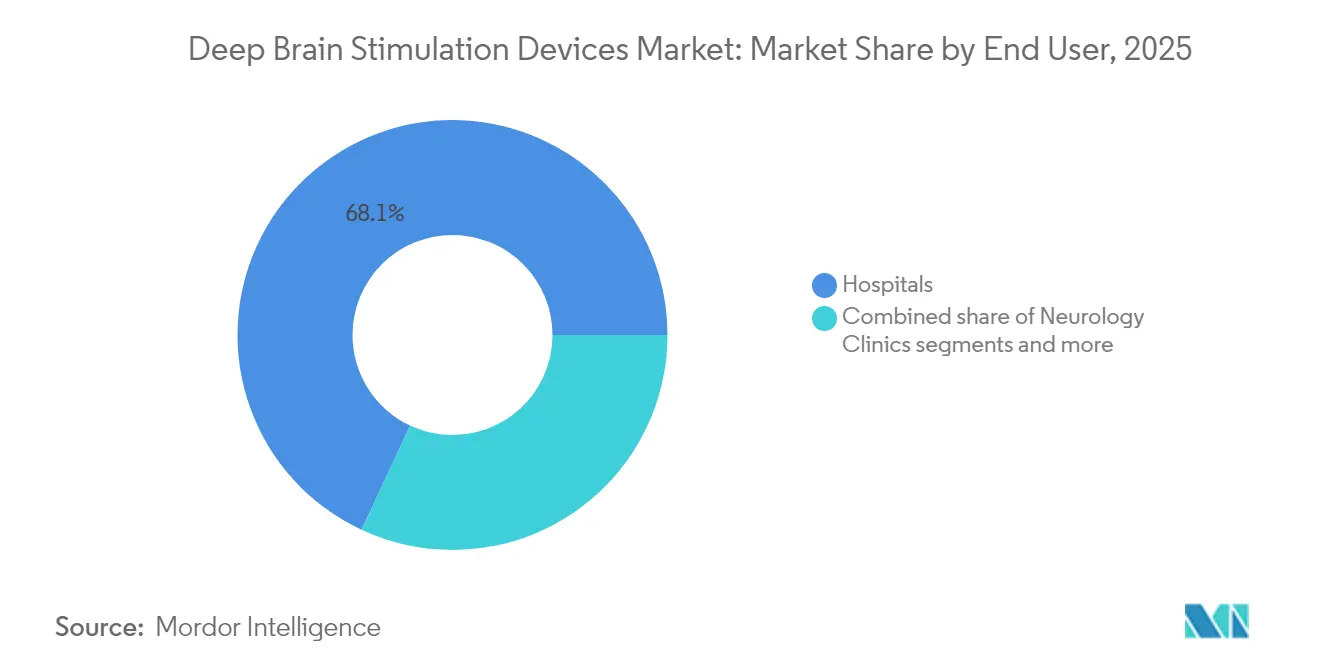
- By end user, Hospitals led with 68.05% revenue share in 2025; Ambulatory Surgical Centers are set to expand at an 10.93% CAGR through 2031.
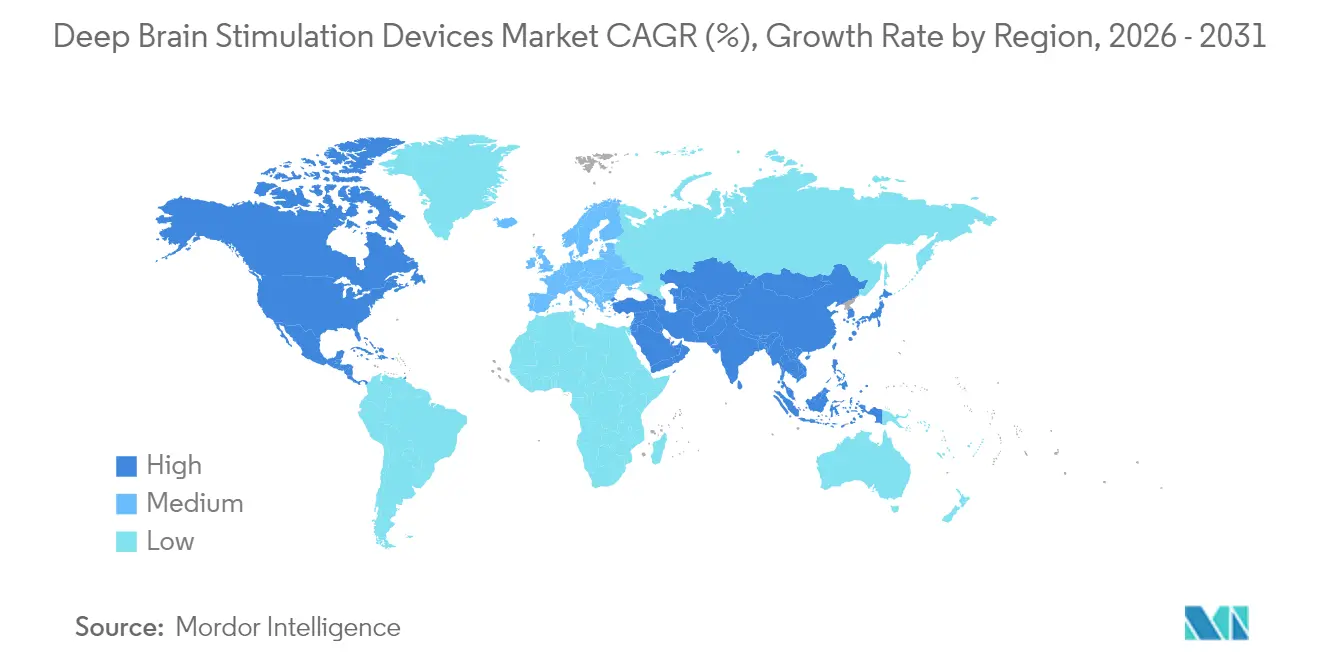
- By geography, North America captured 41.88% of deep brain stimulation devices market share in 2025, whereas Asia-Pacific records the highest projected CAGR at 11.12% to 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Deep Brain Stimulation Devices Market Trends and Insights
Driver Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rapid uptake of sensing-enabled closed-loop IPGs | +2.8% | Global, with early adoption in North America & EU | Medium term (2-4 years) |
| Reimbursement expansion for earlier-stage Parkinson's patients | +1.9% | North America & EU, expanding to APAC | Short term (≤ 2 years) |
| Surge in Asia-Pacific neurosurgeon training programs | +1.6% | APAC core, spill-over to MEA | Long term (≥ 4 years) |
| Miniaturised rechargeable IPGs lengthen replacement cycles | +1.4% | Global | Medium term (2-4 years) |
| FDA fast-track for adaptive DBS algorithms | +1.2% | Global, led by North America | Short term (≤ 2 years) |
| AI-guided patient selection improves responder rates | +1.0% | Global, concentrated in developed markets | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Rapid uptake of sensing-enabled closed-loop IPGs
BrainSense Adaptive DBS won FDA clearance in February 2025, marking the first commercial closed-loop platform that automatically titrates stimulation from real-time neural signals[1]Source: Medtronic, “Medtronic earns U.S. FDA approval for the world's first Adaptive deep brain stimulation system for people with Parkinson's,” Medtronic, news.medtronic.com . Multicenter data show median 50% motor-symptom reduction compared with open-loop systems, alongside a one-third cut in programming visits[2]Source: Parkinson’s Foundation, “New Study Further Personalizes Deep Brain Stimulation,” Parkinson’s Foundation, parkinson.org . Physicians report faster optimization curves and fewer side effects, improving patient satisfaction and health-economic value. Early adoption is strongest in U.S. and German centers where reimbursement rewards outcome-based models. Expanding indications in epilepsy and depression further widen the deep brain stimulation devices market.
Reimbursement expansion for earlier-stage Parkinson’s patients
Medicare removed the “advanced-stage only” criterion in late 2024, instantly enlarging the U.S. eligible population. Private payors aligned policies within six months, accelerating referral volumes. Comparable moves by France’s HAS and Germany’s G-BA support broader European uptake. Earlier intervention improves long-term functional status and reduces levodopa cost burden, reinforcing the deep brain stimulation devices market outlook. Manufacturers are funding outcomes registries to generate real-world evidence that sustains the policy shift.
Surge in Asia-Pacific neurosurgeon training programs
China, India, and South Korea launched VR-based surgical curricula that shorten mastery time while maintaining safety. Shanghai’s Ruijin Hospital now trains 120 fellows annually, a five-fold jump over 2023, and similar hubs in Mumbai and Seoul replicate the model. Each added neurosurgeon enables 50-75 DBS implants yearly, directly boosting regional procedure capacity. Government grants subsidize cadaver labs and simulation suites, reducing the upfront burden on hospitals. These investments underpin the deep brain stimulation devices market’s double-digit growth in Asia-Pacific.
Miniaturized rechargeable IPGs lengthen replacement cycles
Boston Scientific’s Vercise Genus battery lasts up to 15 years and recharges in under 30 minutes, cutting lifetime surgical revisions by 60%. Extended life translates to lower cumulative device spend and fewer infection opportunities, strengthening the value proposition for younger patients. Medtronic’s Percept RC combines long-life power with sensing, blunting competitive differentiation. Payors increasingly favor rechargeables, accelerating their share within the deep brain stimulation devices market.
Restraint Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Post-implant infection rates driving precautionary revisions | -1.8% | Global, higher impact in developing markets | Short term (≤ 2 years) |
| Global shortage of ^99Mo for imaging prolongs diagnosis windows | -1.2% | Global, acute in regions dependent on reactor supply | Medium term (2-4 years) |
| Cyber-security vulnerability disclosures for Bluetooth-enabled IPGs | -0.9% | Global, concentrated in connected device markets | Short term (≤ 2 years) |
| High capital cost of intra-operative imaging suites in South America | -0.7% | South America, extending to emerging markets | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Post-implant infection rates driving precautionary revisions
Hardware-related infections occur in 2.6-6.9% of implants, often requiring complete system explantation and extended antibiotic courses. Revision costs exceed USD 20,000 per episode and stall new referrals when publicized outcomes erode confidence. Summer seasonality and comorbidity prevalence escalate risk, especially in facilities lacking laminar airflow theaters. Antimicrobial-coated leads and stricter peri-operative protocols are lowering rates in high-volume U.S. centers, yet widespread uptake in emerging markets remains limited. Persistent infection fears moderate the deep brain stimulation devices market’s penetration curve.
Global shortage of ^99Mo for imaging prolongs diagnosis windows
DaTscan depends on ^99Mo supply from six aging research reactors. Outages in 2024 lengthened scheduling backlogs to eight weeks in parts of Latin America, delaying surgical clearance. FDA stability-testing rules increased production costs by USD 3 million per site, dissuading smaller compounders. Limited imaging access slows patient throughput and postpones device orders, constraining the deep brain stimulation devices market until isotope supply or alternative MRI biomarkers scale.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Product Type: Devices Retain Dominance as Accessories Accelerate
Devices generated 83.12% revenue in 2025, anchored by implantable pulse generators that integrate sensing, rechargeable batteries, and MRI safety features. Dual-channel generators now surpass single-channel units in new implants, reflecting bilateral procedure growth. Accessories—directional leads, sensing electrodes, and extensions—grow at 10.47% CAGR as closed-loop systems require specialized hardware upgrades. Replacement cycles are shorter for accessories, creating an annuity stream that triples lifetime revenue per patient. Deep brain stimulation devices market size for accessories is projected to reach USD 0.44 billion by 2031, amplified by software-based programming kits adopted by community neurologists. Competitive pressure centers on electrode geometry and material science rather than standalone pricing, protecting margins.
Technological breakthroughs in segmented leads deliver more precise targeting and reduce adverse effects, spurring conversion from legacy systems. Manufacturers bundle upgrade paths with existing generators, raising switching barriers for hospitals. Regulatory clearances in Europe and the United States for adaptive leads position accessories as the primary innovation frontier. Growth in refurbishment and resterilization services for extensions also contributes to segment revenue, especially in cost-sensitive markets. Consequently, accessories will outpace device unit growth yet remain reliant on installed generator bases within the deep brain stimulation devices market.

By Application: Parkinson’s Prevails While Depression Surges
Parkinson’s disease retained 61.21% share in 2025 owing to robust clinical evidence and payor familiarity. Average implant age has dropped from 65 to 58 years since guideline revisions, extending the revenue tail per patient. Essential tremor remains steady, but dystonia benefits from expanded offspring forms like cervical dystonia protocols. Depression is the fastest mover at 10.74% CAGR, driven by Abbott’s TRANSCEND trial and FDA breakthrough device designation for treatment-resistant disease. Deep brain stimulation devices market size for depression is forecast to triple by 2030 should pivotal data secure full approval.
Epilepsy advances through NeuroPace’s responsive neurostimulation, blurring lines between DBS and cranial-closed-loop systems and drawing new competitors into the neuromodulation arena. Obsessive-compulsive disorder remains niche but benefits from cross-disciplinary psychiatric-surgical collaborations. Application diversity mitigates revenue concentration risk while demanding nuanced regulatory and clinical strategies across indications. These trends collectively reinforce the deep brain stimulation devices market.
By End User: Hospitals Lead but Outpatient Sites Gain Traction
Hospitals captured 68.05% of 2025 revenue, leveraging integrated imaging, critical care, and multidisciplinary teams. However, streamlined surgical workflows now allow same-day discharge in selected cohorts, lifting ambulatory surgical center (ASC) appeal at an 10.93% CAGR. ASCs reduce procedure cost by up to 25% and free hospital OR slots for complex cases, a boon amid staffing shortages.
Neurology clinics handle long-term programming and follow-up through cloud-based platforms, expanding patient reach beyond metropolitan hubs. Remote programming cuts travel burden and lifts adherence, especially in rural America and Australia. Capital barriers—USD 3-5 million intra-operative MRI suites—slow ASC diffusion in South America but catalyze public-private imaging ventures. Companies offer turnkey service models that include surgical training and device logistics, smoothing the transition from hospital dominance to mixed-site delivery in the deep brain stimulation devices market.

Geography Analysis
North America remained the revenue anchor with 41.88% deep brain stimulation devices market share in 2025, bolstered by broad Medicare coverage, high neurosurgeon density, and early adoption of closed-loop systems. U.S. growth continues as outpatient implants rise, while Canada’s universality sustains steady volume albeit with tighter device margins.
Europe contributes a quarter of global sales, characterized by stable reimbursement and consistent procedure volumes across leading German, French, and Nordic centers. Adoption of rechargeable generators and directional leads is mature, yet bureaucratic procurement processes elongate replacement cycles.
Asia-Pacific leads expansion at 11.12% CAGR, propelled by China’s pricing frameworks for brain-computer interfaces and aggressive capacity building in tertiary hospitals. Japan’s aging demographics and robust research output sustain premium device demand, whereas India advances value-engineered solutions to address affordability constraints. Local manufacturing partnerships and regulatory harmonization initiatives lower entry barriers, making the region the strategic battleground for the deep brain stimulation devices market. Middle East and Africa record single-digit growth, limited by capital equipment scarcity and reimbursement gaps, yet Gulf states invest in centers-of-excellence that could seed broader adoption.
Competitive Landscape
Market concentration is moderate: Medtronic, Boston Scientific, and Abbott collectively command majority of revenue through broad portfolios and clinician-support ecosystems. Medtronic’s BrainSense set the benchmark for adaptive systems, reinforcing its premium positioning. Boston Scientific differentiates with the Vercise Genus platform’s battery longevity and directional stimulation, while Abbott targets psychiatry with the Liberta rechargeable and TRANSCEND program.
Asian challengers Beijing PINS and SceneRay gain traction in China’s public tenders via cost leadership and local service networks. European innovators such as Newronika secured CE Mark for adaptive DBS, signaling nascent competition in closed-loop algorithms. Strategic moves include Boston Scientific’s February 2025 FDA clearance for Cartesia X leads, Medtronic’s software-update business model, and Abbott’s AI-enabled programming suite.
All players emphasize evidence generation and surgeon training to create stickiness; data-driven programming ecosystems augment device features and erect switching costs. Cybersecurity disclosures around Bluetooth-enabled IPGs spur collaborative hardening efforts, adding a protective moat for incumbents that can afford security certification. Consolidation is expected as niche software or electrode suppliers seek scale within the deep brain stimulation devices market.
Deep Brain Stimulation Devices Industry Leaders
Boston Scientific Corporation
Renishaw PLC
NeuroPace Inc.
Beijing Pinchi Medical Equipment Co., Ltd.
Medtronic
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- March 2025: Newronika received CE Mark approval for its adaptive deep brain stimulation device for Parkinson’s treatment
- February 2025: Medtronic earned FDA approval for BrainSense Adaptive DBS for Parkinson’s patients
Global Deep Brain Stimulation Devices Market Report Scope
As per the scope of this report, a deep brain stimulator is a battery-operated medical device, which is also called a neurostimulator. It is used in neurosurgical procedures, to treat various disabling neurological symptoms. TThe deep brain stimulation devices market is segmented by product type (single-channel deep brain stimulator and dual-channel deep brain stimulator), by application (Parkinson's disease, essential tremor, obsessive-compulsive disorder, epilepsy, dystonia, and other applications), by end user (hospitals, ambulatory surgical centers, and other end users), and geography. (North America, Europe, Asia-Pacific, Middle East and Africa, and South America). The report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD) for the above segments.
| Device | Single-Channel Systems |
| Implantable Pulse Generators | |
| Dual-Channel Systems | |
| Accessories | Leads / Electrodes |
| Extensions & Accessories |
| Parkinson’s Disease |
| Essential Tremor |
| Dystonia |
| Epilepsy |
| Obsessive-Compulsive Disorder |
| Depression (Investigational) |
| Hospitals |
| Neurology Clinics |
| Ambulatory Surgical Centers |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| South Korea | |
| Australia | |
| Rest of Asia-Pacific | |
| South America | Brazil |
| Argentina | |
| Rest of South America | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa |
| By Product Type | Device | Single-Channel Systems |
| Implantable Pulse Generators | ||
| Dual-Channel Systems | ||
| Accessories | Leads / Electrodes | |
| Extensions & Accessories | ||
| By Application | Parkinson’s Disease | |
| Essential Tremor | ||
| Dystonia | ||
| Epilepsy | ||
| Obsessive-Compulsive Disorder | ||
| Depression (Investigational) | ||
| By End User | Hospitals | |
| Neurology Clinics | ||
| Ambulatory Surgical Centers | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| South Korea | ||
| Australia | ||
| Rest of Asia-Pacific | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||


Key Questions Answered in the Report
How large will the deep brain stimulation devices market be by 2031?
It is forecast to reach USD 2.56 billion, up from USD 1.58 billion in 2026 at a 10.18% CAGR.
Which therapeutic area grows fastest in deep brain stimulation?
Treatment-resistant depression shows the strongest trajectory with an 10.74% CAGR through 2031 on the back of pivotal trial momentum.
Why are ambulatory surgical centers gaining share in DBS procedures?
Enhanced recovery protocols and reduced overhead lower per-procedure cost by up to 25%, drawing payor support and boosting ASC volumes.
What technology shift most influences future DBS device adoption?
Sensing-enabled closed-loop generators that auto-adjust stimulation based on neural activity improve outcomes and cut clinic visits.
Which region is set to add the most new DBS capacity?
Asia-Pacific, driven by large-scale neurosurgeon training programs and supportive government neurotechnology policies.
How does battery longevity affect patient economics?
Rechargeable IPGs lasting up to 15 years slash revision surgeries by 60%, lowering lifetime costs and infection exposure.
Page last updated on:









