Liver Disease Therapeutics Market Size
Liver Disease Therapeutics Market Analysis
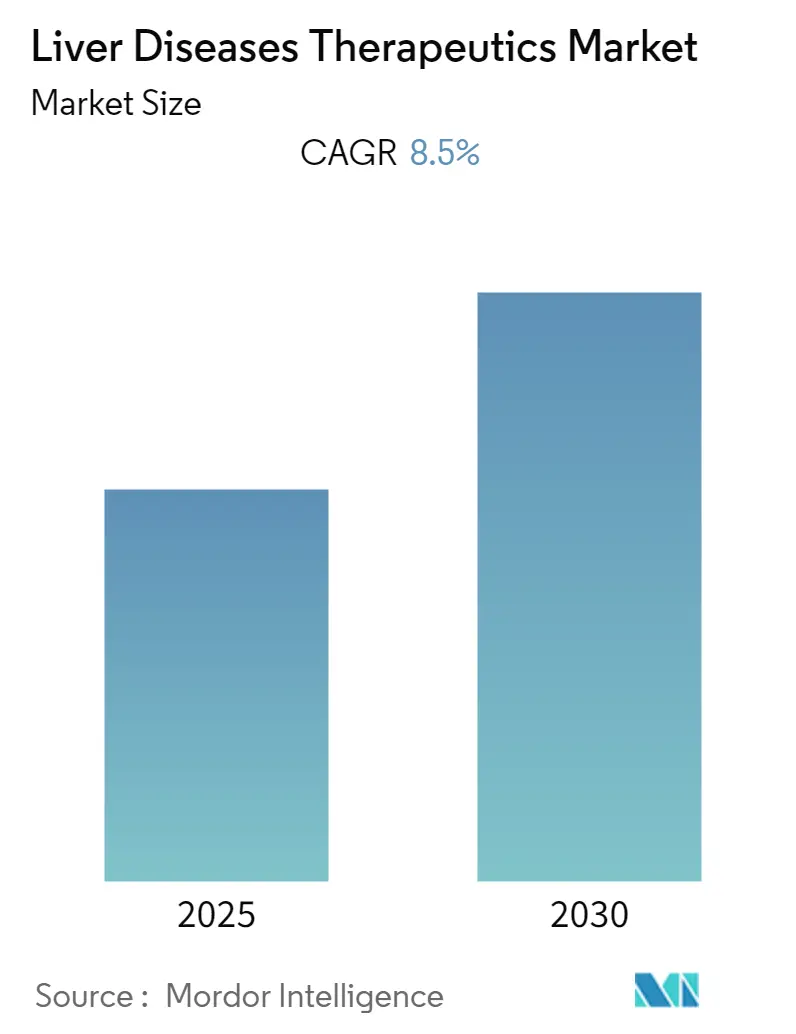
The Liver Diseases Therapeutics Market is expected to register a CAGR of 8.5% during the forecast period.
Since all resources were used to treat COVID-19 patients, it had a big effect on how other diseases were treated. Also, because of the lockdown and the high risk of infection, fewer people went to hospitals and clinics for problems other than COVID-19. This was expected to have a big effect on the market that was being studied. Reports said that COVID-19 may have had a big effect on people with liver diseases, which was good for the market for medicines to treat liver diseases. For example, a May 2021 study by the American Association for the Study of Liver Diseases (AASLD) found a strong link between cirrhosis and immune system problems.This was linked to a poor COVID-19 prognosis as well as alcoholic liver damage and cirrhosis. Also, according to an article in Frontiers in Medicine from September 2021, liver damage in COVID-19 patients can be caused by a number of things, such as systemic inflammation caused by the virus, hypoxia, hepatic congestion, and drug-induced liver disease. The same source also said that COVID-19 was linked to hepatic decompensation and liver-related death in people with cirrhosis.Hence, COVID-19 was predicted to have an effect on the liver disease therapeutics market.
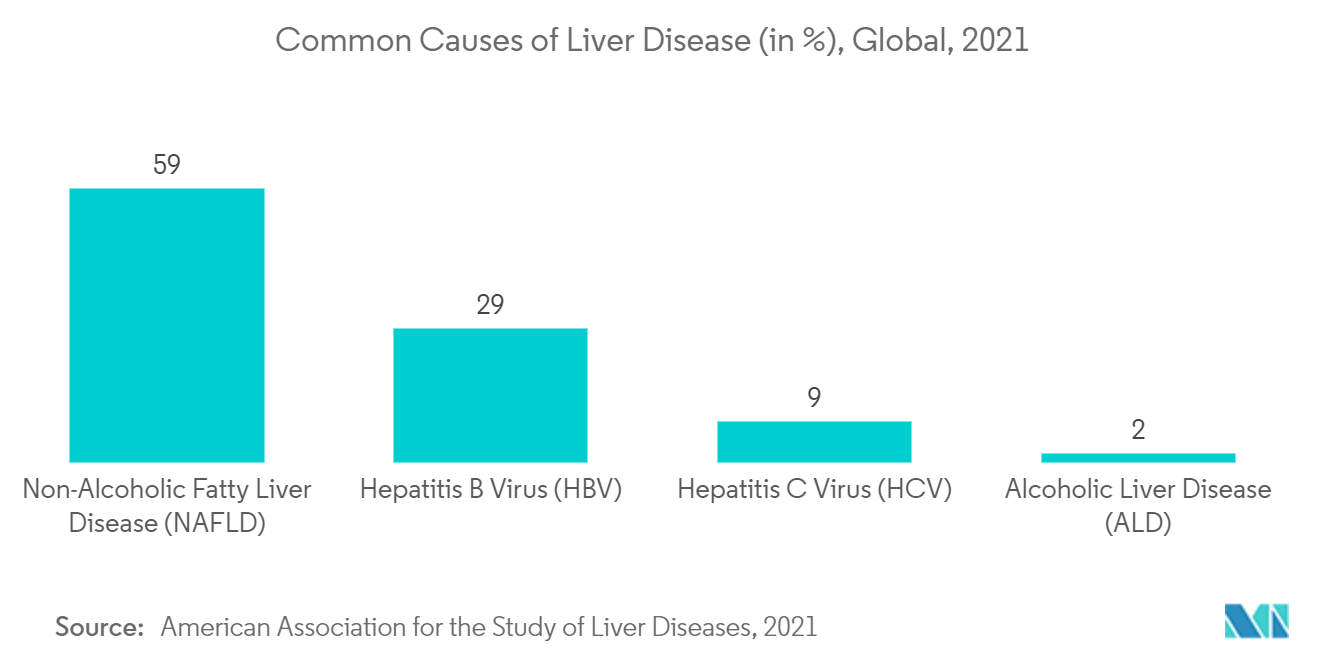
The main things that are likely to drive growth in the liver disease therapeutics market are the high number of people with liver diseases, the rise in alcohol consumption, and bad eating habits. For example, a research study published in the Clinical Liver Disease journal in June 2021 says that chronic liver disease (CLD), which affects about 1.5 billion people around the world, is one of the leading causes of death and illness around the world.Further, as per the American Association for the Study of Liver Diseases's publication in June 2021, the most common causes of prevalent disease are non-alcoholic fatty liver disease (NAFLD) (59%), followed by hepatitis B (29%), hepatitis C (9%), and alcoholic liver disease (ALD) (2%). Additionally, according to the study published in Clinical and Molecular Hepatology in October 2022, the prevalence of nonalcoholic fatty liver disease (NAFLD) is forecasted to be 55.7% by 2040, globally. Therefore, increasing liver diseases have helped drive the overall market.
Also, people who drink more alcohol and are overweight are more likely to get liver disease, which is expected to increase the demand for medicines to treat it. A study that was published in June 2022 in the journal Hepatology found that, for example, a temporary increase in alcohol consumption can greatly increase the risk of long-term illness and death from ALD.During the COVID-19 pandemic, it was also said that up to 25% more alcohol was drunk in the United States. Also, the same source says that by 2040, this increase in drinking could lead to 8,000 more deaths from alcohol-related liver disease, 18,700 cases of liver failure, and 1,000 cases of liver cancer, which is likely to increase the need for effective and advanced treatments.
Also, because liver diseases are becoming more common, the government and other non-profits play a very important role in raising awareness about them all over the world. For example, in January 2023, the International Liver Transplantation Society (ILTS), the International Living Donor Liver Transplantation Study Group (iLDLT), and the Liver Transplant Society of India (LTSI) held Asia's first consensus conference on how to care for patients who get small liver grafts in India. Hence, all these organizations are found promoting awareness of liver-related diseases and support for research and development for the prevention, treatment, and cure of liver diseases. So, the main things that are driving the growth of the market during the study period are the rise in liver diseases around the world, the rise in the number of people who drink too much and eat poorly, and the rise in government efforts to raise awareness about liver diseases.
However, the stringent regulations and the complications associated with the drugs are expected to hinder market growth during the study period.
Liver Disease Therapeutics Market Trends
The Anti Viral Drugs Segment is Expected to Dominate the Market During the Forecast Period
Because more people are getting liver diseases and drinking more alcohol, the need for medicines to treat liver diseases has grown a lot.
Antiviral drugs are a type of medicine used to treat hepatitis. They work by lowering the amount of HBV (hepatitis B virus) in the body and lowering the risk of getting cirrhosis and hepatocellular cancer.Some of the FDA-approved drugs include Mavyret, Zepatier, and Vosevi. These drugs have been widely used for many years. They are safe and effective, especially for patients with signs and symptoms of moderate-to-severe liver impairment or other serious liver problems.
The high incidence of virus-infected liver diseases, including hepatitis, and the efficacy of antiviral treatment are the major factors contributing to the growth of this segment in the studied market. Chronic hepatitis B virus (HBV) infection is still a serious public health concern globally because it can cause cirrhosis and hepatocellular carcinoma (HCC), which increase human mortality and morbidity. Antiviral drugs are often taken to suppress the hepatitis B virus and long-term complications associated with hepatitis B, including liver disease and impairment. As per the study published in the May 2021 issue of BMC Infectious Diseases, the direct-acting antiviral (DAA) drug treatment demonstrated a positive prognostic factor for liver function improvement in HCV (hepatitis C virus) patients. Furthermore, the research published in the August 2022 issue of BMC Gastroenterology demonstrated the effectiveness of antiviral treatment in HBeAg-negative chronic hepatitis B patients with normal or mildly elevated alanine aminotransferase (ALT).
Therefore, the market for antiviral drugs has a significant market share and is anticipated to retain its dominance during the forecast period due to the rising incidence of virally-infected liver diseases and expanding research indicating the effectiveness of antiviral medication therapy.
North America is Expected to Dominate the Liver Disease Therapeutics Market
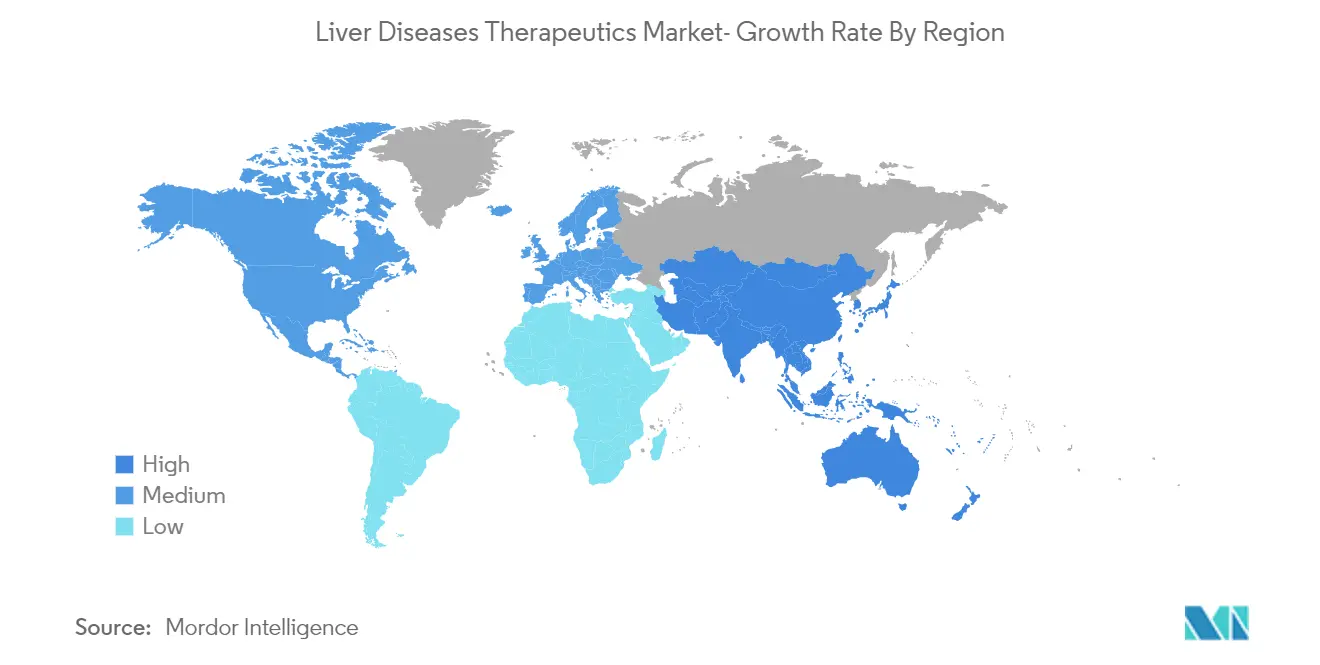
North America is the biggest market because that is where most clinical trial studies are done by research and academic institutions.Changes in lifestyles, like drinking more alcohol and eating less healthily, have also made liver disease more common.Over the next few years, the regional market is expected to grow because patients are becoming more aware of their health and because new products are being made to meet unmet medical needs.
There has been an increase in the incidence of liver diseases in the region. According to the Centers for Disease Control and Prevention, as of January 2023, 4.5 million adults were diagnosed with liver disease in the United States. Further, as per the same source, alcohol consumption is also very high in the country, which is one of the major factors responsible for the increase in the burden of liver disease, and about 25.1% of adults age 18 and older had approximately one heavy drinking day (five or more drinks for men and four or more drinks for women) in the previous years. Hence, the burden of liver diseases in the country is expected to increase over the years, which is anticipated to have a positive impact on the market.
Therefore, due to the rising prevalence of liver diseases, the management of liver diseases has evolved rapidly. To cater to the significant unmet needs in the market, players are focusing on research and development with potential candidates, primarily related to preventing liver disease progression and reducing anticipated liver morbidity and mortality. For instance, in December 2022, Altimmune, Inc. announced positive topline results from its 24-week (12-week extension) trial of Pemvidutide for non-alcoholic fatty liver disease, showing the potential to dramatically lower liver fat content and liver inflammation. Moreover, in January 2022, Madrigal Pharmaceuticals, Inc. announced positive topline clinical data from the placebo-controlled, double-blind version of its phase 3 MAESTRO-NAFLD (Non-Alcoholic Fatty Liver Disease) safety study of Resmetirom.
Hence, the growing prevalence rates of liver diseases and the introduction of new products targeting unmet medical needs are expected to drive regional market growth over the forecast period.
Liver Disease Therapeutics Industry Overview
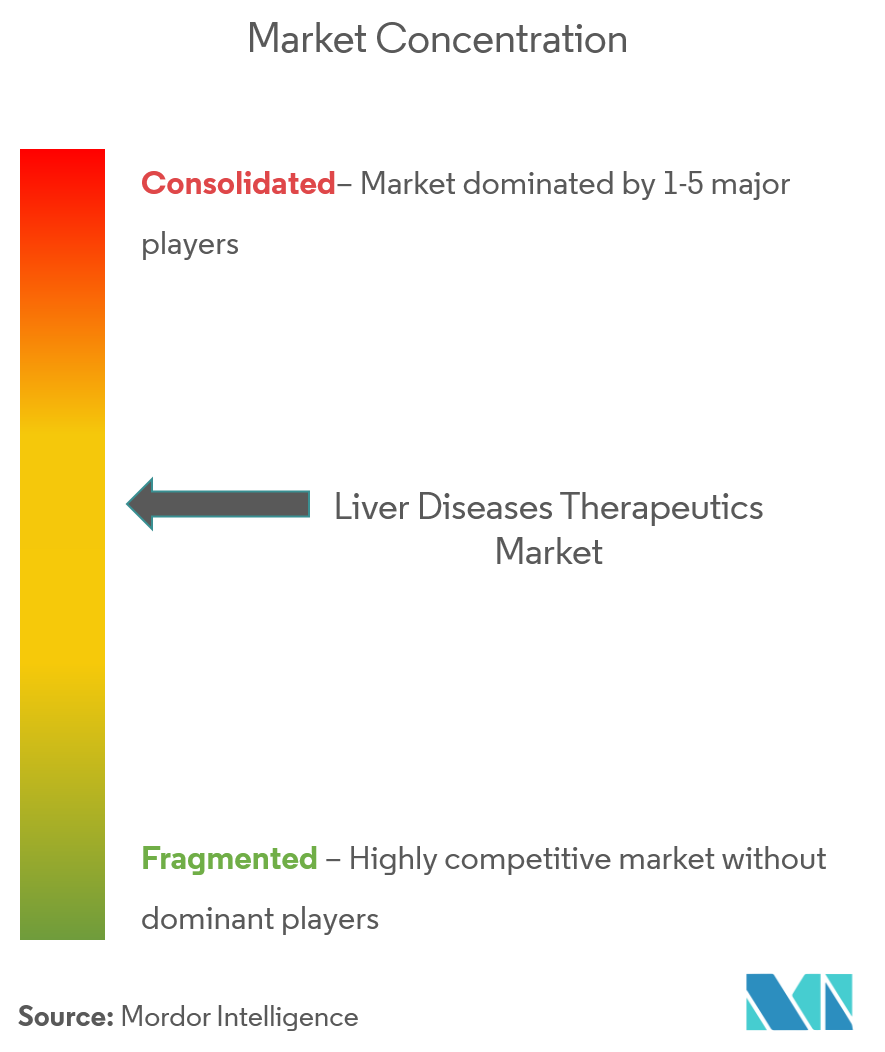
The liver disease therapeutics market is fragmented, with many prominent players. In terms of market share, a few of the major players are currently dominating the market. Some of the companies that are currently dominating the market are Abbott Laboratories, Gilead Sciences, Novartis AG, Sanofi, and Astellas Pharma, Inc., among others.
Liver Disease Therapeutics Market Leaders
-
Abbott Laboratories
-
Gilead Sciences
-
Novartis AG
-
Sanofi
-
Astellas Pharma Inc.
- *Disclaimer: Major Players sorted in no particular order
Liver Disease Therapeutics Market News
- January 2023: Takeda and Arrowhead Pharmaceuticals Inc. announced topline results from the Phase 2 SEQUOIA clinical study of investigational Fazirsiran for the treatment of liver disease associated with alpha-1 antitrypsin deficiency (AATD-LD).
- January 2022: Merck received conditional liver cancer treatment approval for its drug Keytruda based on its second confirmatory study. Keytruda slashed the risk of death by 21% over placebo in hepatocellular carcinoma patients in Asia.
Liver Disease Therapeutics Industry Segmentation
As per the scope of the report, liver disease is either inherited or caused by factors that damage the liver, such as viruses and the use of alcohol. Liver disease, if not treated, damages the liver and further leads to liver failure. Therefore, some of the major drugs used in treating various types of liver diseases include vaccines, immunosuppressants, chemotherapy drugs, and anti-viral drugs. The liver disease therapeutics market is segmented by treatment type (anti-viral drugs, targeted therapy, immunosuppressant drugs, chemotherapy drugs, immunoglobulin, vaccines, and other treatment types), end user (hospitals, ambulatory surgery centers, and other end users), and geography (North America, Europe, Asia-Pacific, the Middle East and Africa, and South America). The market report also covers the estimated market sizes and trends for 17 different countries across major regions globally. The report offers the value (in USD million) for the above segments.
| By Treatment Type | Anti Viral Drugs | ||
| Targeted Therapy | |||
| Immunosuppressant Drugs | |||
| Chemotherapy Drugs | |||
| Immunoglobulin | |||
| Vaccines | |||
| Other Treatment Types | |||
| By End-User | Hospitals | ||
| Ambulatory Surgery Centers | |||
| Other End-Users | |||
| Geography | North America | United States (By Treatment Type and End-User) | |
| Canada (By Treatment Type and End-User) | |||
| Mexico (By Treatment Type and End-User) | |||
| Europe | Germany (By Treatment Type and End-User) | ||
| United Kingdom (By Treatment Type and End-User) | |||
| France (By Treatment Type and End-User) | |||
| Italy (By Treatment Type and End-User) | |||
| Spain (By Treatment Type and End-User) | |||
| Rest of Europe (By Treatment Type and End-User) | |||
| Asia-Pacific | China (By Treatment Type and End-User) | ||
| Japan (By Treatment Type and End-User) | |||
| India (By Treatment Type and End-User) | |||
| Australia (By Treatment Type and End-User) | |||
| South Korea (By Treatment Type and End-User) | |||
| Rest of Asia-Pacific (By Treatment Type and End-User) | |||
| Middle-East and Africa | GCC (By Treatment Type and End-User) | ||
| South Africa (By Treatment Type and End-User) | |||
| Rest of Middle-East and Africa (By Treatment Type and End-User) | |||
| South America | Brazil (By Treatment Type and End-User) | ||
| Argentina (By Treatment Type and End-User) | |||
| Rest of South America (By Treatment Type and End-User) | |||
| Anti Viral Drugs |
| Targeted Therapy |
| Immunosuppressant Drugs |
| Chemotherapy Drugs |
| Immunoglobulin |
| Vaccines |
| Other Treatment Types |
| Hospitals |
| Ambulatory Surgery Centers |
| Other End-Users |
| North America | United States (By Treatment Type and End-User) |
| Canada (By Treatment Type and End-User) | |
| Mexico (By Treatment Type and End-User) | |
| Europe | Germany (By Treatment Type and End-User) |
| United Kingdom (By Treatment Type and End-User) | |
| France (By Treatment Type and End-User) | |
| Italy (By Treatment Type and End-User) | |
| Spain (By Treatment Type and End-User) | |
| Rest of Europe (By Treatment Type and End-User) | |
| Asia-Pacific | China (By Treatment Type and End-User) |
| Japan (By Treatment Type and End-User) | |
| India (By Treatment Type and End-User) | |
| Australia (By Treatment Type and End-User) | |
| South Korea (By Treatment Type and End-User) | |
| Rest of Asia-Pacific (By Treatment Type and End-User) | |
| Middle-East and Africa | GCC (By Treatment Type and End-User) |
| South Africa (By Treatment Type and End-User) | |
| Rest of Middle-East and Africa (By Treatment Type and End-User) | |
| South America | Brazil (By Treatment Type and End-User) |
| Argentina (By Treatment Type and End-User) | |
| Rest of South America (By Treatment Type and End-User) |
Liver Disease Therapeutics Market Research FAQs
What is the current Liver Diseases Therapeutics Market size?
The Liver Diseases Therapeutics Market is projected to register a CAGR of 8.5% during the forecast period (2025-2030)
Who are the key players in Liver Diseases Therapeutics Market?
Abbott Laboratories, Gilead Sciences, Novartis AG, Sanofi and Astellas Pharma Inc. are the major companies operating in the Liver Diseases Therapeutics Market.
Which is the fastest growing region in Liver Diseases Therapeutics Market?
Asia-Pacific is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Liver Diseases Therapeutics Market?
In 2025, the North America accounts for the largest market share in Liver Diseases Therapeutics Market.
What years does this Liver Diseases Therapeutics Market cover?
The report covers the Liver Diseases Therapeutics Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Liver Diseases Therapeutics Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on: June 16, 2024
Our Best Selling Reports
Liver Cirrhosis Treatment Industry Report
Statistics for the 2025 Liver Diseases Therapeutics market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Liver Diseases Therapeutics analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.