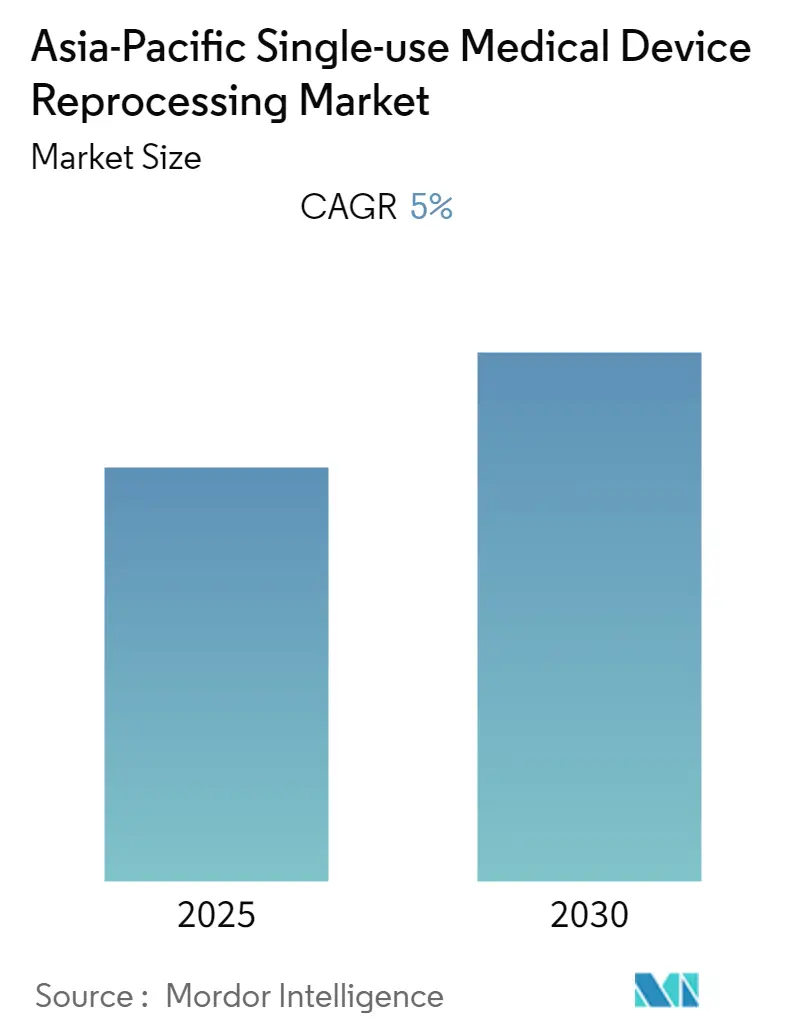
Asia-Pacific Single-use Medical Device Reprocessing Market Analysis by Mordor Intelligence
The Asia-Pacific Single-use Medical Device Reprocessing Market is expected to register a CAGR of 5% during the forecast period.
The COVID-19 pandemic negatively affected the single-use medical reprocessing market due to the impact of reprocessing activities. Many business activities were affected worldwide due to COVID-19 restrictions, which led to a decline in market capitalization and businesses. But now, conditions are improving with the vaccination process, and the market is expected to be at its pre-COVID era status. The COVID-19 pandemic led to a massive surge in various medical devices and instruments, such as ventilators, oxygen concentrators, pulse oximeters, suction pumps, central venous catheters, and thermocyclers. It has also created an opportunity for the studied market in the future.
Factors such as cost savings through reprocessing single-use medical devices, increasing regulatory pressure to reduce the volume of medical waste, the rising burden of chronic diseases, and the growing geriatric population will boost the market studied. Also, the new activities launched in the market and the key strategies adopted, such as mergers and acquisitions, are expected to increase the studied market. As per the research article published in December 2021 by Environmental Health, the healthcare system of high-income countries mostly depends on a linear supply chain that contains single-use disposable medical devices. It increases healthcare expenditure and creates other problems, such as waste and pollution. It indirectly but severely affects the surrounding ecosystem. Thus, reprocessing medical devices reduces the amount of medical waste generated and propels the market growth.
Additionally, growing demands for recycling and reusability due to factors such as rising awareness among the public and campaigns from various NGOs working to reduce medical waste and increase the recycling measure are also expected to boost the growth of the single-use medical device reprocessing market during the forecast period. Thus, due to the factors mentioned above, the studied market is expected to grow significantly during the study period.
However, concerns such as potential material alteration during reprocessing, cross infection with reprocessed devices, and a general notion regarding the quality of reprocessed single-use medical devices among healthcare providers will impede the growth of the studied market.
Asia-Pacific Single-use Medical Device Reprocessing Market Trends and Insights
Sequential Compression Sleeves Segment is Expected to Witness Growth Over the Forecast Period
Compression sleeves are inflatable ones that connect to an air pump to augment blood flow in a limb for blood clot prevention. These devices will record a large share owing to the increasing number of interventions and the consequent economic load demand for assessing SUDs' reuse. Japan does not have any regulations for the reprocessing of single-use medical devices. Therefore, regulatory authorities, such as the MHLW (Japan), in collaboration with the FDA, are focusing on generating new medical instrument reprocessing guidelines for Japan. Therefore all these factors are expected to drive the overall market.
Sequential compression sleeves are used widely worldwide due to the high burden of diseases such as deep vein thrombosis and pulmonary embolism. These diseases will further increase over the years. As per the study published in March 2021 by the Indian Journal of Vascular and Endovascular Surgery, there were 518,111 admissions, with a VTE incidence of 19.49/10,000 cases. Males contributed to 55% of patients in this study. Acute deep-vein thrombosis (DVT) (less than 14 days of presentation) contributed to 66% of the patients. The age group between 41 and 60 years had the maximum incidence of DVT of 43.9%. Hence, the class II device segment will grow due to the rising medical device demand.
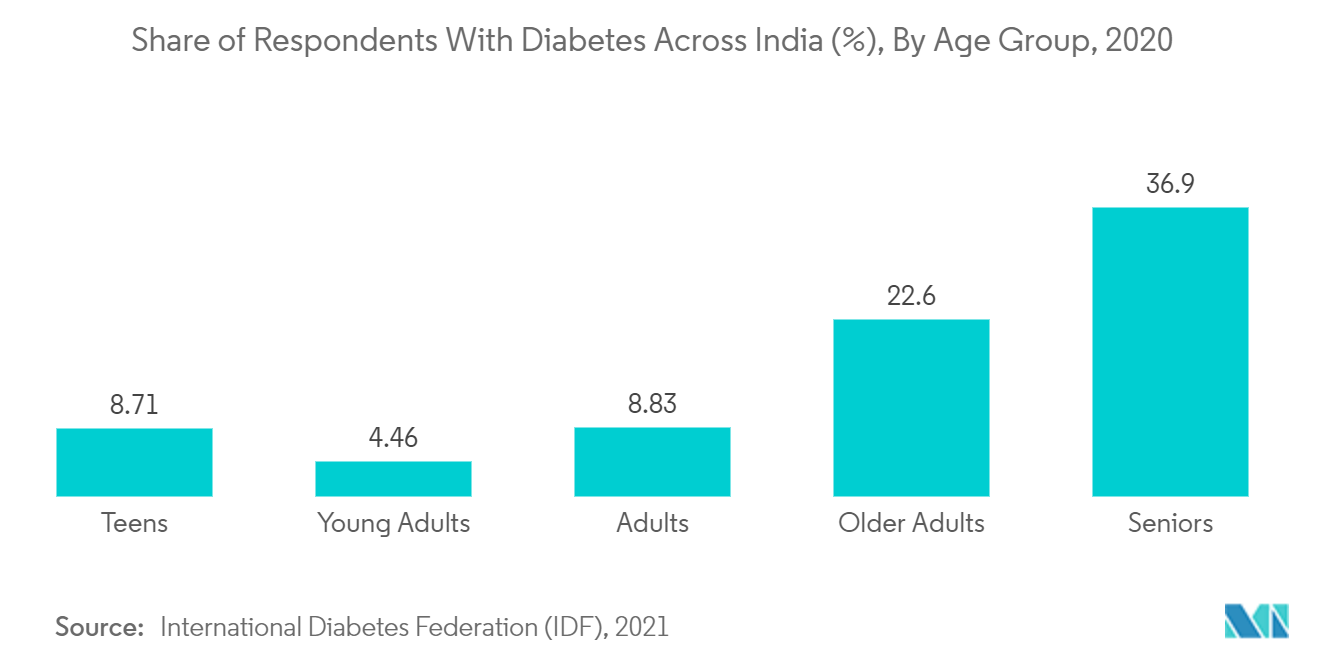


China is Expected to Witness Growth Over the Forecast Period
China holds a significant share in the single-use medical device reprocessing market and is expected to show a similar trend over the forecast period, mainly due to the increasing cost of reusable medical devices and the rising prevalence of cardiovascular diseases in the region.
Increasing cases of cardiovascular diseases and related surgical procedures are the primary growth factors. CVD is the leading cause of healthcare burden in China. The prevalence and incidence of the disease are increasing with time. The research article published in April 2022 by the International Journal of Public Health mentioned that CVD is one of the significant healthcare burdens in China. And as per the article, currently, 290 million people suffering from cardiac disorders (13 million), Stroke, coronary heart disease (11 million), rheumatic heart disease (2.5 million), heart failure (4.5), congenital heart disease (2 million), pulmonary heart disease (5 million), and hypertension (245 million) in China. Also, the source mentioned above that out of the total mortality related to CVD, 40% occurs in China alone. The prevalence of cardiac diseases generates a need for cardiovascular device monitoring and diagnostic devices. This high prevalence of diseases requiring class I and II devices will significantly boost the market's growth.
Thus, due to the factors mentioned above, the studied market is expected to grow significantly during the study period.


Competitive Landscape
The Asia-Pacific Single-Use Medical Devices Reprocessing Market is fragmented and moderately competitive. The major players in the Asia-Pacific single-use medical device reprocessing market include Medline Industries, Inc., NEScientific, Inc., ReNu Medical Inc., Soma Technology, Inc., Sterilmed Inc. (Johnson & Johnson), and Stryker Corporation.
Asia-Pacific Single-use Medical Device Reprocessing Industry Leaders
Medline Industries, Inc.,
NEScientific, Inc.
ReNu Medical Inc.
Sterilmed Inc. (Johnson & Johnson)
Stryker Corporation
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- August 2022- Ecolab Inc. expanded its Healthcare division to serve hospitals and healthcare facilities in Southeast Asia. A leader in infection prevention solutions and services, Ecolab Healthcare, offers a comprehensive array of hygiene and infection prevention solutions for hospitals, surgical centers, and healthcare facilities. They also provide instrument & endoscopy reprocessing, surgical drapes, and environmental hand hygiene.
- April 2022- The 'state-of-the-art' Central Sterile Services Department (CSSD) of AIIMS Bhubaneswar received 2nd prize under the Government Health Care Organization category in the Consortium of Accredited Health Care Organizations Awareness Compliances and Excellence (CAHO-ACE) program for the year 2021.


Asia-Pacific Single-use Medical Device Reprocessing Market Report Scope
As per the scope of the report, reprocessing single-use medical devices (SUDs) is an exercise of inspecting, cleaning, function testing, sterilizing, and packaging to clinically and carefully use them again. A reprocessed single-use device is an original device that has previously been used and subjected to additional processing and manufacturing for a further single use on a patient. The reuse of single-use devices has been increasing as a cost-saving measure. The Asia-Pacific Single-use Medical Device Reprocessing Market is Segmented by Device Type (Class I Devices (Laparoscopic Graspers, Scalpels, Tourniquet Cuffs, and Other Class I Devices) and Class II Devices (Pulse Oximeter Sensors, Sequential Compression Sleeves, Catheters and Guidewires, and Other Class II Devices)) and Geography (China, Japan, India, Australia, South Korea, and Rest of Asia-Pacific). The report offers the value in (USD million) for the above segments.
| Class I Devices | Laparoscopic Graspers |
| Forceps | |
| Scissors | |
| Other Class I Devices | |
| Class II Device | Pulse Oximeter Sensors |
| Sequential Compression Sleeves | |
| Catheters and Guidewires | |
| Other Class II Devices |
| China |
| Japan |
| India |
| Australia |
| South Korea |
| Rest of Asia-Pacific |
| By Device Type | Class I Devices | Laparoscopic Graspers |
| Forceps | ||
| Scissors | ||
| Other Class I Devices | ||
| Class II Device | Pulse Oximeter Sensors | |
| Sequential Compression Sleeves | ||
| Catheters and Guidewires | ||
| Other Class II Devices | ||
| Geography | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||


Key Questions Answered in the Report
What is the current Asia-Pacific Single-use Medical Device Reprocessing Market size?
The Asia-Pacific Single-use Medical Device Reprocessing Market is projected to register a CAGR of 5% during the forecast period (2025-2030)
Who are the key players in Asia-Pacific Single-use Medical Device Reprocessing Market?
Medline Industries, Inc.,, NEScientific, Inc., ReNu Medical Inc., Sterilmed Inc. (Johnson & Johnson) and Stryker Corporation are the major companies operating in the Asia-Pacific Single-use Medical Device Reprocessing Market.
What years does this Asia-Pacific Single-use Medical Device Reprocessing Market cover?
The report covers the Asia-Pacific Single-use Medical Device Reprocessing Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Asia-Pacific Single-use Medical Device Reprocessing Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
Asia-Pacific Single-use Medical Device Reprocessing Market Report
Statistics for the 2025 Asia-Pacific Single-use Medical Device Reprocessing market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Asia-Pacific Single-use Medical Device Reprocessing analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.





