Waldenstrom's Macroglobulinemia (WM) Treatment Market Size and Share
Waldenstrom's Macroglobulinemia (WM) Treatment Market Analysis by Mordor Intelligence
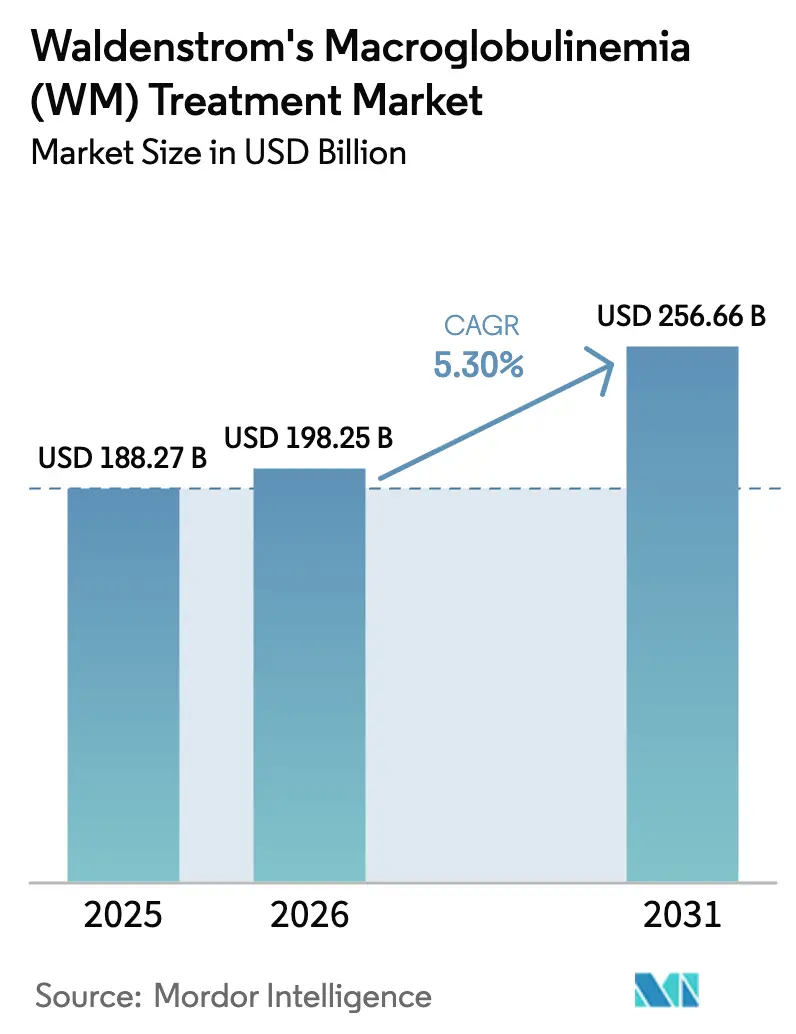
The Waldenstrom's Macroglobulinemia Treatment Market size is projected to expand from USD 188.27 billion in 2025 and USD 198.25 billion in 2026 to USD 256.66 billion by 2031, registering a CAGR of 5.30% between 2026 to 2031.
The increasing prevalence and incidence of cases of Waldenstrom's macroglobulinemia and increasing drug approvals by the regulatory bodies are escalating the growth of the market. Regulatory advancements are driving momentum in the industry. In January 2024, the FDA granted accelerated approval to zanubrutinib for previously treated patients. This was followed by the FDA's acceptance of BeiGene's supplemental biologics license application in October 2025, expanding the drug's use to frontline therapy. These developments collectively improve access to therapy and streamline treatment initiation. Additionally, precision diagnostics are optimizing care pathways. The routine detection of the MYD88 L265P mutation now informs BTK inhibitor selection in academic settings, accelerating time-to-treatment and reducing cycles of less effective therapies.
Key Report Takeaways
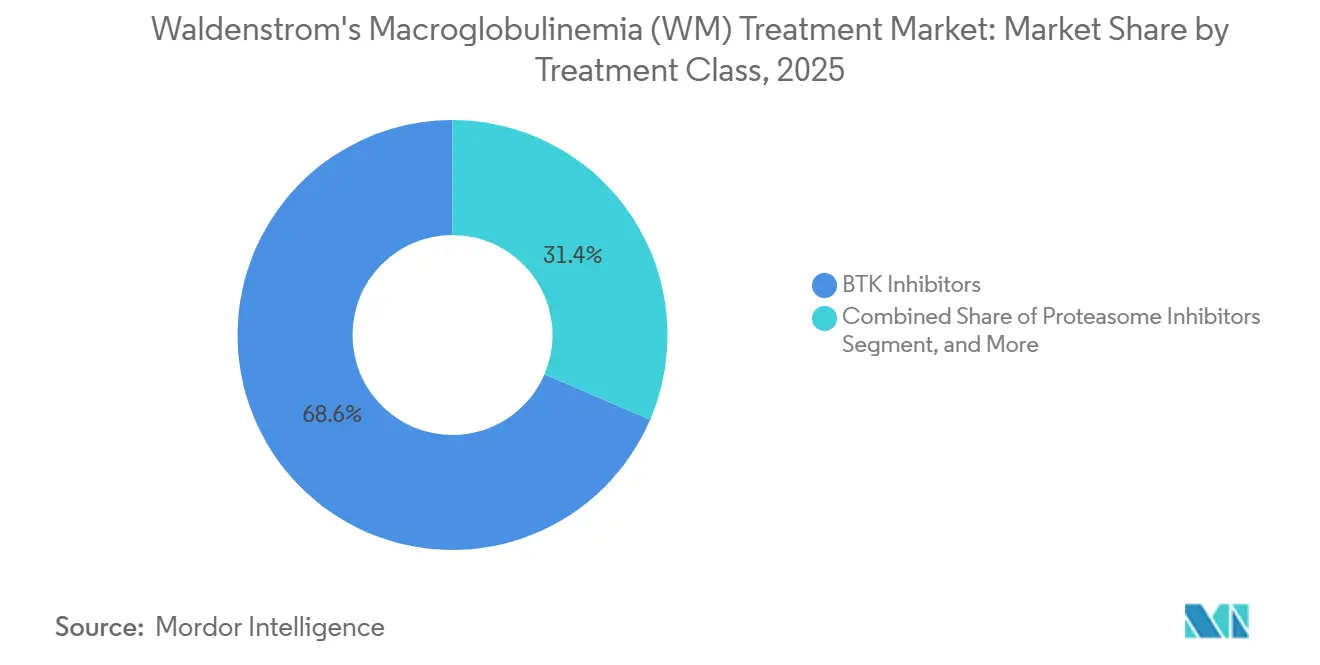
- By treatment class, BTK inhibitors led with 68.56% revenue share in 2025, while proteasome inhibitors are forecast to expand at a 5.87% CAGR through 2031.
- By line of therapy, first-line held 55.45% revenue share in 2025, while second-line is projected to grow at a 6.39% CAGR through 2031.
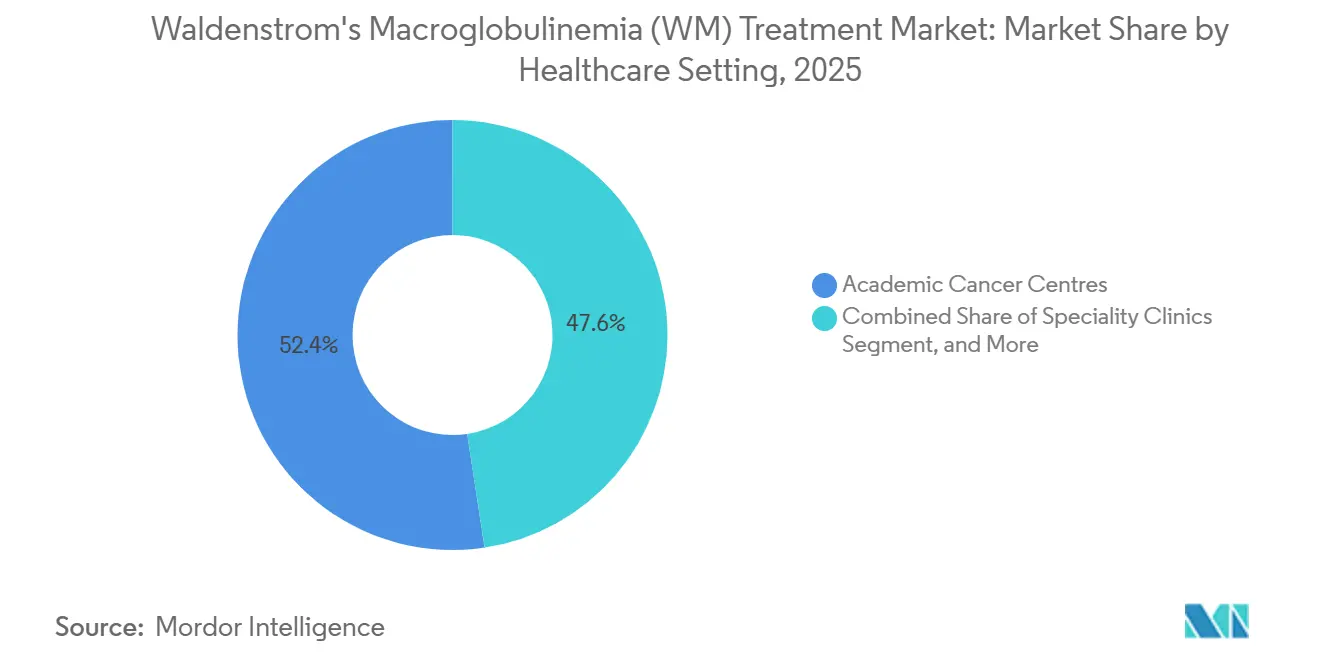
- By healthcare setting, academic cancer centers accounted for 52.37% revenue share in 2025, while specialty clinics are forecast to expand at a 6.80% CAGR through 2031.
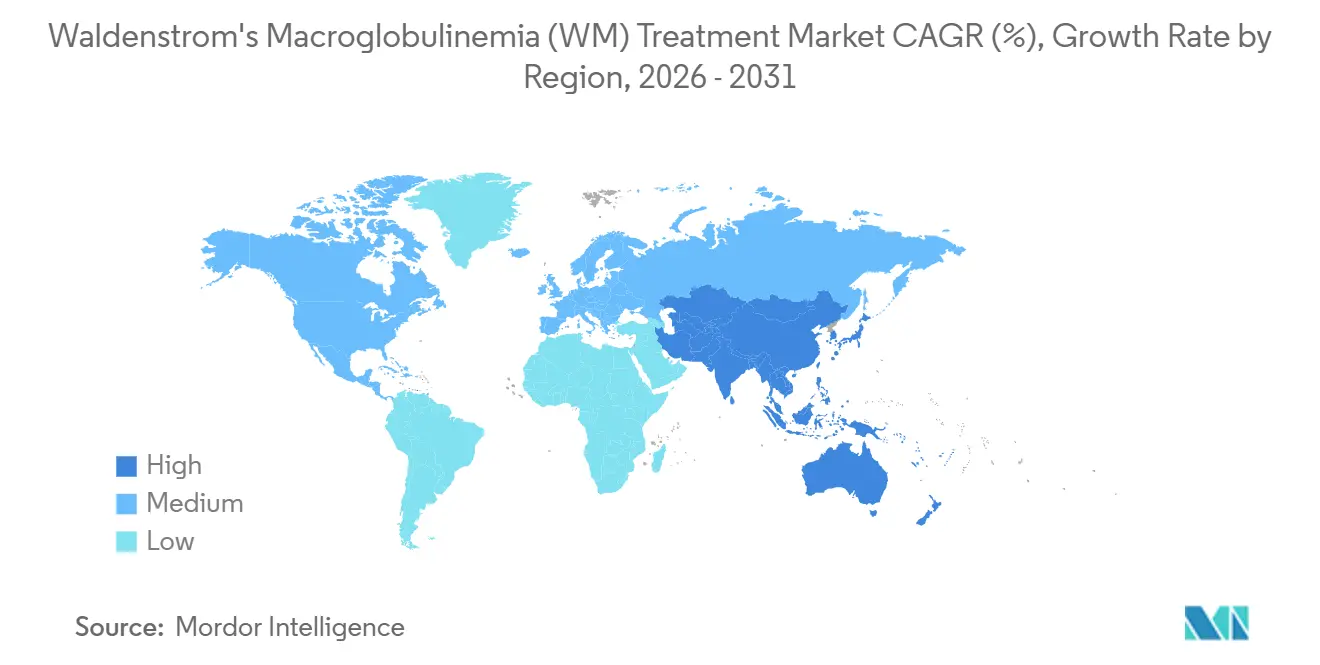
- By geography, North America held 45.62% in 2025, while Asia-Pacific is projected to advance at a 6.15% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Waldenstrom's Macroglobulinemia (WM) Treatment Market Trends and Insights
Drivers Impact Analysis
| DRIVER | (~) % IMPACT ON CAGR FORECAST | GEOGRAPHIC RELEVANCE | IMPACT TIMELINE |
|---|---|---|---|
| Growing diagnosed prevalence via next-gen genomic testing | +0.9% | Global, with North America and Europe leading adoption | Short term (≤ 2 years) |
| FDA/EMA approvals & label expansions of BTK inhibitors | +1.2% | North America, Europe, Asia-Pacific (China, Japan) | Medium term (2-4 years) |
| Ageing population enlarging treatment-eligible cohort | +0.7% | Global, particularly North America, Europe, Japan | Long term (≥ 4 years) |
| Non-covalent BTK degraders addressing resistance gaps | +0.8% | North America, Europe, select Asia-Pacific markets | Medium term (2-4 years) |
| Liquid-biopsy MRD tools triggering earlier intervention | +0.6% | North America, Europe, Australia | Short term (≤ 2 years) |
| Advocacy-funded trial acceleration in rare haematologics | +0.4% | Global, with concentration in North America and Europe | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Growing Diagnosed Prevalence Via Next-Gen Genomic Testing
Next-generation sequencing panels that detect MYD88 L265P and CXCR4 mutations reduced the median time-to-diagnosis from 14.3 months in 2020 to 6.8 months in 2025. This advancement expanded the patient population eligible for treatment and enabled earlier intervention for high-risk individuals. In March 2024, Medicare coverage was extended to Foundation Medicine’s FoundationOne Heme test. This test sequences 406 genes and, with significant payment support towards its USD 5,800 list price, has driven increased adoption in community settings, promoting standardized testing practices. Launched in September 2024, Illumina’s TruSight Oncology 500 assay offers a 7-day turnaround time, expediting decision-making compared to traditional methods and facilitating the timely initiation of BTK inhibitors.[1]Illumina, “TruSight Oncology 500 Product Overview,” Illumina, illumina.com Additionally, the European Medicines Agency’s 2024 directive mandated MYD88 mutation testing before prescribing BTK inhibitors, making it a regulatory requirement across the region.[2]European Medicines Agency, “Guidance on MYD88 Testing Prior to BTK Inhibitor Use,” European Medicines Agency, ema.europa.eu These advancements have redefined the diagnostic framework for the Waldenström's Macroglobulinemia market and strengthened the adoption of molecularly guided therapy selection across treatment settings.
FDA/EMA Approvals & Label Expansions of BTK Inhibitors
In January 2024, the FDA granted accelerated approval to zanubrutinib for previously treated WM, expanding access to this selective BTK inhibitor, recognized for its favorable tolerability in routine clinical practice. Similarly, the EMA issued a conditional marketing authorization in May 2024, enabling reimbursement across 27 EU member states and improving patient access within hospital formularies. In October 2025, Acalabrutinib received FDA priority review for frontline use, supported by ELEVATE-WM data demonstrating a 94% overall response rate, thereby enhancing competitive options for initial therapy selection. Japan’s PMDA approved zanubrutinib in December 2024, with the national insurance framework capping co-pays at JPY 100,000, reducing financial barriers for older patients who are more likely to require treatment. These approvals and label expansions are expected to drive significant near-term volume growth in the Waldenström's Macroglobulinemia market across the United States, the European Union, and Japan.
Ageing Population Enlarging Treatment-Eligible Cohort
By 2030, the global population aged 65 and older is expected to grow from 771 million in 2022 to 994 million, increasing the at-risk population for WM and driving consistent therapy demand throughout the decade.[3]United Nations, “World Population Prospects 2022,” United Nations, un.org In the United States, the number of adults aged 70 and above is projected to reach 43.2 million by 2030, which is likely to boost diagnostic volumes if the age-specific incidence remains stable.[4]U.S. Census Bureau, “Population Projections,” U.S. Census Bureau, census.gov In 2024, individuals aged 65 and older accounted for 21.3% of Europe's population, with some countries exceeding 23%, a trend linked to higher referral volumes to tertiary centers. In Japan, citizens aged 75 and above represented 15.5% of the population in 2024, reflecting a rise in hematologic cancer cases and increased pressure on academic centers.[5]Eurostat, “Population Structure and Ageing,” European Commission, ec.europa.eu Median overall survival for WM has improved from 8.3 years in 2015 to 12.7 years in 2024, extending therapy duration and expanding the treatment base in the Waldenström's Macroglobulinemia market.
Non-Covalent BTK Degraders Addressing Resistance Gaps
Covalent BTK inhibitors primarily target the C481 residue; however, mutations at C481S, observed in approximately 30% of relapsed patients, disrupt binding and drive resistance. This highlights the need for alternative therapies with different binding mechanisms. Non-covalent BTK inhibitors and degraders have demonstrated the ability to retain activity against C481S-mutant clones, effectively restoring disease control in ibrutinib-refractory cases. Nurix’s NX-5948, a cereblon-recruiting BTK degrader, entered Phase 1/2 trials in August 2025. Interim results showed a 67% partial response rate among 12 evaluable WM patients, supporting the continued development of targeted protein degradation for this patient group. BeiGene’s BGB-16673 achieved a 78% overall response rate in a Phase 1 study involving patients who had progressed on ibrutinib, indicating potential sequencing strategies as resistance emerges. In October 2025, the FDA granted Fast Track designation to NX-5948, accelerating review timelines and supporting its expedited development plans.
Restraint Impact Analysis
| RESTRAINT | (~) % IMPACT ON CAGR FORECAST | GEOGRAPHIC RELEVANCE | IMPACT TIMELINE |
|---|---|---|---|
| High annual therapy cost & restricted reimbursement | -0.7% | Global, particularly emerging markets and Europe | Medium term (2-4 years) |
| Limited WM expertise in low-resource regions | -0.5% | Sub-Saharan Africa, South Asia, Latin America | Long term (≥ 4 years) |
| Cardiotoxicity concerns curbing combo-regimen uptake | -0.3% | Global, with heightened scrutiny in North America and Europe | Short term (≤ 2 years) |
| Radio-isotope supply bottlenecks for I-131 therapies | -0.2% | Global, with manufacturing concentrated in North America | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
High Annual Therapy Cost & Restricted Reimbursement
With an annual list price of USD 179,000, zanubrutinib imposes significant pressure on payer budgets in the United States, resulting in utilization management measures that can delay treatment initiation. In November 2024, NICE declined routine NHS funding for zanubrutinib, citing an incremental cost-effectiveness ratio of GBP 87,000 (approximately USD 110,000), which exceeds the UK's GBP 30,000 threshold. Similarly, in March 2024, CADTH recommended against public reimbursement for ibrutinib as a frontline treatment for WM due to uncertainties in the long-term economic model, limiting its adoption in Canada's public programs.
Limited WM Expertise in Low-Resource Regions
In Sub-Saharan Africa, fewer than 120 hematologists hold subspecialty certification in lymphoma. This shortage significantly limits the capacity for timely diagnoses and evidence-based treatments, particularly in remote regions. A comparative analysis highlights that in low-resource settings, diagnosis is delayed by 11 months after symptom onset, compared to 6.8 months in high-income countries, further compounding the disease burden at presentation. In India, a workforce survey from 2024 shows that out of 1,200 medical oncologists, only 340 completed hematologic malignancy fellowships. Additionally, the majority of these specialists are concentrated in metropolitan centers, restricting regional access to WM-specific care. Telemedicine has partially addressed these gaps, with services like eConsult managing 420 WM consultations in 2025. However, reimbursement policies for asynchronous care continue to vary across states in the United States.
Segment Analysis
By Treatment Class: BTK Inhibitors Dominate, Proteasome Inhibitors Gain
In 2025, BTK inhibitors captured a dominant 68.56% share of the treatment-class revenue, reflecting strong prescriber confidence and their position as the preferred option for eligible patients in the Waldenström's Macroglobulinemia market. Data from clinical trials highlighted the advantages of zanubrutinib over ibrutinib, with zanubrutinib demonstrating a longer median progression-free survival of 42.7 months compared to 20.3 months for ibrutinib, emphasizing the durability and continuity of selected BTK regimens. Additionally, zanubrutinib was associated with fewer Grade 3 or higher adverse events than ibrutinib, a critical consideration when cardiac risks influence treatment decisions. These clinical outcomes reinforce the central role of BTK inhibitors in first-line and early-relapse treatments for Waldenström's Macroglobulinemia. At the same time, the expanding pipeline of non-covalent agents and degraders continues to drive attention toward addressing resistance mechanisms. The regulatory progress of acalabrutinib in frontline WM further supports the expectation that multiple BTK options, differentiated by clinical profiles, will coexist, enabling more refined patient-level decisions as clinical guidelines evolve.
Proteasome inhibitors are projected to grow the fastest, with a compound annual growth rate (CAGR) of 5.87% through 2031. This growth is driven by the increasing adoption of bortezomib-based regimens, particularly among patients who develop atrial fibrillation on BTK therapy, a safety concern that significantly impacts treatment sequencing in both community and academic settings.

By Line of Therapy: Second-Line Settings Accelerate
In 2025, first-line therapy accounted for 55.45% of total line-of-therapy revenue, while second-line settings are anticipated to grow at a 6.39% CAGR through 2031. This growth is driven by factors such as resistance biology and safety considerations, which are influencing real-world treatment sequences. China's progress in reimbursing zanubrutinib has enhanced its frontline accessibility. Simultaneously, U.S. regulatory decisions in 2025 have played a pivotal role in shaping its adoption. These developments collectively supported its uptake at the initial stages of treatment. On the other hand, the future of second-line treatments depends on addressing the C481S-mediated resistance. This resistance occurs in approximately 30% of relapsed cases, prompting a shift to either non-covalent agents or BTK degraders as they gain maturity. Nurix's NX-5948 demonstrated a 67% partial response rate in patients with ibrutinib-refractory WM during a 2025 interim analysis. This outcome not only attracted investor interest but also highlighted degradation as a promising strategy in the Waldenström's Macroglobulinemia market. Data from registries indicate that after two prior treatment lines, the median progression-free survival drops to 9.2 months. With a limited number of approved agents, sequencing options become restricted, emphasizing the clinical importance of introducing new mechanisms in later treatment lines.
By Healthcare Setting: Specialty Clinics Rise
In 2025, academic cancer centers accounted for 52.37% of revenue in healthcare settings, reflecting their specialized expertise and leadership in clinical trials, particularly in enrolling patients into WM protocols. A significant portion of WM clinical trials is conducted exclusively at these academic centers, emphasizing their dominance in addressing complex sequencing challenges and providing access to novel mechanisms. Specialty clinics are projected to grow at a CAGR of 6.80% through 2031, driven by advancements such as liquid-biopsy MRD tools and telemedicine, which enable timely care in community settings while reducing referral delays. In 2025, Mayo Clinic's eConsult program managed 420 WM cases, demonstrating reduced travel costs per patient and presenting a practical approach to delivering high-quality care outside major tertiary centers.

Geography Analysis
In 2025, North America, with its established prescriber base and payer structures, is expected to maintain its leadership in accommodating high-cost oncology drugs. Meanwhile, the Asia-Pacific region is projected to experience rapid growth through 2031, driven by expanding reimbursement schemes for BTK inhibitors in the Waldenström's Macroglobulinemia market. Improved market access in China and Japan is already increasing initiation rates, while Australian subsidies have reduced out-of-pocket expenses that previously hindered uptake. Europe, while holding a significant market volume, is advancing at a slower pace. Decisions such as NICE’s 2024 stance on zanubrutinib are shaping treatment choices, resulting in slower alignment with jurisdictions offering early access.
Asia-Pacific’s growth trajectory is further supported by its large and aging population, which is expected to increase the number of eligible patients over time. This demographic trend amplifies the impact of enhanced reimbursements in major countries on the Waldenström's Macroglobulinemia market. The adoption of molecular diagnostics in routine evaluations by clinical teams is narrowing the gap between symptom identification and treatment initiation. This development is increasing the proportion of patients benefiting from targeted therapies earlier in their treatment journey. In Japan and Australia, affordable co-pay structures and national formularies are ensuring the continuation of therapy, which is critical for diseases requiring long-term management. In contrast, the Middle East and Africa, along with South America, continue to face challenges such as limited expert availability and funding, which are keeping their market shares relatively small despite recent policy initiatives.
Competitive Landscape
The Waldenström's Macroglobulinemia market exhibited a moderate concentration, with the top three companies collectively commanding a majority share. Notably, no single entity surpassed the 28% mark, indicating potential opportunities for new entrants to focus on resistance biology or target underserved regions. Competition primarily revolves around label expansions and safety differentiation. For example, BeiGene received FDA acceptance in October 2025 for its frontline application of zanubrutinib. If approved, this would expand the drug's usage to earlier stages of care. Furthermore, combination therapies are gaining traction; the pairing of venetoclax with rituximab achieved an impressive 92% overall response rate in 2024. This not only underscored the efficacy of well-tolerated regimens but also intensified pricing and positioning challenges for single-agent alternatives.
Emerging mechanisms are capturing the attention of both investors and clinicians. Nurix, for instance, advanced its NX-5948 with promising interim data and, in October 2025, secured a notable USD 85 million in funding. This move highlights the growing interest in targeted protein degradation, especially in the context of resistant Waldenström's Macroglobulinemia. Meanwhile, Cellectar's CLR 131, a radiotherapeutic, carved out a unique niche but grappled with radio-isotope supply challenges, hindering its immediate scalability. On the other hand, InnoCare's orelabrutinib, a domestic BTK option in China, showcased significant sales momentum through 2025. This success story underscores the potential of regional champions in solidifying market access within their local landscapes.
Waldenstrom's Macroglobulinemia (WM) Treatment Industry Leaders
TG Therapeutics, Inc.
Curis, Inc.
X4 Pharmaceuticals, Inc.
Nurix Therapeutics, Inc.
BeOne Medicines GmbH
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- January 2026: ImmunityBio, Inc. reported updated efficacy and safety results from its ongoing QUILT-106 clinical study evaluating an off-the-shelf allogeneic CD19 chimeric antigen receptor natural killer cell therapy (CAR-NK). This CD19 t-haNK (CAR-NK) therapy, a high-affinity natural killer (NK) cell treatment, is designed to express a CD19-specific chimeric antigen receptor (CAR). It is used in combination with rituximab (an anti-CD20) for treating patients with Waldenström Non-Hodgkins lymphoma, a rare B-cell malignancy.
- June 2025: Cellectar Biosciences, Inc. announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Therapy Designation to iopofosine I 131. This innovative, first-in-class cancer-targeting agent utilizes a phospholipid ether as a radioconjugate monotherapy for treating relapsed/refractory Waldenstrom macroglobulinemia (r/r WM).
- April 2025: Nurix Therapeutics announced that the FDA granted Orphan Drug Designation to bexobrutideg (NX-5948) for the treatment of Waldenström macroglobulinemia (WM). Bexobrutideg, an orally bioavailable and brain-penetrant Bruton tyrosine kinase (BTK) degrader, is being assessed in a dose-escalation (phase 1a) and cohort-expansion (phase 1b) study for adult patients with relapsed or refractory B-cell malignancies.
Global Waldenstrom's Macroglobulinemia (WM) Treatment Market Report Scope
As per the scope of the report, Waldenström macroglobulinemia, also known as lymphoplasmacytic lymphoma, is an indolent subtype of non-Hodgkin lymphoma that originates in the bone marrow and begins producing abnormal white blood cells that hinder normal body function and cause significant complications. The primary factor responsible for this disorder is the release of interleukin-6 (IL-6) from dendritic cells, which promotes the growth of plasmacytoid lymphocytes.
The Global Waldenstrom's Macroglobulinemia (WM) Treatment Market is segmented by treatment type, line of therapy, healthcare setting, and geography. By treatment type, the market is segmented into BTK inhibitors, proteasome inhibitors, BCL-2 inhibitors, PI3K/mTOR inhibitors, plasmapheresis, and chemotherapy & others. By line of therapy, the market is segmented into first-line, second-line, and third-line & beyond. By healthcare setting, the market is segmented into academic cancer centers, community/regional hospitals, and specialty clinics. By geography, the market is segmented into North America, Europe, Asia-Pacific, the Middle East and Africa, and South America. The market report also covers the estimated market sizes and trends for 17 different countries across major regions globally. The report offers market size and forecasts in value (USD) for the above segments.
| BTK Inhibitors |
| Proteasome Inhibitors |
| BCL-2 Inhibitors |
| PI3K/mTOR Inhibitors |
| Plasmapheresis |
| Chemotherapy & Others |
| First-Line |
| Second-Line |
| Third-Line & Beyond |
| Academic Cancer Centres |
| Community/Regional Hospitals |
| Speciality Clinics |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Treatment Class | BTK Inhibitors | |
| Proteasome Inhibitors | ||
| BCL-2 Inhibitors | ||
| PI3K/mTOR Inhibitors | ||
| Plasmapheresis | ||
| Chemotherapy & Others | ||
| By Line of Therapy | First-Line | |
| Second-Line | ||
| Third-Line & Beyond | ||
| By Healthcare Setting | Academic Cancer Centres | |
| Community/Regional Hospitals | ||
| Speciality Clinics | ||
| By Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the current size and growth outlook for the Waldenström's Macroglobulinemia market?
The Waldenström's Macroglobulinemia market size is USD 188.27 billion in 2026 and is set to grow at a 5.3% CAGR to 2031, reaching USD 256.66 billion.
Which treatments lead adoption in Waldenström's Macroglobulinemia and why?
BTK inhibitors lead with 68.56% treatment-class revenue in 2025, supported by durability and tolerability signals from head-to-head evidence such as the ASPEN trial.
Which regions are expanding fastest for Waldenström's Macroglobulinemia?
Asia-Pacific is the fastest growing region with a projected 6.15% CAGR through 2031, aided by Chinas reimbursement and broader regional access decisions.
What are the key barriers to access in Waldenström's Macroglobulinemia?
High annual therapy costs, reimbursement denials or restrictions, limited specialist availability in low-resource regions, and safety monitoring demands for combination regimens remain the main hurdles.
How is resistance to BTK inhibitors being addressed in Waldenström's Macroglobulinemia?
Non-covalent BTK inhibitors and BTK degraders like NX-5948 retain activity against C481S-mutant disease and are advancing with regulatory support, including FDA Fast Track designation.
Which care settings are shaping patient management in Waldenström's Macroglobulinemia?
Academic centers retain a leading revenue role, while specialty clinics are growing faster due to MRD tools and telemedicine that enable more care to stay local.
Page last updated on:









