Spain In-vitro Diagnostics Market Analysis by Mordor Intelligence
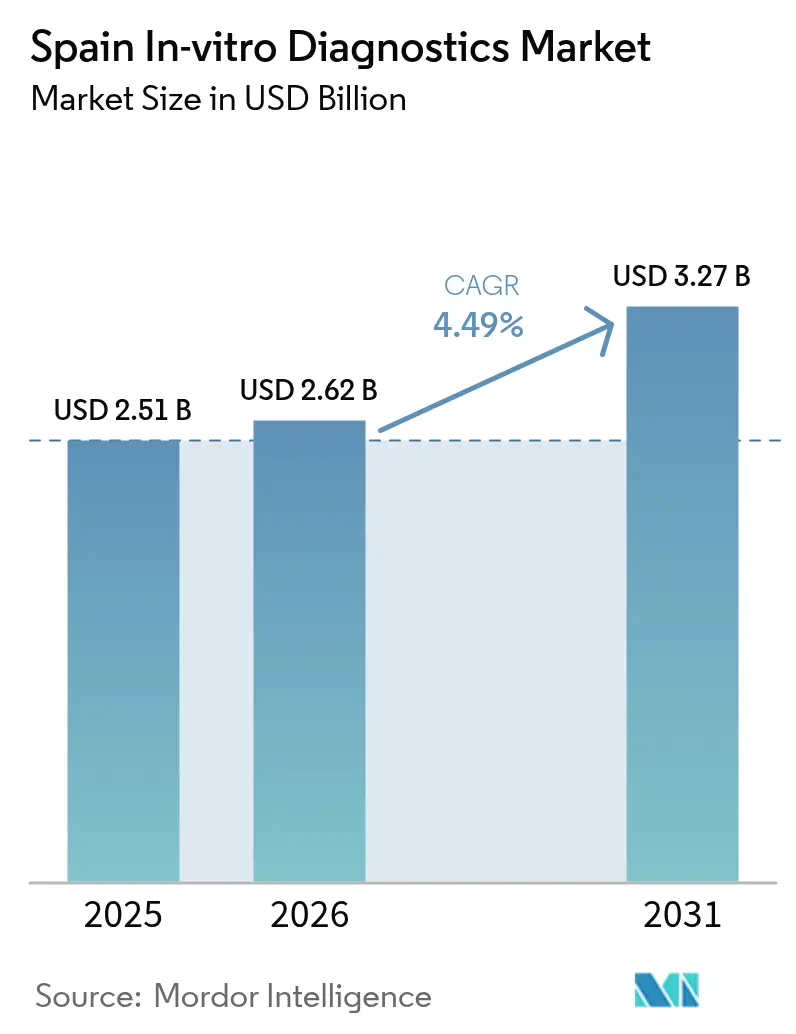
Spain IVD market size in 2026 is estimated at USD 2.62 billion, growing from 2025 value of USD 2.51 billion with 2031 projections showing USD 3.27 billion, growing at 4.49% CAGR over 2026-2031. Continued momentum is rooted in an aging population, higher chronic-disease incidence, and the country’s full transition to the EU In Vitro Diagnostic Regulation (IVDR). Strong demand for frequent renal, diabetes, and hypertension screening is lifting test volumes, while hospital groups in Madrid and Catalonia are modernizing laboratories with AI-enabled analyzers that shorten turnaround times. Molecular assay innovators are benefiting from European Investment Bank financing, and Spain’s Digital Health Strategy is steering budgets toward connected instruments that meet IVDR traceability rules. At the same time, decentralized procurement across 17 autonomous communities is nudging suppliers toward value-based contracts that link reagent spending to clinical outcomes.
Key Report Takeaways
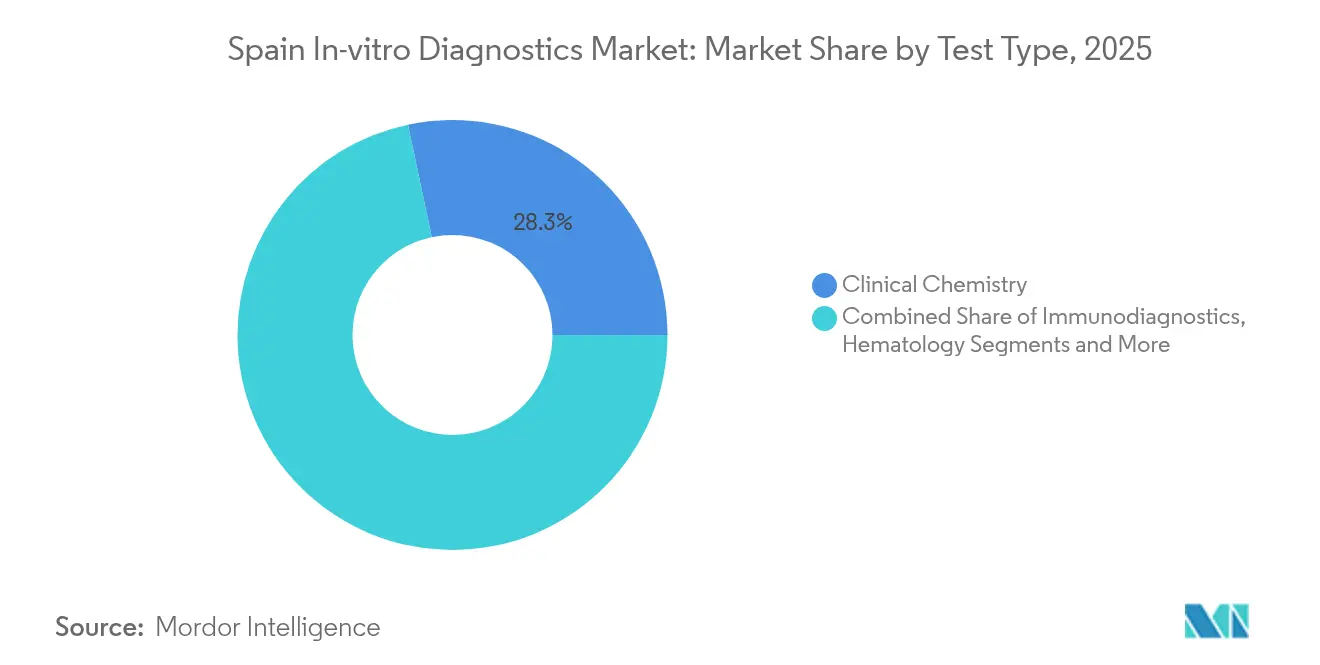
- By test type, clinical chemistry led with 28.32% revenue share in 2025, while molecular diagnostics is projected to advance at a 9.38% CAGR to 2031.
- By product, reagents & consumables captured 70.35% of the Spain IVD market share in 2025, and software & services is forecast to grow at an 8.01% CAGR through 2031.
- By usability, disposable devices commanded 82.75% of the Spain IVD market size in 2025, whereas reusable devices are expected to expand at an 8.12% CAGR to 2031.
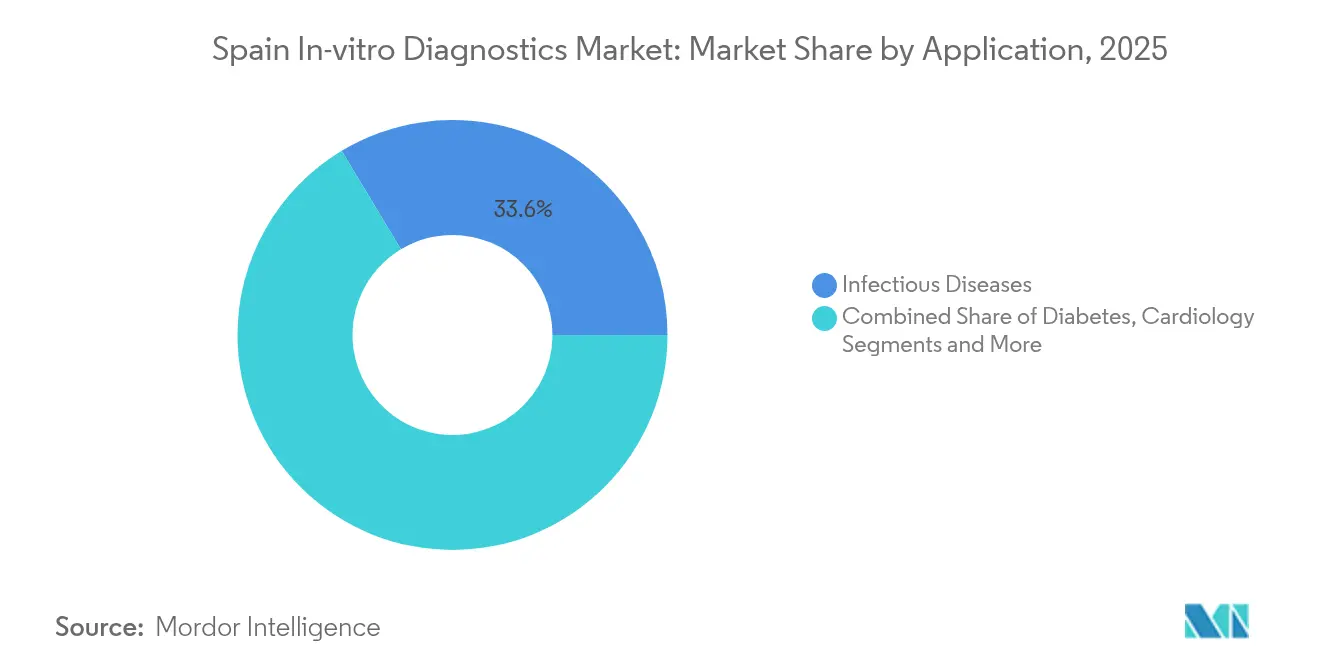
- By application, infectious-disease testing accounted for a 33.62% share in 2025, and oncology diagnostics is on track for a 9.42% CAGR between 2026 and 2031.
- By end user, hospital & reference laboratories held 60.92% revenue share in 2025, while home-care and ambulatory point-of-care settings are growing the fastest at a 10.18% CAGR.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Spain In-vitro Diagnostics Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising burden of chronic and lifestyle diseases | +1.8% | Asturias, Castile and León, Galicia | Long term (≥ 4 years) |
| Rapid technological innovation in molecular and immunodiagnostics | +1.2% | Madrid, Barcelona, Valencia | Medium term (2-4 years) |
| Expansion of point-of-care testing across primary and home settings | +0.9% | Catalonia, Madrid | Medium term (2-4 years) |
| Government and EU investment programs for digital lab modernization | +0.7% | National | Short term (≤ 2 years) |
| Growth of precision medicine and companion-diagnostics adoption | +0.5% | Madrid, Barcelona, Valencia, Seville | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Rising Burden of Chronic & Lifestyle Diseases
About 15.1% of Spanish adults live with chronic kidney disease, a figure that continues to climb with population aging. Higher prevalence of diabetes, cancer, and cardiovascular disorders is prompting earlier, more frequent testing in primary care clinics. The CARABELA-CKD program standardizes nephrology pathways and lifts demand for creatinine, eGFR, and micro-albumin tests across the Spain IVD market. Laboratories in Galicia and Asturias report double-digit yearly increases in renal panels, pushing reagent distributors to boost stock levels. Regional health authorities now bundle screening targets into hospital financing agreements, an approach that ties purchasing volumes to chronic-care metrics. Collectively, these factors underpin consistent baseline growth for routine and specialty assays.
Rapid Technological Innovation in Molecular & Immunodiagnostics
The European Investment Bank’s EUR 20 million loan to Universal DX accelerates liquid-biopsy development for early colorectal-cancer detection[1]European Investment Bank, “Universal DX Liquid-Biopsy Financing,” eib.org. Spanish startups mix next-generation sequencing with machine-learning algorithms to profile multiple biomarkers in one run, lowering per-test costs. University hospitals in Valencia validate multiplex respiratory panels that deliver 90-minute results and reduce inpatient isolation days. Immunodiagnostic platforms now integrate chemiluminescent detection with automated calibration, raising sensitivity for thyroid and cardiac markers. As IVDR pushes traceability and performance benchmarking, local firms adopt cloud-based quality-control dashboards that feed directly into notified-body audits, strengthening compliance while sharpening competitive differentiation.
Expansion of Point-of-Care Testing Across Primary & Home Settings
Catalonia’s EUR 580 million primary-care improvement plan channels fresh funds into rapid analyzers for glucose, CRP, and coagulation monitoring. General practitioners deploy handheld readers linked to electronic health records, allowing same-visit treatment adjustments for chronic patients. In Madrid, pharmacies pilot antigen-test kiosks that transmit anonymized data to regional surveillance databases within minutes, supporting real-time outbreak tracking. Supply-chain managers report that barcode-enabled cartridge systems cut wastage by 12% compared with manual strips, making point-of-care economics more favorable. Home-based INR monitoring also gains traction as insurers reimburse remote data uploads, easing pressure on hospital anticoagulation clinics.
Government & EU Investment Programs for Digital Lab Modernization
Spain’s Recovery & Resilience Plan earmarks EUR 800 million for high-tech cancer-diagnostic devices, driving replacement cycles for outdated analyzers[2]OECD/European Observatory, “Recovery and Resilience Plan,” oecd.org. The PERTE for Cutting-edge Health adds EUR 2.36 billion for AI integration and a national health data lake. University Hospital La Paz in Madrid installs robotic sample-track lines that lift hourly throughput by 40% while improving specimen traceability. Regional tenders now require middleware interoperability, prompting mid-tier suppliers to partner with software vendors for IVDR-ready connectivity. Collectively, these capital injections speed lab automation and promote standards-based data exchange across the Spain IVD market.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Stringent and evolving EU IVDR regulatory landscape | -0.9% | National | Short term (≤ 2 years) |
| Regional reimbursement delays and budget constraints | -0.7% | Andalusia, Extremadura, Murcia | Medium term (2-4 years) |
| Shortage of skilled laboratory personnel and training gaps | -0.5% | Rural provinces | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Stringent & Evolving EU IVDR Regulatory Landscape
Seventy-three percent of Spanish laboratories say they lack adequate guidance to complete IVDR conformity files[3]Association for Molecular Pathology, “EU IVDR Survey,” amp.org. High-risk assays must meet new performance-study and post-market-surveillance rules by May 2025, yet notified-body capacity remains tight. The 2024/1860 amendment extends certain timelines but also adds supply-chain traceability duties, raising administrative workloads. Smaller reagent makers divert R&D budgets toward documentation, slowing product-pipeline turnover. Because each autonomous community interprets EU law through its own procurement filters, suppliers face variable local checklists that complicate national launches.
Regional Reimbursement Delays & Budget Constraints
Only 9.49% of reimbursement decisions reference formal HTA conclusions, exposing gaps in Spain’s evidence-based funding process. Andalusia and Extremadura apply tight annual spending caps that postpone adoption of new oncology panels, whereas Madrid greenlights them within six months. Draft royal-decree proposals aim to create a single national HTA framework, but interim uncertainty restrains hospital buyers. Point-of-care devices struggle most: without a uniform tariff code, clinics finance them from discretionary budgets, slowing roll-out despite proven efficiency gains.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Test Type: Molecular Diagnostics Reshaping Diagnostic Paradigms
Clinical chemistry generated 28.32% of the Spain IVD market size in 2025, supported by its central role in metabolic, renal, and hepatic panels. Routine analyzers achieve high throughput and cost efficiency, which stabilizes reagent demand. However, molecular diagnostics is posting a 9.38% CAGR as hospitals expand oncology and infectious-disease gene testing. The Spain IVD market benefits from Seegene and Werfen’s joint venture, which promises syndromic PCR menus tailored to local antimicrobial-resistance profiles. As next-generation sequencing costs fall, regional centers pilot cancer-risk screening programs financed through EU innovation funds, cementing growth.
A shift toward hybrid panels that combine immunoassay and PCR markers blurs traditional segment boundaries. Laboratories deploy reflex-testing protocols that trigger confirmatory molecular runs after abnormal chemistry results, raising cross-segment reagent consumption. IVDR mandates traceable lot-release data, encouraging integration of chemistry and molecular middleware. These developments reinforce molecular diagnostics’ strategic importance while ensuring clinical chemistry retains volume leadership in the Spain IVD market.

By Product: Software Integration Driving Diagnostic Intelligence
Reagents & consumables represented 70.35% of the Spain IVD market share in 2025, reflecting high test frequency and replenishment cycles. Automated inventory modules tied to laboratory-information systems trim expiry-related wastage, yet tight tender pricing keeps margins thin. Software & services—currently under 10% of revenues—will outpace other categories at an 8.01% CAGR as digital pathology, cloud middleware, and AI analytics become procurement priorities. Hospitals adopt subscription models that bundle instrument leasing with predictive-maintenance modules, converting capital outlays into operating expenses.
Instrument vendors now embed open APIs so third-party algorithms can interrogate analyzer data, turning platforms into clinical-decision hubs. Spain’s Digital Health Strategy sets interoperability standards that favor HL7-FHIR messaging, nudging suppliers to certify integrations. In turn, reagent makers collaborate with software firms to package quality-control dashboards, tying consumable sales to analytical-performance guarantees. This convergence between hardware, reagents, and analytics drives holistic purchasing decisions across the Spain IVD market.
By Usability: Disposable Dominance Reflects Infection-Control Priorities
Disposable IVD devices accounted for 82.75% of 2025 revenues, a figure cemented by post-pandemic infection-control protocols. Single-use cassettes simplify staff workflows and align with IVDR sample-traceability rules, outweighing per-test cost premiums. Environmental policymakers, however, press laboratories to adopt circular-economy practices, spurring interest in reusable plastics and validated reprocessing cycles. New polymer blends withstand autoclave temperatures without leaching, supporting an 8.12% CAGR for reusable formats through 2031.
Manufacturers position hybrid solutions—disposable microfluidic chips housed in reusable readers—to balance sustainability and biosafety. Procurement teams include carbon-footprint criteria in tenders, rewarding suppliers that offer recycling schemes. As a result, the Spain IVD market observes gradual convergence between disposability and reusability, with device design increasingly optimized for both infection control and ecological stewardship.
By Application: Oncology Diagnostics Driving Precision-Medicine Adoption
Infectious-disease assays delivered 33.62% of Spain IVD market revenues in 2025, buoyed by persistent respiratory-virus surveillance and antimicrobial-resistance screening. Yet oncology diagnostics will expand at a 9.42% CAGR, propelled by non-invasive liquid-biopsy initiatives funded by EU and national grants. Early-detection programs in Barcelona now include annual circulating-tumor-DNA tests for high-risk populations, elevating demand for ultra-sensitive PCR reagents.
Diabetes monitoring remains a sizeable niche due to sustained prevalence and government emphasis on chronic-disease management. Cardiology assays increasingly pair high-sensitivity troponin with digital ECG analytics, providing faster rule-out of acute coronary syndromes in emergency departments. Advanced multiplex platforms allow autoimmune-panel consolidation, shortening diagnostic journeys for systemic lupus and rheumatoid arthritis patients. These dynamics keep application portfolios diverse while reinforcing oncology’s role as a future growth anchor for the Spain IVD market.

By End User: Home-Care Settings Disrupting Traditional Testing Paradigms
Hospital and reference laboratories captured 60.92% of Spain IVD market revenues in 2025, underpinned by integrated networks that serve acute-care and specialist clinics. Automation upgrades yield high productivity, yet capacity constraints persist amid rising test complexity. Home-care and ambulatory point-of-care channels will rise at a 10.18% CAGR as telehealth reimbursement widens. Pharmacies in Catalonia pilot HbA1c testing services linked to virtual endocrinology consults, cutting referral times by two weeks.
Diagnostic laboratories negotiate reagent-rental contracts that swap higher menu breadth for lower upfront instrument fees, aligning with variable outpatient demand. Academic centers leverage sequencing cores for translational-research contracts, blurring lines between patient testing and research. Community health centers expand basic panels such as lipid and thyroid profiles using portable analyzers, enhancing access in underserved areas. Collectively these shifts point to a distributed yet interconnected ecosystem that keeps the Spain IVD market resilient and patient-centric.
Geography Analysis
Madrid and Catalonia together account for over 35.24% of the Spain IVD market size, supported by dense hospital networks and concentrated R&D funding. Regional health ministries funnel EU cohesion funds into molecular-oncology hubs that attract multinational clinical-trial activity. Andalusia, although budget-constrained, shows steady uptake of high-throughput chemistry analyzers driven by chronic-care programs, while Galicia and Asturias focus on renal and cardiac panels aligned with their aging demographics. In central Castilla-La Mancha, procurement pools negotiate multi-year reagent contracts that stabilize pricing yet slow product refresh cycles. Valencia integrates AI-driven triage tools within public hospitals, accelerating imaging-to-lab data handoffs. Basque Country’s technology park hosts IVD startups that export middleware solutions, illustrating regional specialization within the Spain IVD industry. Across the Canary and Balearic Islands, maritime logistics shape inventory practices, favoring compact instruments and extended-shelf-life reagents. These geographic nuances require suppliers to tailor channel strategies, bolstering the Spain IVD market’s heterogeneity while broadening its growth base.
Competitive Landscape
The top vendors—Roche, bioMeriux, Bio-Rad Laboratories, and Danaher Corp—collectively hold significant revenue, indicating moderate concentration. Global firms leverage scale in reagents and automation, whereas Spanish specialists compete in niche molecular panels, software, and instrument service contracts. Werfen’s partnership with Seegene brings syndromic PCR portfolios under a local manufacturing umbrella, tightening lead times and easing IVDR conformity. Roche pilots bundle contracts that guarantee uptime for digital immunoassay lines, shifting risk to the supplier side.
Small- to mid-size startups focus on AI-assisted pathology and point-of-care connectivity, often collaborating with academic incubators. PERTE grants accelerate prototype validation, although many newcomers still outsource final assembly to larger contract manufacturers. Procurement decentralization leads established players to maintain region-specific sales teams, while distributors bundle multi-brand reagents to win provincial hospital bids. Technology, regulatory agility, and service quality thus become pivotal differentiators in the Spain IVD market.
Spain In-vitro Diagnostics Industry Leaders
Becton, Dickinson and Company
Bio-Rad Laboratories Inc.
F. Hoffmann-La Roche AG
Danaher Corporation (Beckman Coulter, Cepheid)
bioMerieux SA
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- May 2025: Miura Partners invested new capital in Saesco to scale local manufacturing of lab consumables and strengthen domestic supply resilience.
- October 2024: Seegene and Werfen created a Spanish NewCo to co-develop syndromic real-time PCR panels for respiratory and gastrointestinal pathogens.
Spain In-vitro Diagnostics Market Report Scope
As per the scope of this report, in vitro diagnostics involves medical devices and consumables that are utilized to perform in vitro tests on various biological samples. They are used for the diagnosis of various medical conditions, such as infectious diseases, cardiovascular diseases, diabetes, and cancer, among other diseases. The Spain In-Vitro Diagnostics Market is segmented by Test Type (Clinical Chemistry, Molecular Diagnostics, and Other Techniques), Product (Instrument, Reagent, and Other Products), Usability (Disposable IVD Devices and Reusable IVD Devices), Application (Infectious Disease, Diabetes, Cancer, Cardiology, Autoimmune Diseases and Other Applications), and End-users (Diagnostic Laboratories, Hospitals and Clinics and Other End-users). The report offers the value (in USD million) for the above segments.
| Clinical Chemistry |
| Immunodiagnostics |
| Molecular Diagnostics |
| Hematology |
| Microbiology |
| Coagulation |
| Point-of-Care (POC) Tests |
| Instruments & Analyzers |
| Reagents & Consumables |
| Software & Services |
| Disposable IVD Devices |
| Reusable IVD Devices |
| Infectious Disease |
| Diabetes |
| Oncology (Cancer) |
| Cardiology |
| Autoimmune Disorders |
| Other Applications |
| Hospitals & Clinics |
| Diagnostic Laboratories |
| Academic & Research Institutes |
| Home-Care / Ambulatory POC Settings |
| Other End-Users |
| By Test Type | Clinical Chemistry |
| Immunodiagnostics | |
| Molecular Diagnostics | |
| Hematology | |
| Microbiology | |
| Coagulation | |
| Point-of-Care (POC) Tests | |
| By Product | Instruments & Analyzers |
| Reagents & Consumables | |
| Software & Services | |
| By Usability | Disposable IVD Devices |
| Reusable IVD Devices | |
| By Application | Infectious Disease |
| Diabetes | |
| Oncology (Cancer) | |
| Cardiology | |
| Autoimmune Disorders | |
| Other Applications | |
| By End-User | Hospitals & Clinics |
| Diagnostic Laboratories | |
| Academic & Research Institutes | |
| Home-Care / Ambulatory POC Settings | |
| Other End-Users |


Key Questions Answered in the Report
How large is the Spain IVD market in 2026?
The Spain IVD market size is valued at USD 2.62 billion in 2026.
What is the expected growth rate for Spain's in-vitro diagnostics through 2031?
Revenue is projected to rise at a 4.49% CAGR, reaching USD 3.27 billion by 2031.
Which test segment is expanding the fastest?
Molecular diagnostics leads with a forecast 9.38% CAGR, driven by oncology and infectious-disease panels.
Why are disposable IVD devices prevalent in Spain?
Infection-control protocols and streamlined workflows push disposable devices to an 82.75% share of total sales.
How is IVDR affecting market entry?
New conformity requirements raise documentation costs and lengthen approval timelines, especially for smaller manufacturers.
What region shows the highest adoption of digital lab technology?
Madrid and Catalonia invest most heavily in AI-enabled analyzers and data-sharing platforms under EU modernization schemes.
Page last updated on: