South Africa Neurology Devices Market Analysis by Mordor Intelligence
The South Africa Neurology Devices Market is expected to register a CAGR of 4.5% during the forecast period.
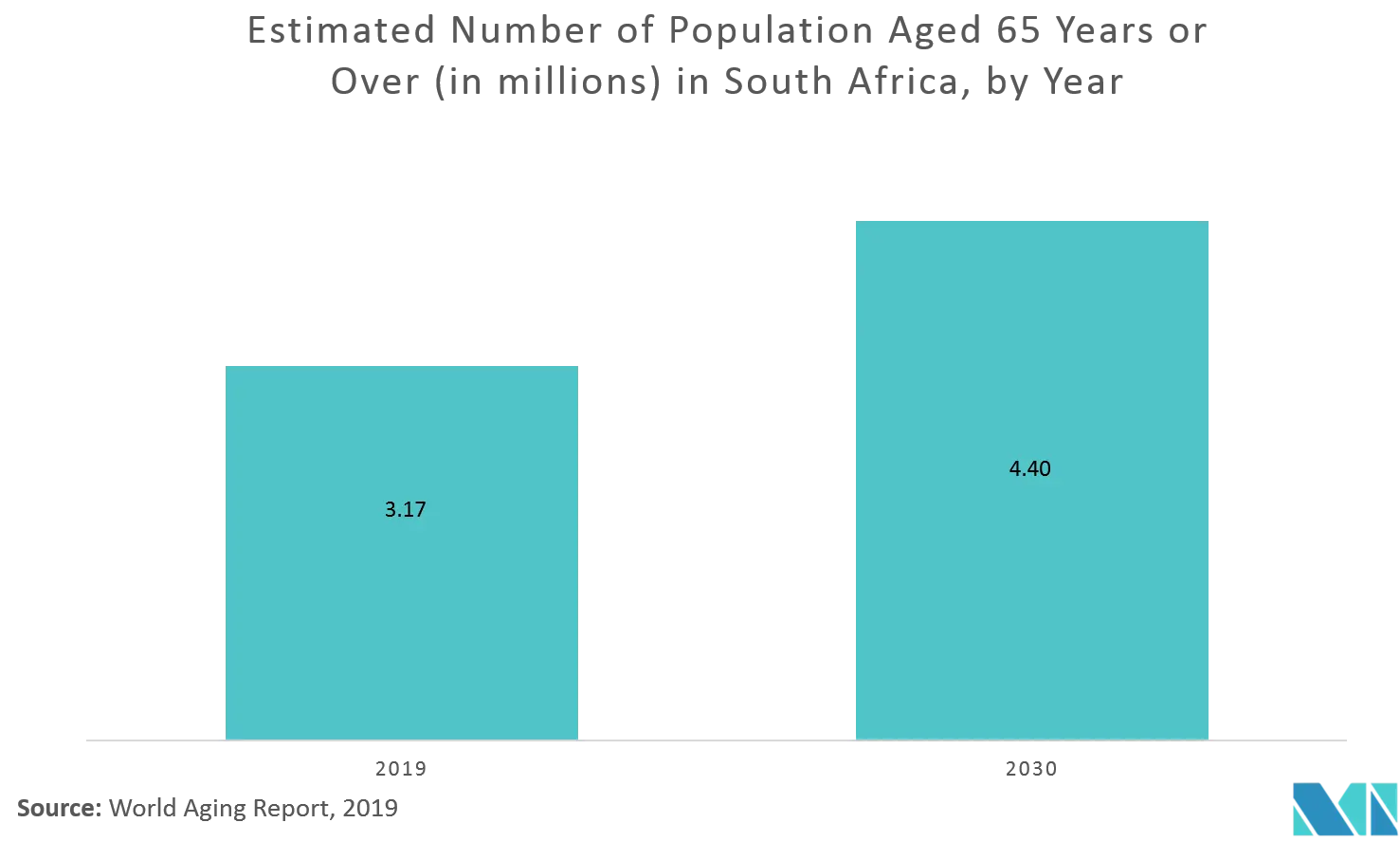
According to the World Population Ageing 2019 report, in South Africa population aged 65 years or over in 2019 was 3.171 million (5.4%) and this number is projected to reach to 4.404 million (6.7%) in 2030. This is expected to consistently increase the dementia patient population which boost the interest of existing players to invest more in this country.
Thus, owing to the above factors the market is expected to show significant growth over the forecast period.
South Africa Neurology Devices Market Trends and Insights
Sacral Nerve Stimulation Devices is the Segment under Neurostimulation Devices that is Expected to Grow Fastest during the Forecast Period
The sacral nerve stimulation devices are mostly used for patients for whom drug therapy or other types of medications are not successful. It is performed with the help of a small device, which is capable of sending electrical impulses to the targeted sacral nerves located in the lower back region. These devices are found to be successful in treating bladder problems. Hence, with increasing incidences of overactive bladder, the sacral nerve stimulation devices segment is expected to grow during the forecast period.
Sacral nerve stimulation devices are an effective and safe treatment for patients suffering from fecal and urinary dysfunction. Increasing demand for minimally invasive therapy, rising prevalence of urinary bladder diseases, and technological advancements such as the introduction of novel coatings and engineering strategies are the main factors driving the growth of the market.
In South Africa, Medtronic Plc offers "The InterStim II System" the neurostimulator for bladder and bowel control which delivers sustained improvements in quality of life for people with overactive bladder (OAB), non-obstructive urinary retention, and chronic fecal incontinence (FI).


Competitive Landscape
The South Africa Neurology Devices Market is fragmented competitive. There are market participants involved in R&D for the development of advanced stimulation and neuromodulation techniques with minimally invasive procedures that are more preferable to the patients. Furthermore, many private companies are showing great interest in developing novel neurological devices to earn huge profits. In the coming years few other players are expected to enter into the market.
South Africa Neurology Devices Industry Leaders
B. Braun Melsungen AG
Stryker Corporation
Medtronic PLC
Abbott Laboratories
Johnson and Johnson
- *Disclaimer: Major Players sorted in no particular order




South Africa Neurology Devices Market Report Scope
As per the scope of the report, neurological devices are medical devices that help to diagnose, prevent, and treat a variety of neurological disorders and conditions, such as Alzheimer's disease, Parkinson's disease, major depression, and traumatic brain injury. The growth of neurological devices is being driven through an emphasis on developing superior neuromodulation and neurostimulation techniques, leveraging minimally invasive procedures that benefit patients. The South Africa Neurology Devices Market is segmented by type of device (Cerebrospinal Fluid Management Device, Interventional Neurology Device, Neurosurgery Device, Neurostimulation Device, and Other Type of Devices).
| Cerebrospinal Fluid Management Devices | |
| Interventional Neurology Devices | Interventional/Surgical Simulators |
| Neurothrombectomy Devices | |
| Carotid Artery Stents | |
| Others | |
| Neurosurgery Devices | Neuroendoscopes |
| Stereotactic Systems | |
| Other Neurosurgery Devices | |
| Neurostimulation Devices | Spinal Cord Stimulation Devices |
| Deep Brain Stimulation Devices | |
| Sacral Nerve Stimulation Devices | |
| Other Neurostimulation Devices | |
| Other Types of Devices |
| By Type of Device | Cerebrospinal Fluid Management Devices | |
| Interventional Neurology Devices | Interventional/Surgical Simulators | |
| Neurothrombectomy Devices | ||
| Carotid Artery Stents | ||
| Others | ||
| Neurosurgery Devices | Neuroendoscopes | |
| Stereotactic Systems | ||
| Other Neurosurgery Devices | ||
| Neurostimulation Devices | Spinal Cord Stimulation Devices | |
| Deep Brain Stimulation Devices | ||
| Sacral Nerve Stimulation Devices | ||
| Other Neurostimulation Devices | ||
| Other Types of Devices | ||


Key Questions Answered in the Report
What is the current South Africa Neurology Devices Market size?
The South Africa Neurology Devices Market is projected to register a CAGR of 4.5% during the forecast period (2025-2030)
Who are the key players in South Africa Neurology Devices Market?
B. Braun Melsungen AG, Stryker Corporation, Medtronic PLC, Abbott Laboratories and Johnson and Johnson are the major companies operating in the South Africa Neurology Devices Market.
What years does this South Africa Neurology Devices Market cover?
The report covers the South Africa Neurology Devices Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the South Africa Neurology Devices Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
South Africa Neurology Devices Market Report
Statistics for the 2025 South Africa Neurology Devices market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. South Africa Neurology Devices analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.





