Global Rare Hemophilia Factors Market Size and Share
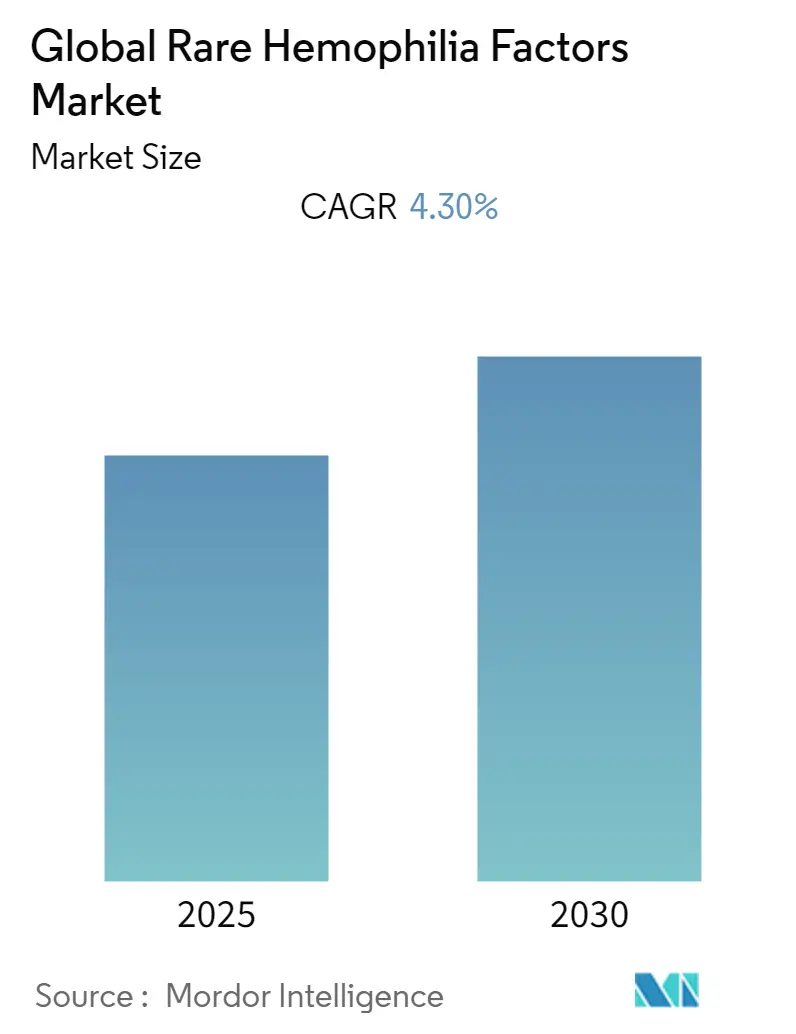
Global Rare Hemophilia Factors Market Analysis by Mordor Intelligence
The Global Rare Hemophilia Factors Market is expected to register a CAGR of 4.3% during the forecast period.
The COVID-19 outbreak notably impacted the healthcare sector. As per the article published in 2022 under the title 'Acquired Hemophilia A Developed Post COVID-19 Vaccine: An Extremely Rare Complication', eight days after receiving the first dose of the COVID-19 vaccine, an elderly man with a history of autoimmune illnesses (polymyalgia rheumatica) and hepatitis C virus experienced the development of Acquired Hemophilia A (AHA). Moreover, as per the article published in 2022 under the title 'The Mixed Effects of the First COVID-19 Lockdown on Patients With Hemophilia', the study results showed that the frequency of bleeding episodes remained unchanged or decreased in 81% of children and 78% of adults with hemophilia during the lockdown. Hence as per the factors mentioned above, COVID-19 had a significant impact on the rare hemophilia factors market.
Certain factors are driving the market growth including rising adoption and approval of new treatment techniques, a growing number of government initiatives, and funding. For instance as per the article published in 2021 under the title 'Recent Advances in the Treatment of Hemophilia: A Review', a new era of prevention and treatment of bleeding started with the introduction of a bi-specific monoclonal antibody that clones the effects of FVIII in the tenase complex, moreover other non-replacement therapies are also in development.
Similarly, according to the World Hemophilia Federation's annual global survey conducted in a period of 1999 to 2020, the number of people affected with hemophilia was 347,026 in 2020. Scientists have reported that hemophilia is three times more prevalent in men worldwide than previously thought and is still associated with a poorer life expectancy, especially in low-income countries. Therefore, the increase in hemophilia is associated with the increasing demand for the Rare Hemophilia Factors Market.
As per the aforementioned factors, the rare hemophilia factors market is likely to grow over the forecast period. However, the high cost associated with hemophilia treatment restrains the market growth.
Global Rare Hemophilia Factors Market Trends and Insights
Fresh Frozen Plasma (FFP) is Expected to Hold Significant Market Share in the Treatment Segment
Plasma transfusion is indicated for the management of bleeding or to prevent bleeding before an urgent invasive procedure in patients requiring the replacement of multiple coagulation factors. Fresh frozen plasma is the fluid portion of a unit of whole blood that is frozen in a designated time frame, usually within 8 hours. FFP contains all coagulation factors except platelets. FFP contains fibrinogen (400 to 900 mg/unit), albumin, protein C, protein S, antithrombin, and tissue factor pathway inhibitor. FFP is administered intravenously. FFP is to be ABO compatible with the recipient's red cells.
Research and development in the field of fresh frozen plasma fuels market growth. For instance, as per the article published in 2020 under the title 'Recent advances in the use of fresh frozen plasma, cryoprecipitate, immunoglobulins, and clotting factors for transfusion support in patients with hematologic disease', the use of plasma and cryoprecipitates is increasing in the treatment of coagulopathies like disseminated intravascular coagulation (DIC), which affect many plasma proteins, as well as liver disease, which affects fewer plasma proteins. These conditions are becoming more common because there are no effective treatments for any of these conditions. Furthermore, as per the article published in 2021 under the title 'Outcomes of Patients Who Undergo Transfusion of Fresh Frozen Plasma: A Prospective, Observational, Multicentre Cohort Study in Hiroshima, Japan', FFP has been transfused appropriately in at least 63.8% of cases and it is important to promote appropriate use of FFP so as not to waste blood resources.
Because of the aforementioned factors, fresh frozen plasma (FFP) is anticipated to face growth in the rare hemophilia factors market over the forecast period.
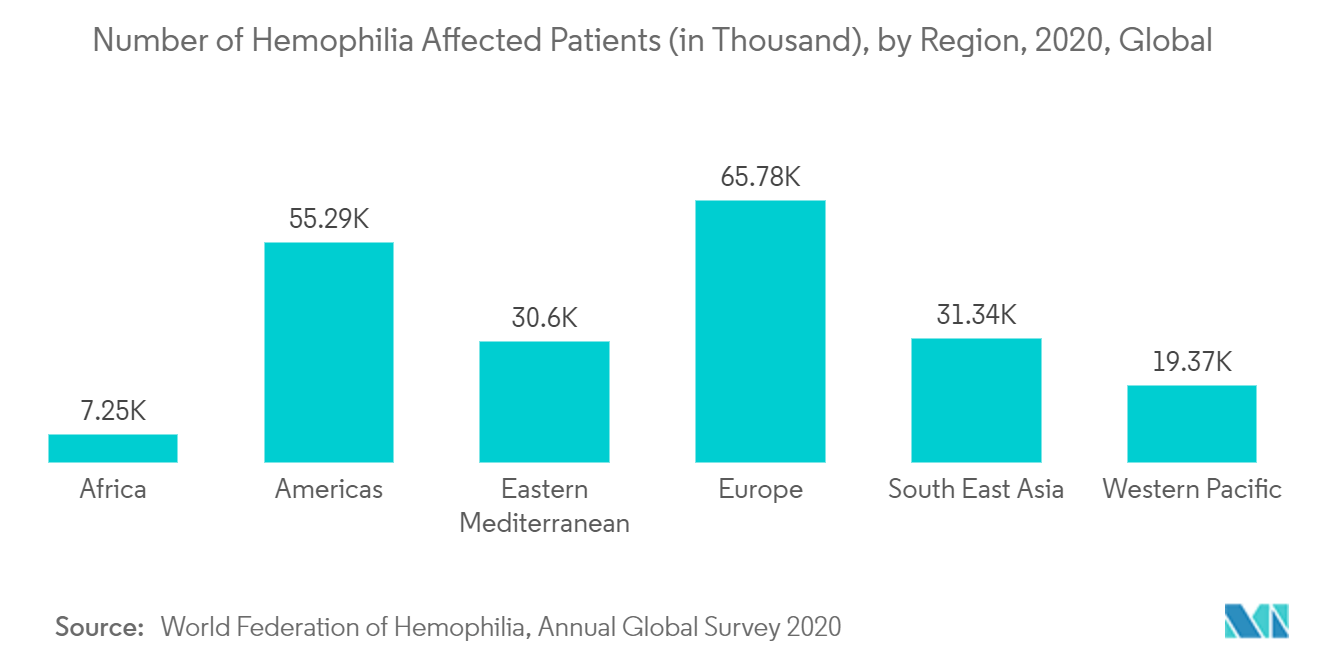


North America Dominates the Market and Expected to do Same in the Forecast Period
The major factors driving the growth of the rare hemophilia factors market in the region include the therapeutic advances in hemophilia, which led to a wave of new Food and Drug Administration-approved agents and increasing awareness about technologically advanced products, increasing prevalence of hemophilia, and the presence of a huge patient pool.
According to the World Federation of Hemophilia 2020 report, there were about 14,816 patients with hemophilia (Hemophilia A affected 11,790 people and Hemophilia B affected with 3,026 people) in the United States in 2020. In Canada a total of 4,709 people were affected with hemophilia, 'Hemophilia A' affected 3,223 people and 'Hemophilia B' affected 701 people. The high prevalence of hemophilia cases in these regions is expected to drive the growth of the market in this region due to the higher adoption of therapies for hemophilia.
Increased awareness regarding the importance of bleeding disorders fuels market growth. For instance, each March, National Hemophilia Foundation provides awareness of inheritable blood and bleeding disorders during Bleeding Disorders Awareness Month. This month patients and families with hemophilia, von Willebrand disease, rare factor disorders, and more, share their lives, stories, struggles, and successes.
Hence as per the factors mentioned above, North America is expected to witness growth in the rare hemophilia factors market over the forecast period.


Competitive Landscape
The global Rare Hemophilia Factors market is moderately competitive and consists of a number of major players. The companies like Baxter Healthcare, Bayer Healthcare, Bio Products Laboratory Ltd., Biogen, CSL Behring, Novo Nordisk, Pfizer, Inc., Takeda Pharmaceutical Co. Ltd., among others, hold the substantial market share in the Rare Hemophilia Factors market.
Global Rare Hemophilia Factors Industry Leaders
Bayer AG
Pfizer, Inc.
Takeda Pharmaceutical Co. Ltd.
Sanofi S.A
Novo Nordisk
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- In June 2022, the United States Food and Drug Administration granted breakthrough therapy designation to 'efanesoctocog alfa' for hemophilia A.Efanesoctocog alfa is the first factor VIII therapy to be awarded Breakthrough Therapy designation by the FDA. This designation is based on XTEND-1 Phase 3 study data demonstrating clinically meaningful prevention of bleeds and superiority in the prevention of bleeding episodes compared to prior prophylaxis factor treatment.
- In May 2022, Takeda Pharmaceutical Co Ltd announced the expansion of its rare diseases portfolio in India with the launch of Adynovate. Adynovate is an innovative extended half-life recombinant Factor VIII (rFVIII) treatment, using established technology for hemophilia A patients.


Global Rare Hemophilia Factors Market Report Scope
As per the scope of the report, hemophilia is a genetic bleeding disorder that prevents the blood from clotting normally. The primary symptom is uncontrolled, often spontaneous bleeding in different areas of the body. The deficiency of fibrinogen, prothrombin, factor V (FV), FVII, FVIII, FIX, FX, FXI, and FXIII, called rare coagulation disorders (RCDs), may result in coagulopathies leading to spontaneous or post trauma and post surgery hemorrhages. The Rare Hemophilia Factors Market is segmented by Treatment (Factor Concentrate, Fresh Frozen Plasma, Cryoprecipitate, Others) and Geography (North America, Europe, Asia-Pacific, Middle-East and Africa, and South America). The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD million) for the above segments.
| Factor Concentrate |
| Fresh Frozen Plasma (FFP) |
| Cryoprecipitate |
| Others |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Treatment | Factor Concentrate | |
| Fresh Frozen Plasma (FFP) | ||
| Cryoprecipitate | ||
| Others | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the current Global Rare Hemophilia Factors Market size?
The Global Rare Hemophilia Factors Market is projected to register a CAGR of 4.3% during the forecast period (2025-2030)
Who are the key players in Global Rare Hemophilia Factors Market?
Bayer AG, Pfizer, Inc., Takeda Pharmaceutical Co. Ltd., Sanofi S.A and Novo Nordisk are the major companies operating in the Global Rare Hemophilia Factors Market.
Which is the fastest growing region in Global Rare Hemophilia Factors Market?
Asia-Pacific is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Global Rare Hemophilia Factors Market?
In 2025, the North America accounts for the largest market share in Global Rare Hemophilia Factors Market.
What years does this Global Rare Hemophilia Factors Market cover?
The report covers the Global Rare Hemophilia Factors Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Global Rare Hemophilia Factors Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
Global Rare Hemophilia Factors Market Report
Statistics for the 2025 Global Rare Hemophilia Factors market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Global Rare Hemophilia Factors analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.





