Global Prostate Specific Antigen (PSA) Test Market Size and Share
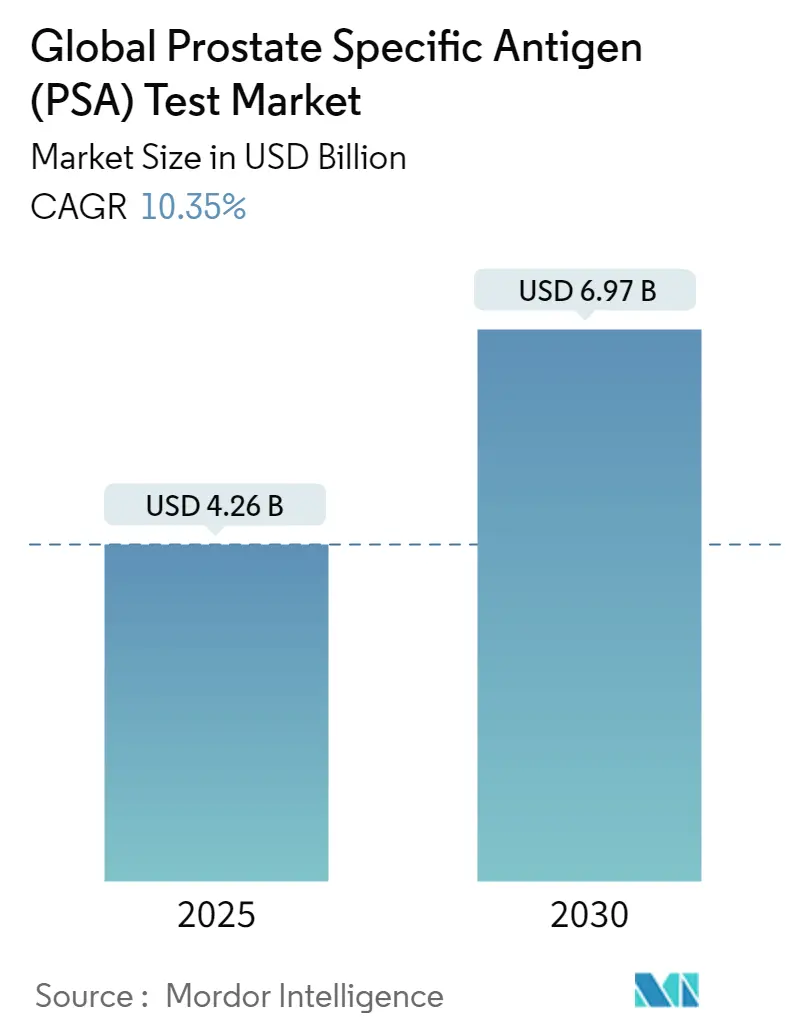
Global Prostate Specific Antigen (PSA) Test Market Analysis by Mordor Intelligence
The Global Prostate Specific Antigen Test Market size is estimated at USD 4.26 billion in 2025, and is expected to reach USD 6.97 billion by 2030, at a CAGR of 10.35% during the forecast period (2025-2030).
The COVID-19 outbreak has impacted non-COVID illnesses, such as cancer. The number of prostate cancer diagnoses decreased during the first COVID-19 wave. This trend was mostly restored in the second half of 2020. However, the number remained lower than predicted. According to a study released in April 2022 titled "Impact of the COVID-19 Outbreak on Prostate Cancer Care in the Netherlands" the first COVID-19 wave saw a 17 percent decrease in prostate cancer diagnoses. From May onwards, the number of diagnoses began to return to around 95% of what was projected by the end of 2020. Hence, market players faced numerous challenges as disruptions in the supply chain were observed. However, cancer care and maintaining high standards of diagnosis and treatment have been the major priorities of the national healthcare bodies and international healthcare systems worldwide.
The increasing prevalence of prostate cancer is a major factor that propels the demand for diagnostic products. Prostate cancer is the most common cancer among men, except for skin cancer. As per the American Society of Clinical Oncology (ASCO) updates from August 2022, prostate cancer is the world's fourth most commonly diagnosed cancer. As per the ASCO, an estimated 268,490 men in the United States were diagnosed with prostate cancer in 2021. Also, according to the same source, an estimated 1,414,259 people were diagnosed with prostate cancer in 2020. Hence, this creates a need for an advanced product that is projected to support the market growth over the forecast period.
The development of technologically advanced products is also driving the market. For example, a highly portable and quick prostate cancer screening kit could provide early warning to communities with a higher incidence of prostate cancer, such as African American males with limited health care access. The low-cost proof-of-concept test combines a test strip and a compact cube-shaped 1.6-inch reader to quantify a prostate cancer marker known as the prostate-specific antigen (PSA) from a drop of blood in minutes.
In addition, the launch of several studies to better prostate cancer diagnostics is aiding market expansion. For instance, new hospitals throughout England planned to join a groundbreaking trial in September 2021 that attempts to improve the analysis of prostate cancer samples using Artificial Intelligence. Six new NHS trusts were thought to kick-start this groundbreaking trial, which will be funded as part of the Eur140 million NHSX AI in Health and Care awards. Galen Prostate is the name of the AI technology developed by Ibex Medical Analytics, a health tech startup. The money will allow hospitals to compare the AI results against a pathologist's study using biopsies from 600 males over 14 months. AI technology can shorten diagnosis timelines, increase biopsy analysis accuracy, and free up clinicians' important time.
Besides, market players are also taking initiatives that are contributing to the market's growth. For instance, in June 2021, Lucida Medical, a Cambridge startup, announced that it had joined the Edison Accelerator, a program created by GE Healthcare in collaboration with Wayra UK to promote early-stage and technologically sophisticated enterprises creating AI solutions for healthcare. Lucida Medical's participation in the Edison Accelerator program is a significant step toward the company's goal of disrupting the cancer diagnostic pathway with technology that more accurately detects prostate cancer by analyzing MRI, allowing radiologists to save time and patients to receive the best possible diagnosis and treatment.
The availability of products in the market has also contributed to the market growth. For instance, in March 2020, Proclarix, a blood-based test for prostate cancer diagnosis, was made commercially available in Europe by Proteomedix, a Swiss cancer diagnostics business.
Thus, owing to the abovementioned factors, the market is expected to show growth over the forecast period. However, the high cost of diagnosis significantly hinders the market's growth.
Global Prostate Specific Antigen (PSA) Test Market Trends and Insights
The Preliminary Test Segment is Expected to Hold a Major Market Share in the Prostate-Specific Antigen Test Market
PSA screening has been more widely available, allowing for earlier detection and treatment, which has resulted in higher survival rates. PSA testing discovers previously undetected and tiny lesions that may or may not progress to a more advanced cancer stage. Most men over 50 and those at risk of acquiring the disease should get these tests since they help diagnose the disease before serious symptoms arise.
The increasing incidence of prostate cancer among men creates the need for advanced diagnostic products, thereby driving the market's growth. Prostate cancer is the most common cancer in American men. As per the American Cancer Society updates from January 2022, it is estimated that about 268,490 new cases of prostate cancer will be diagnosed in the United States in 2022.
Moreover, according to Globocan 2020, prostate cancer is the second most common type of cancer in the male population. As per the data published by the same source, the prevalence of prostate cancer was found to be 1,414,259, or 7.3% of the total cancer cases worldwide. As prostate cancer is more common in the elderly population, the rising aging population in the world is also expected to propel market growth. For instance, according to the World Population Aging Highlight of 2020 published by the United Nations, the global geriatric population (people age 65 years or more) was 727 million in 2020, and it is expected to reach 1.5 billion by the end of 2050. Thus, the increasing number of prostate cancer cases is expected to increase the demand for the preliminary test, thus, driving the segment's growth.
As a result of the aforementioned factors, the preliminary test segment is predicted to develop significantly throughout the forecast period.
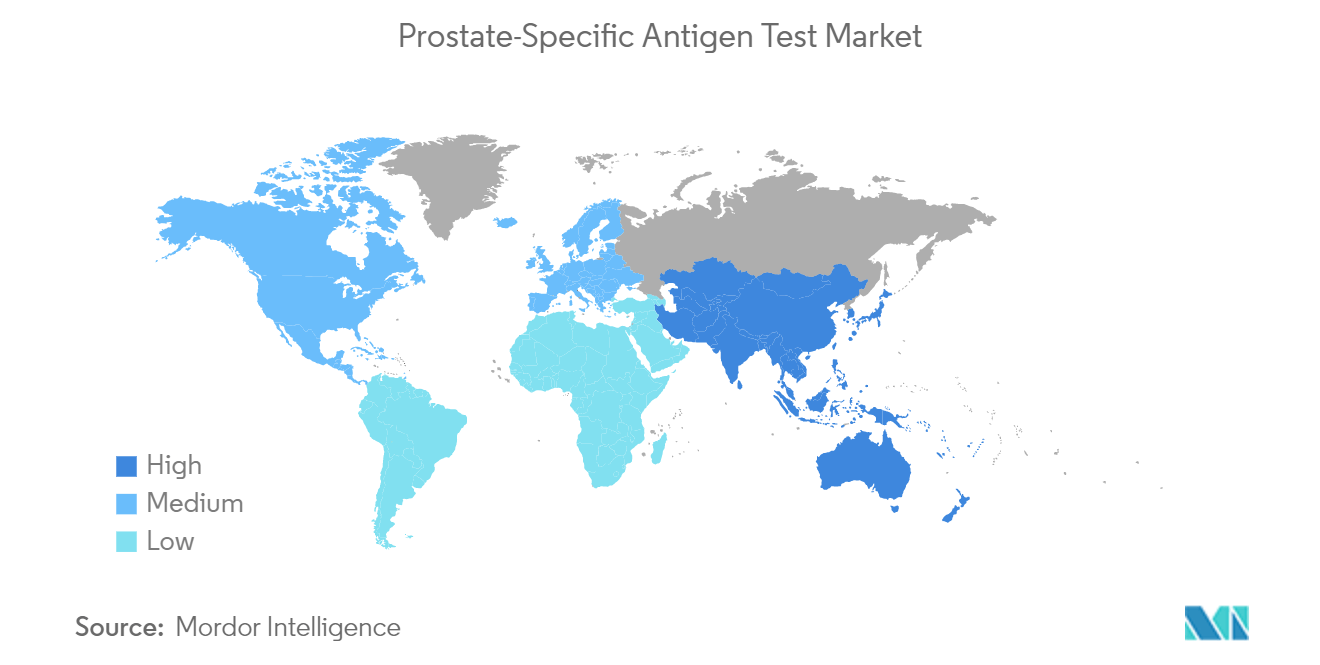
North America is Expected to Hold a significant share in the market and is expected to do the Same in the Forecast Period.
North America is anticipated to hold a significant market share, owing to the factors such as the increasing prevalence of prostate cancer, coupled with the rising awareness and increasing research and development in this region. However, the current impact of the COVID-19 pandemic on cancer care in the United States has resulted in decreases and delays in identifying new cancers and the delivery of treatment. As per the research article published in October 2021, "Impact of COVID-19 on Cancer Care: How the Pandemic Is Delaying Cancer Diagnosis and Treatment for American Seniors", significant decreases in screening for prostate cancer were observed in 2020 compared with 2019, with the most significant reduction occurring in April for prostate (56%) screenings. Thus, it is expected to increase the burden of prostate cancer in the region, in turn, it is expected to boost the growth of the market over the forecast period.
The market's expansion is further aided by favorable government backing in the United States. For example, Datar Cancer Genetics reported in February 2022 that its TriNetra-Prostate blood test, created to identify early-stage prostate cancer, has been designated a breakthrough device by the United States Food and Drug Administration. Paige Prostate, the first artificial intelligence (AI) based program developed to detect areas at risk of being malignant in prostate biopsy photos, was also approved by the United States Food and Drug Administration (FDA) in September 2021. This program allows pathologists to make a higher number of correct malignant tissue diagnoses, perhaps helping to address the growing number of cancer cases worldwide.
As a result of the aforementioned factors, the region is predicted to show significant growth during the forecast period.
Competitive Landscape
The prostate-specific antigen test market is fragmented and competitive and consists of several major players. Some of the players operating in the market are Accuquik Test Kits, Bayer AG, Bio-Rad Laboratories, Inc., Beckman Coulter, Inc., Fujirebio (H.U. Group company), General Electric Company, Abcam PLC, Lomina AG, Laboratory Corporation of America Holdings, OPKO Health, Inc., Hanzhou Testsea biotechnology Co. LTD, and Proteomedix.
Global Prostate Specific Antigen (PSA) Test Industry Leaders
Bio-Rad Laboratories, Inc.
Accuquik Test Kits
OPKO Health, Inc.
bioMérieux SA
Beckman Coulter, Inc.
- *Disclaimer: Major Players sorted in no particular order
Recent Industry Developments
- In April 2022, The Milton and Carroll Petrie Department of Urology at Mount Sinai launched the Mount Sinai Robert F. Smith Mobile Prostate Cancer Screening Unit to support prostate health in the Black community.
- In March 2022, miR Scientific, a New York-based healthcare company dedicated to transforming global cancer management, and Leonie Hill Capital, a Singapore-based organization focused on large-scale, sophisticated healthcare access and distribution, announced the signing of a collaboration agreement to partner in the commercial launch of the award-winning miR Sentinel Prostate Cancer Test in Singapore and Southeast Asia.
Global Prostate Specific Antigen (PSA) Test Market Report Scope
As per the scope of this report, the prostate-specific antigen test is a blood test used to screen for prostate cancer. The test measures the amount of prostate-specific antigen (PSA) in the blood. PSA is a protein produced by both cancerous and noncancerous tissue in the prostate, a small gland that sits below a man's bladder. The Prostate-Specific Antigen Test Market is Segmented by Test Type (Preliminary Tests, and Confirmatory Tests (Pca3 Test, Trans-Rectal Ultrasound, and Biopsy)), and Geography (North America, Europe, Asia-Pacific, Middle East, and Africa, and South America). The market report also covers the estimated market sizes and trends for 17 countries across major global regions. The report offers the value (USD million) for the above segments.
| Preliminary Tests | |
| Confirmatory Tests | Pca3 Test |
| Trans-Rectal Ultrasound | |
| Biopsy |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle-East and Africa | GCC |
| South Africa | |
| Rest of Middle-East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Test Type | Preliminary Tests | |
| Confirmatory Tests | Pca3 Test | |
| Trans-Rectal Ultrasound | ||
| Biopsy | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle-East and Africa | GCC | |
| South Africa | ||
| Rest of Middle-East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||
Key Questions Answered in the Report
How big is the Global Prostate Specific Antigen (PSA) Test Market?
The Global Prostate Specific Antigen (PSA) Test Market size is expected to reach USD 4.26 billion in 2025 and grow at a CAGR of 10.35% to reach USD 6.97 billion by 2030.
What is the current Global Prostate Specific Antigen (PSA) Test Market size?
In 2025, the Global Prostate Specific Antigen (PSA) Test Market size is expected to reach USD 4.26 billion.
Who are the key players in Global Prostate Specific Antigen (PSA) Test Market?
Bio-Rad Laboratories, Inc., Accuquik Test Kits, OPKO Health, Inc., bioMérieux SA and Beckman Coulter, Inc. are the major companies operating in the Global Prostate Specific Antigen (PSA) Test Market.
Which is the fastest growing region in Global Prostate Specific Antigen (PSA) Test Market?
Asia-Pacific is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Global Prostate Specific Antigen (PSA) Test Market?
In 2025, the North America accounts for the largest market share in Global Prostate Specific Antigen (PSA) Test Market.
What years does this Global Prostate Specific Antigen (PSA) Test Market cover, and what was the market size in 2024?
In 2024, the Global Prostate Specific Antigen (PSA) Test Market size was estimated at USD 3.82 billion. The report covers the Global Prostate Specific Antigen (PSA) Test Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Global Prostate Specific Antigen (PSA) Test Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
Global Prostate Specific Antigen (PSA) Test Market Report
Statistics for the 2025 Global Prostate Specific Antigen (PSA) Test market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Global Prostate Specific Antigen (PSA) Test analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.




