Pharmaceutical Analytical Testing Market Size
Pharmaceutical Analytical Testing Market Analysis
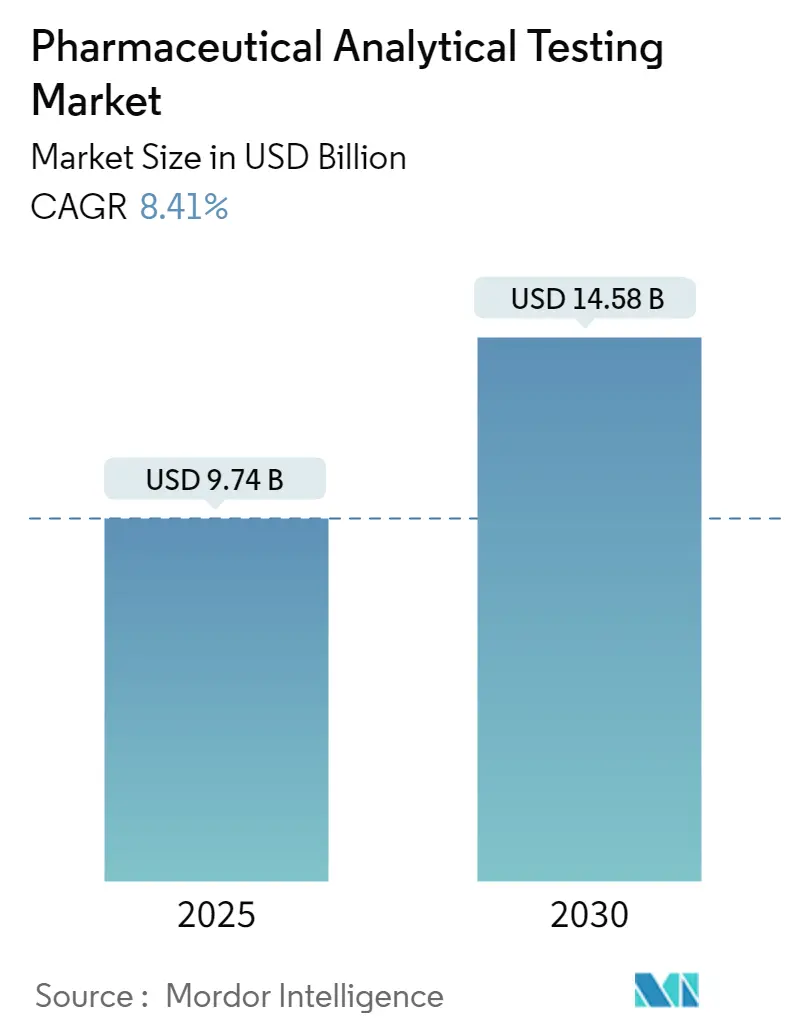
The Pharmaceutical Analytical Testing Market size is estimated at USD 9.74 billion in 2025, and is expected to reach USD 14.58 billion by 2030, at a CAGR of 8.41% during the forecast period (2025-2030).
Various factors, such as the increasing number of clinical trials, the rising focus on Pharmaceutical Analytical Testing Services of biologics and biosimilars, and the increased trend of outsourcing laboratory testing services, are expected to boost the market growth over the forecast period.
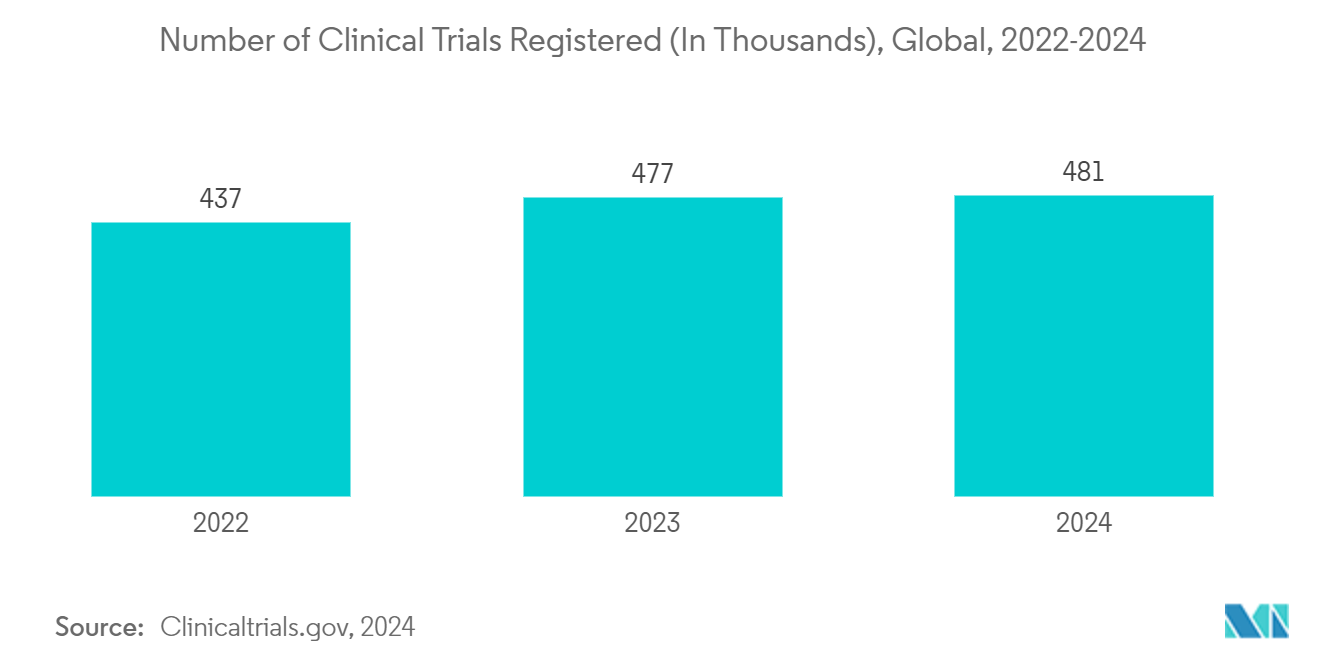
The increase in clinical trials for developing new drugs is anticipated to boost the need for Pharma Testing Services and contribute to market growth. For instance, in February 2024, according to clinicaltrials.gov, there were 481,599 studies from locations comprising 50 states and 223 countries. Additionally, in December 2023, Mission Therapeutics, a United Kingdom-based company, received clinical trial authorization from the Medicines and Healthcare Products Regulatory Agency (MHRA) to evaluate the MTX325 in a Phase-I study for Parkinson's disease. Thus, with the high number of clinical trials, it is anticipated that Pharmaceutical Analytical Services are likely to be employed and contribute to market growth.
Additionally, initiatives taken by health regulatory bodies, such as the World Health Organization, to boost clinical trial activities are expected to propel the growth of the Pharmaceutical Analytical Testing Services Market over the forecast period. For instance, in November 2023, the World Health Organization Global Clinical Trials forum presented the global vision for developing clinical research infrastructure in various therapeutic areas such as cancer, cardiovascular disease, and metabolic and neurological disorders. It facilitates the ethics and regulatory approval process. Therefore, government initiatives for promoting clinical trials and research are anticipated to fuel the adoption of Pharmaceutical Analytical Services, contributing to market growth.
Moreover, the growing emphasis on analytical assessments of biosimilars and biologics drives the demand for Pharmaceutical Analysis Services and contributes to market growth. For instance, in February 2022, an article published in the Frontiers in Bioengineering and Biotechnology mentioned that analytical and biosimilarity assessments were conducted to ensure the approval of biosimilars worldwide. Thus, Pharmaceutical Analytical Services are highly employed, with a growing focus on analytical testing to ensure the approval process and safety of the molecules, thus driving the market growth.
The market players' strategic activities, such as collaborations and partnerships among Pharmaceutical Testing Companies, are anticipated to boost their capabilities to provide Pharmaceutical Analytical Services, which is expected to bolster the market growth. For instance, in July 2023, Recipharm opened a new analytical laboratory in Bengaluru, India, to promote its testing capabilities for nitrosamines, extractable and leachable, and elemental impurity testing worldwide. Additionally, in December 2023, Sterling Accuris Diagnostics acquired Vaibhav Analytical Services, a provider of analytical testing services based in Ahmedabad, India. With this acquisition, the company intends to expand its services to new horizons, especially in the realm of Pharmaceutical Analytical Testing. Such developments are estimated to propel market growth over the forecast period.
Therefore, owing to the aforementioned factors, such as the rise in clinical trials, growing attention on analytical testing of biologics and biosimilars, and the rising trend of outsourcing laboratory testing services, the Pharmaceutical Outsourcing Analytical Testing Market is anticipated to grow over the forecast period. However, the complex regulatory framework for maintaining laboratories and challenges in developing proper analytical techniques will likely impede market growth.
Pharmaceutical Analytical Testing Market Trends
Stability Testing Segment is Expected to Exhibit a Significant Market Growth Over the Forecast Period
Pharmaceutical Stability Testing is conducted to evaluate the capability of the drug to retain its properties throughout its shelf-life. The stability studies of pharmaceutical products are one of the most important parameters for developing new drugs and formulations. The stability testing is done based on five parameters: chemical, physical, microbiological, therapeutic, and toxicity. The degradation of any of these parameters can lead to health hazards.
Factors such as the growing government initiatives and guidelines to support stability testing, advantages of stability testing, and key strategies adopted by the market players are expected to boost the growth of the Pharmaceutical Stability & Storage Services Market over the forecast period.
The regulatory bodies are involved in developing guidelines to ensure the stability of the testing of pharmaceutical products. For instance, in July 2023, the European Medicines Agency published guidelines on stability testing for active ingredients and pharmaceutical products to ensure their safety. These guidelines mandate pharmaceutical companies to conduct stability testing, which is expected to drive segment growth in the Pharmaceutical Quality Control Market over the forecast period.
Additionally, various studies were conducted to perform stability testing with different analytical techniques for determining drug quality, efficacy, and safety. For instance, in March 2023, a study published in the Turkish Journal of Pharmaceutical Sciences reported that stability testing was conducted for the biosimilar monoclonal antibodies candidate (TUR01) with advanced analytical techniques such as isoelectric focusing on capillary electrophoresis, capillary electrophoresis-sodium dodecyl sulfate, and others.
In addition to the above context, the study showed drug stability at ≤-65°C and 5 ± 3°C for at least 18 months. Thus, Pharmaceutical Analytical Testing ensures that the drugs are highly stable, which in turn is expected to propel the adoption of such techniques across drug manufacturing companies, as it provides the safety and quality of biosimilars, hence contributing to segment growth.
Key players' strategic initiatives, such as product launches, approvals, and partnerships, are expected to fuel the segment's growth. For instance, in November 2023, CatSci Ltd established a partnership with Reach Separations in which the company can leverage CatSci's expertise in analytical science covering various services, including International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) stability testing.
Additionally, in July 2023, ten23 Health launched quality control services in Switzerland for product release and stability testing of clinical and commercial drugs as per the international current good manufacturing practice (cGMP) standards. Such developments are estimated to boost the segment growth during the forecast period.
Therefore, the aforementioned factors, such as government initiatives, market players' strategies, and the effectiveness of Pharmaceutical Stability Testing and Pharmaceutical Laboratory Services, are anticipated to drive the segment growth over the forecast period.
North America Holds a Significant Share in the Market and Expected to do Same During The Forecast Period
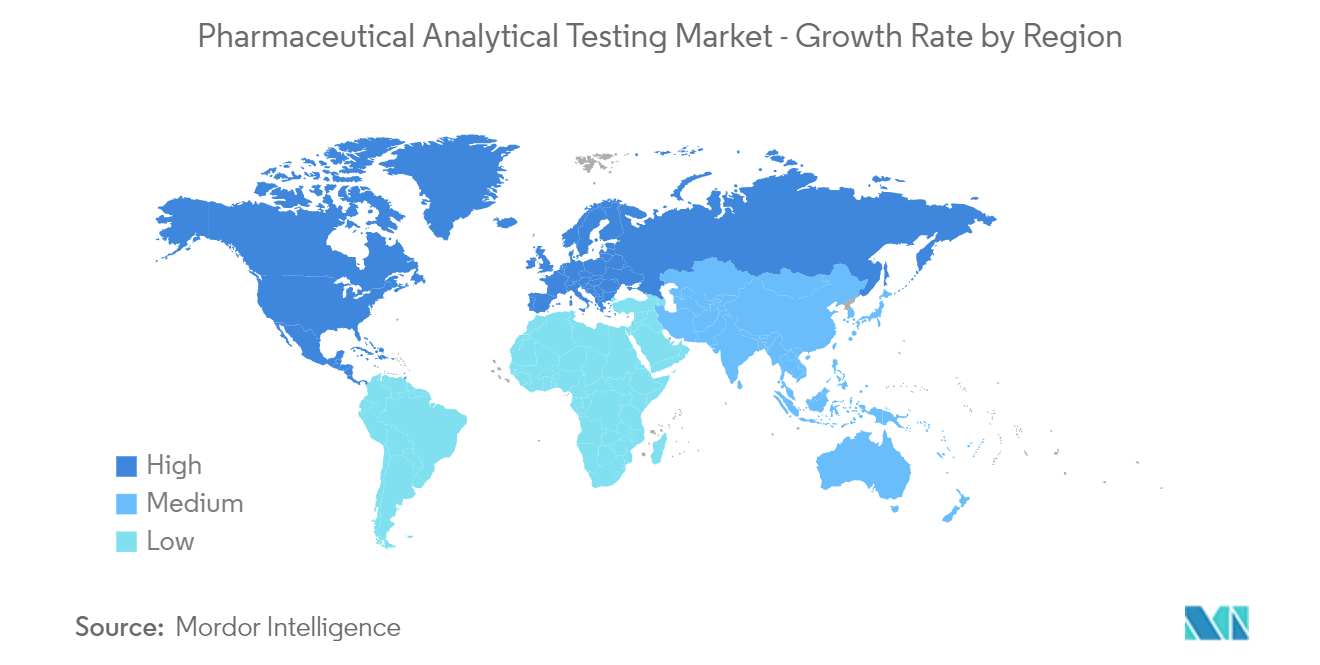
North America is expected to occupy a significant market share in the Pharmaceutical Analytical Testing Market owing to the increasing number of R&D activities, investments, and strategic initiatives taken by the market players in the region.
The region's need for Pharmaceutical Analytical Services is growing due to increased clinical research activities and drug approvals. For instance, in January 2024, the United States Food and Drug Administration's Center for Drug and Evaluation approved 55 new drugs in 2023, which are indicated for treating various infectious diseases. Also, as of February 2024, about 146,311 clinical trials were registered in the United States, contributing 30% of the total registered clinical trials. Thus, the significant number of drug approvals and clinical trials in the region is anticipated to boost the market growth in the area during the forecast period.
Furthermore, growing investments in clinical research fuel the demand for Pharmaceutical Analytical Testing Services to develop safe and high-quality pharmaceutical products and drive market growth in the region. For instance, the Pharmaceutical Research and Manufacturers of America (PhRMA) trade group represents the leading biopharmaceutical research companies in the United States. In 2022, its member companies' research and development (R&D) expenditure reached about USD 101 billion worldwide, in which the United States accounted for 21% of R&D investment. Therefore, significant research and development expenditures for conducting clinical trials will likely employ Pharmaceutical Analytical Testing to ensure safety at each drug development stage and are expected to propel the market growth.
The strategic initiatives, including product launches, approvals, acquisitions, and partnerships, not only enhance the capabilities and service offerings of key market players but also drive the broader adoption of Pharmaceutical Analytical Services. For instance, in January 2024, Alcami Corporation acquired Pacific Pharmaceutical Services, Inc. to improve its pharma storage and services capabilities in Nevada, ensuring the stability and security of storing pre-clinical and clinical materials. Additionally, in December 2022, Pharmacopeia opened its advanced manufacturing technology lab in Richmond, Virginia, with the launch of analytical services to boost drug manufacturing activities and medication supply chain resilience. Such developments are estimated to propel market growth in the region.
Therefore, the increasing number of clinical trials, increase in research and development expenditures, and product launches are expected to boost the demand for Pharmaceutical Analytical Testing Market growth in the North American region, ensuring high-quality and safe drug development.
Pharmaceutical Analytical Testing Industry Overview
The Pharmaceutical Analytical Testing Market is fragmented in nature due to the presence of several companies globally. Various strategies are adopted by the market players such as product launches, collaborations, and acquisitions to expand their business in the market. The studied market consists of several international and local companies that hold most of the market shares and are well known. Some of the key players in the market are Steris, Intertek Pharmaceutical Services, Pace Analytical Services, SGS SA, and Boston Analytical, among others.
Pharmaceutical Analytical Testing Market Leaders
-
Steris
-
Intertek Pharmaceutical Services
-
Pace Analytical Services
-
SGS SA
-
Boston Analytical
- *Disclaimer: Major Players sorted in no particular order
Pharmaceutical Analytical Testing Market News
- March 2024: LGM Pharma invested over USD 2 million to expand its analytical testing services and include drug delivery suppository manufacturing capabilities by 50% in its facility in Rosenburg, Texas.
- January 2024: Kindeva Drug Delivery increased its analytical service capabilities by launching a new global business unit, which provides integrated and stand-alone analytical support to pharmaceutical, biopharmaceutical, and medical device companies.
Pharmaceutical Analytical Testing Industry Segmentation
As per the scope of the report, pharmaceutical analytical testing is majorly used in pharmaceutical and biopharmaceutical companies for a series of clinical trials on newly manufactured drugs to ensure safety. This pharmaceutical analytical testing market is segmented by service type and geography. By service type, the market is segmented into bioanalytical testing, method development & validation, stability testing, drug substances testing, and other service types. By geography, the market is segmented into North America, Europe, Asia-Pacific, the Middle East and Africa, and South America. For each segment, the market size is provided in terms of USD value.
| By Service Type | Bioanalytical Testing | ||
| Method Development & Validation | |||
| Stability Testing | |||
| Drug Substances Testing | |||
| Other Service Types | |||
| Geography | North America | United States | |
| Canada | |||
| Mexico | |||
| Europe | Germany | ||
| United Kingdom | |||
| France | |||
| Italy | |||
| Spain | |||
| Rest of Europe | |||
| Asia-Pacific | China | ||
| Japan | |||
| India | |||
| Australia | |||
| South Korea | |||
| Rest of Asia-Pacific | |||
| Middle East and Africa | GCC | ||
| South Africa | |||
| Rest of Middle East and Africa | |||
| South America | Brazil | ||
| Argentina | |||
| Rest of South America | |||
| Bioanalytical Testing |
| Method Development & Validation |
| Stability Testing |
| Drug Substances Testing |
| Other Service Types |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
Pharmaceutical Analytical Testing Market Research FAQs
How big is the Pharmaceutical Analytical Testing Market?
The Pharmaceutical Analytical Testing Market size is expected to reach USD 9.74 billion in 2025 and grow at a CAGR of 8.41% to reach USD 14.58 billion by 2030.
What is the current Pharmaceutical Analytical Testing Market size?
In 2025, the Pharmaceutical Analytical Testing Market size is expected to reach USD 9.74 billion.
Who are the key players in Pharmaceutical Analytical Testing Market?
Steris, Intertek Pharmaceutical Services, Pace Analytical Services, SGS SA and Boston Analytical are the major companies operating in the Pharmaceutical Analytical Testing Market.
Which is the fastest growing region in Pharmaceutical Analytical Testing Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2025-2030).
Which region has the biggest share in Pharmaceutical Analytical Testing Market?
In 2025, the North America accounts for the largest market share in Pharmaceutical Analytical Testing Market.
What years does this Pharmaceutical Analytical Testing Market cover, and what was the market size in 2024?
In 2024, the Pharmaceutical Analytical Testing Market size was estimated at USD 8.92 billion. The report covers the Pharmaceutical Analytical Testing Market historical market size for years: 2021, 2022, 2023 and 2024. The report also forecasts the Pharmaceutical Analytical Testing Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Our Best Selling Reports
Pharmaceutical Analytical Testing Industry Report
The global pharmaceutical analytical testing services market is experiencing significant growth, driven by the increasing demand for analytical testing services and the rise in drug approvals and clinical trials. Enhanced government efforts to bolster analytical testing capabilities, alongside the growing need for vaccine testing, are major contributors to this market expansion. The sector's focus on bioanalytical testing and method development is crucial in addressing the urgent need for meticulous drug testing to ensure the safety and efficacy of new pharmaceuticals.
Despite challenges such as developing appropriate analytical techniques and navigating a complex regulatory landscape, there are lucrative opportunities in stability testing and the trend towards outsourcing laboratory testing services. North America leads the market, underscored by intense R&D activities and numerous clinical trials, signaling a promising future for innovation within the healthcare analytical testing services market.
The market analysis highlights the importance of industry reports and market data in understanding the market size and trends. The industry overview provides insights into the market segmentation and the value it brings. Market leaders play a crucial role in shaping the market outlook and driving market growth. The industry statistics and market forecast offer a comprehensive market review and predictions for the future.
Industry information and research companies provide valuable industry research, contributing to the overall industry sales and market value. The industry outlook and market predictions are essential for understanding the market trends and growth rate. The market forecast and market overview are pivotal in understanding the market segmentation and the industry's future direction.
The report example and report pdf offer detailed insights into the market, providing a robust foundation for industry analysis. The market leaders and research companies are instrumental in driving market growth and ensuring the industry's success. The industry trends and market data are crucial for understanding the market's dynamics and making informed decisions.
Overall, the pharmaceutical analytical testing services market is poised for significant growth, with numerous opportunities for innovation and development. The industry's focus on meticulous drug testing, combined with the trend towards outsourcing and enhanced government efforts, ensures a bright future for the market.