Pediatric Clinical Trials Market Size and Share
Pediatric Clinical Trials Market Analysis by Mordor Intelligence
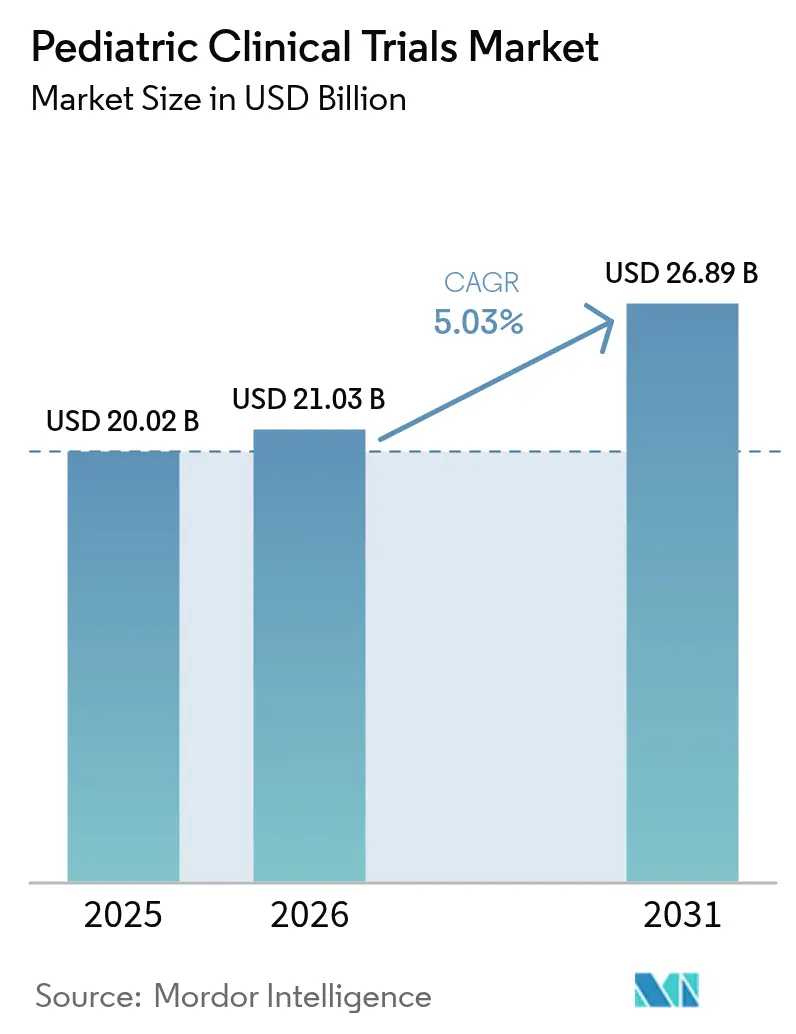
The pediatric clinical trials market size was valued at USD 20.02 billion in 2025 and estimated to grow from USD 21.03 billion in 2026 to reach USD 26.89 billion by 2031, at a CAGR of 5.03% during the forecast period (2026-2031). Strong regulatory incentives, notably the US Pediatric Research Equity Act (PREA) and the EU Paediatric Regulation, keep trial volumes rising as every new molecular entity targeting children must present age-appropriate evidence. Mandatory early evaluation of oncology drugs under the RACE for Children Act sustains a high share of cancer-focused protocols. Parallel trends—including rising chronic disease prevalence among children, the shift toward decentralized and AI-enabled study designs, and deeper outsourcing to pediatric-specialist contract research organizations (CROs)—are expanding both the scope and geographic reach of the pediatric clinical trials market. North America remains the largest regional hub, but Asia-Pacific is accelerating fastest as regulators in South Korea, Taiwan, and Australia streamline review pathways and introduce fiscal incentives for sponsors.
Key Report Takeaways
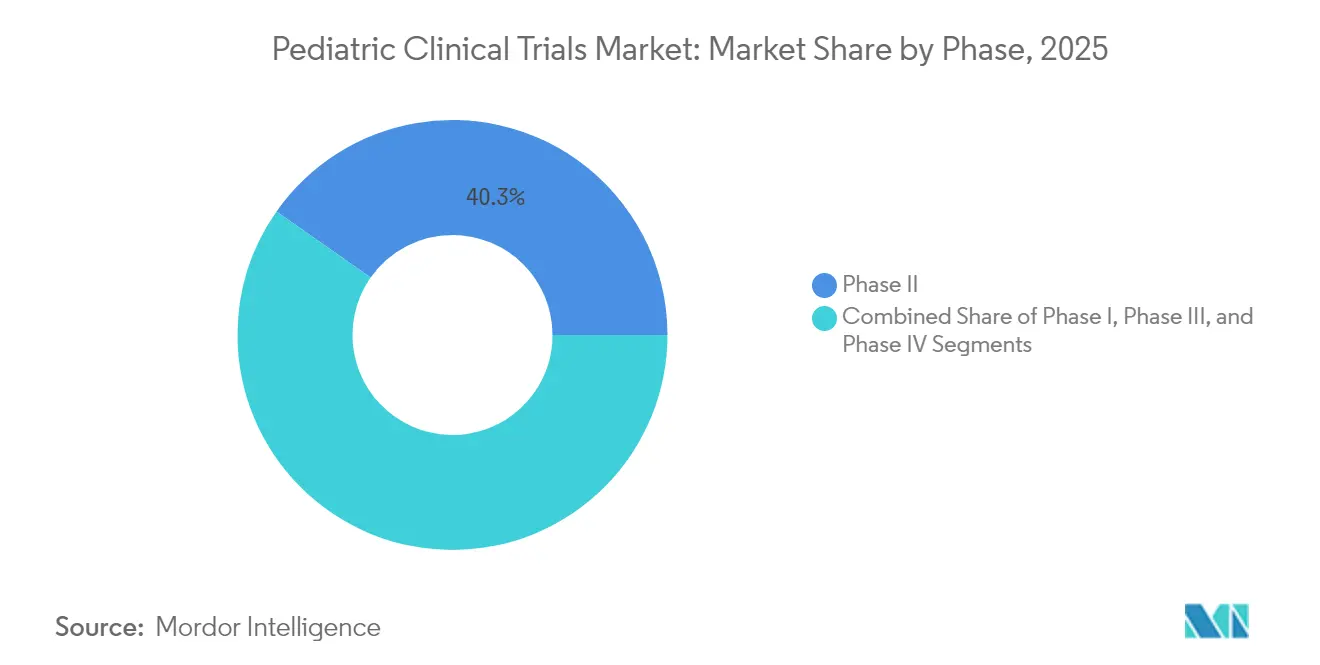
- By phase, Phase II accounted for 40.26% of pediatric clinical trials market share in 2025, while Phase I is projected to expand at a 6.96% CAGR through 2031.
- By study design, interventional drug studies held 64.93% share of the pediatric clinical trials market size in 2025, whereas observational cross-sectional studies will advance at a 7.44% CAGR to 2031.
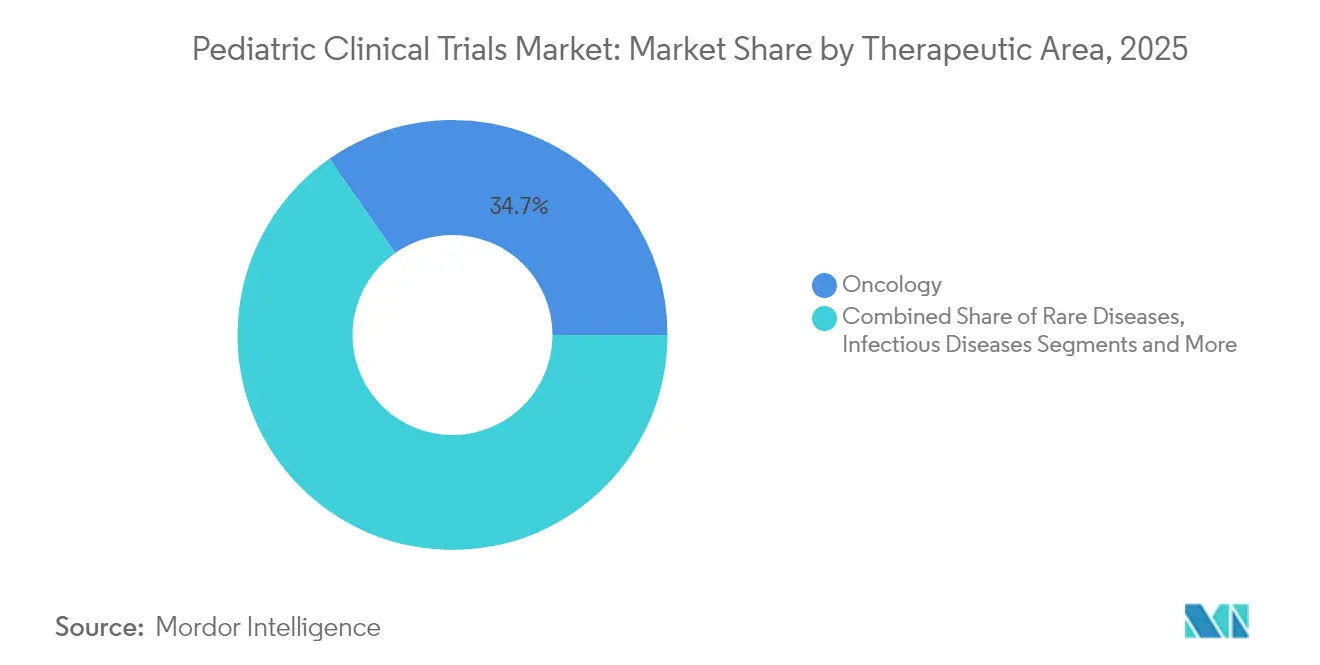
- By therapeutic area, oncology captured 34.71% of pediatric clinical trials market share in 2025, and rare diseases are forecast to grow at an 8.26% CAGR over the same horizon.
- By sponsor type, pharmaceutical and biopharmaceutical companies represented 48.72% of pediatric clinical trials market size in 2025; government and academic institutions show the highest CAGR at 8.15% through 2031.
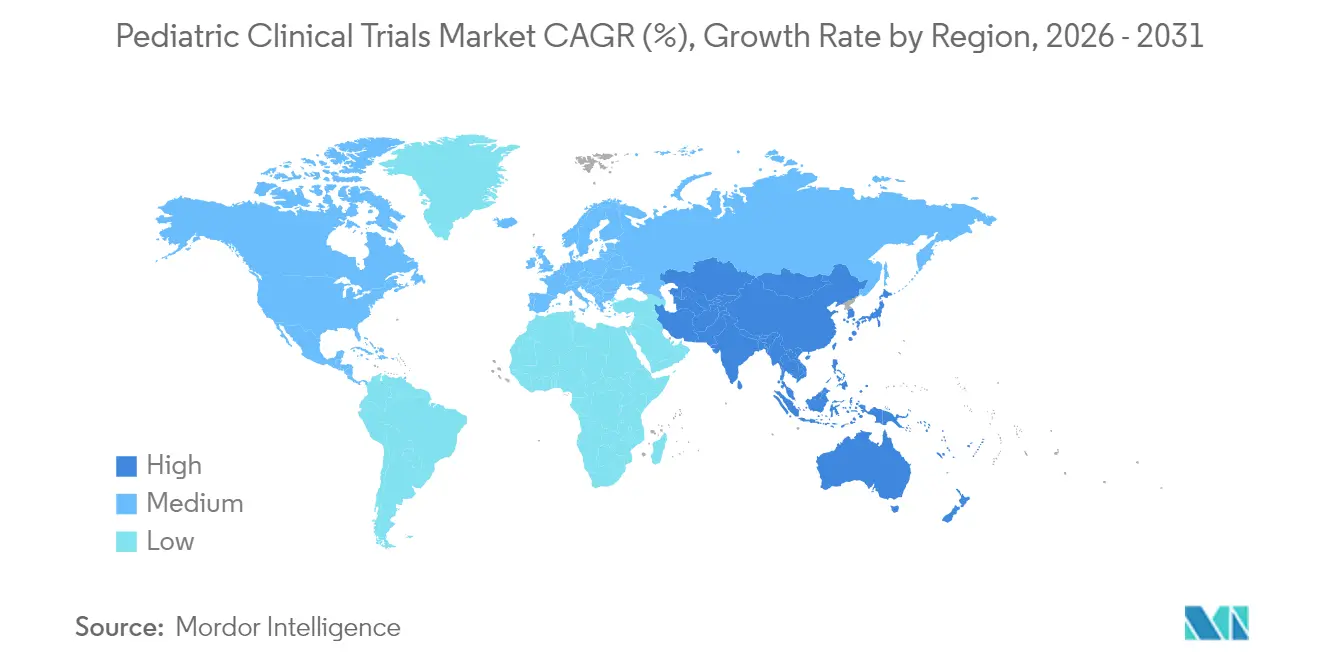
- By geography, North America led with 39.02% market share in 2025, whereas Asia-Pacific is expected to register a 7.18% CAGR between 2026 and 2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Pediatric Clinical Trials Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Regulatory incentives (US PREA, EU Paediatric Regulation) | +1.8% | Global, strongest in North America & EU | Long term (≥ 4 years) |
| Rising prevalence of chronic pediatric diseases | +1.2% | Global, higher burden in developed markets | Medium term (2-4 years) |
| Outsourcing surge to pediatric-specialist CROs | +0.9% | Global, led by North America, expanding to APAC | Medium term (2-4 years) |
| Decentralized/virtual trial adoption for children | +0.7% | North America & EU early adoption, APAC following | Short term (≤ 2 years) |
| AI-driven adaptive designs cutting sample size | +0.5% | North America & EU leading, selective APAC adoption | Medium term (2-4 years) |
| RACE for Children Act-led oncology trial boom | +0.4% | US-focused with global spillover effects | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Regulatory Incentives (US PREA, EU Paediatric Regulation)
Long-standing mandates have reshaped drug-development economics by requiring Pediatric Investigation Plans in Europe and pediatric study plans under PREA in the United States. Convergence continues with the FDA’s 2024 acceptance of ICH E11A extrapolation guidelines, enabling streamlined dose-finding that leverages adult data where scientifically justified[1]WCG Clinical, “The FDA Accepts ICH E11A on Pediatric Extrapolation,” wcgclinical.com. These policies reduce sequential adult-to-child timelines, lifting demand for specialized pediatric protocols, and the 2025 Innovation in Pediatric Drugs Act proposes stronger enforcement, signaling durable growth.
Rising Prevalence of Chronic Pediatric Diseases
US survey data show persistent increases in asthma and mental-health diagnoses among youth, directly expanding the therapeutic pipeline for pediatric respiratory, endocrine, and neurological agents. Asthma alone continues to impose disparate burdens on non-Hispanic Black children, highlighting equity gaps that prospective studies are beginning to address. The earlier onset and longer treatment windows typical of chronic childhood diseases amplify the requirement for child-friendly formulations and robust long-term safety datasets, prompts that collectively boost the pediatric clinical trials market.
Outsourcing Surge to Pediatric-Specialist CROs
Trial complexity is motivating sponsors to partner with CROs that field board-certified pediatricians and family-centric recruitment teams. IQVIA has already completed 359 pediatric studies across 101 countries, enrolling 221,000 children. ICON manages 399 pediatric trials spanning 117,000 participants and 16,630 sites. Such specialization supports age-appropriate pharmacokinetics, decentralized consent workflows, and innovative dosing strategies—capabilities now recognized as essential in the pediatric clinical trials market.
Decentralized/Virtual Trial Adoption for Children
Hybrid and fully decentralized models are overcoming distance, time, and school-schedule barriers that deter families from participation. The FDA’s 2024 guidance explicitly endorses remote data capture to broaden access for underrepresented pediatric populations. ICON reports a 10% recruitment lift in pediatric studies that incorporate electronic patient-reported outcomes, with 90% of enrolled families preferring travel times under one hour when occasional on-site visits remain necessary.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Ethical complexities & informed-consent hurdles | -0.8% | Global, stricter in EU & developed markets | Long term (≥ 4 years) |
| Limited recruitable patient pools | -1.1% | Global, pronounced in rare diseases | Medium term (2-4 years) |
| Scarcity of child-friendly drug formulations | -0.6% | Global, variable by region | Medium term (2-4 years) |
| Post-pandemic site staffing shortages | -0.9% | North America & EU, selective APAC impact | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Ethical Complexities & Informed-Consent Hurdles
Dual requirements for parental permission and age-appropriate assent introduce added administrative layers that can delay study start-up and drive costs. A multicenter Canadian survey found wide divergence in feasibility perceptions for obtaining assent within 48 hours of PICU admission. International trials face further institutional review board variation; the PARITY orthopedic oncology study secured approvals from only 46 of 91 interested sites due to resource constraints. Harmonized consent templates and electronic documentation are gradually easing this burden but will remain a headwind for the pediatric clinical trials market.
Limited Recruitable Patient Pools
Systematic reviews show that merely 10% of eligible children enroll in trials, with socioeconomic status, language, and prior therapy heavily influencing participation. Children with rare diseases pose an even greater challenge as prevalence per indication often falls below 2 per 100,000. Efforts such as telehealth-enabled screening and community-based satellite sites are improving reach, yet recruitment remains the most significant brake on pediatric clinical trials market size expansion.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Phase: Early-Stage Trials Drive Innovation Pipeline
Phase II maintained the largest slice of the pediatric clinical trials market size at 40.26% in 2025 as sponsors used proof-of-concept readouts to fine-tune age-appropriate dosing and accrual strategies. Phase I trials, spurred by the RACE Act and ICH E11A framework, are projected to deliver a 6.96% CAGR by 2031 as sponsors move earlier to test mechanism-based therapies in children. Adaptive, model-based escalation designs minimize exposure while accelerating go/no-go decisions—a practice now standard in oncology and rare metabolic disorders.
Phase III programs remain pivotal for labeling, yet heightened reliance on extrapolation data permits smaller randomized cohorts. As a result, Phase III’s proportional pediatric clinical trials market share could erode marginally even though absolute study counts rise. Post-marketing Phase IV surveillance is expanding for chronic therapies where lifetime exposure necessitates pharmacovigilance across developmental stages, leveraging real-world data and registries to capture growth and neurocognitive endpoints.
Note: Segment shares of all individual segments available upon report purchase
By Study Design: Drug Interventions Dominate Amid Observational Growth
Interventional drug protocols held 64.93% pediatric clinical trials market share in 2025, reflecting regulatory imperatives for child-specific pharmacokinetic and safety data. Observational cross-sectional studies will contribute the fastest 7.44% CAGR to 2031 as regulators accept real-world evidence to support supplemental labeling, especially in ultra-rare diseases where randomized trials are infeasible.
Device interventions, although smaller in count, are rising steadily in diabetes tech and neuromonitoring, propelled by the need to validate sensor accuracy and alert thresholds in infants. Behavioral and cohort studies complement drug trials by characterizing adherence patterns, school attendance impacts, and psychosocial outcomes critical to holistic benefit-risk assessment in the pediatric clinical trials industry.
By Therapeutic Area: Oncology Leadership Challenged by Rare Disease Innovation
Oncology preserved 34.71% of pediatric clinical trials market share in 2025 on the back of molecularly targeted therapeutics and immunotherapies mandated under the RACE Act. Rare disease programs, however, are set to eclipse all others in growth with an 8.26% CAGR as sponsors pursue Priority Review Voucher incentives and leverage gene-editing platforms to address monogenic disorders.
Infectious-disease trials pivoted after the COVID-19 pandemic toward RSV monoclonal antibodies such as nirsevimab, exemplifying accelerated licensure paths for preventive biologics in neonates. Respiratory, metabolic, and neurologic areas also show steady expansion, fueled by chronic burden trends and breakthroughs in gene-therapy vectors crossing the blood–brain barrier.
Note: Segment shares of all individual segments available upon report purchase
By Sponsor Type: Academic Institutions Accelerate Public Health Focus
Pharmaceutical and biopharmaceutical firms retained 48.72% pediatric clinical trials market size in 2025, largely to fulfill mandatory pediatric post-marketing requirements. Government and academic institutions, backed by NIH and EU Horizon grants, will log an 8.15% CAGR to 2031 as they target public-health gaps such as neonatal sepsis and adolescent mental health where commercial incentives are limited.
CROs gain traction as intermediaries, blending industry resources with academic mentorship programs for investigator training. Hybrid sponsor models—pharma supplying investigational product while universities lead protocol design—are flourishing, aligning economic efficiency with scientific rigor and broadening the pediatric clinical trials industry’s collaborative fabric.
Geography Analysis
North America commanded 39.02% of the pediatric clinical trials market in 2025 due to PREA-driven mandates, a dense network of children’s hospitals, and reliable reimbursement for trial-related procedures. Institutional capacity initiatives, such as Lurie Children’s plan to open a specialty pharmacy in 2026, reinforce integrated research-to-care models. Staffing shortages persist but are mitigated by remote-monitoring adoption and site-support alliances.
Asia-Pacific will post a 7.18% CAGR through 2031, buoyed by South Korea’s centralized IRB review, Taiwan’s fast-track approvals, and Australia’s decentralized trial guidelines that slash start-up times by up to three months. China’s expansion of its National Rare Disease List and investment in provincial referral networks further enlarge patient pools. Lower operational costs and rapidly digitizing healthcare records enhance the region’s attractiveness to multinational sponsors aiming to diversify recruitment.
Europe benefits from a harmonized regulatory environment via the Paediatric Committee (PDCO) and maintains robust academic-industry collaboration. Still, post-Brexit regulatory divergence demands duplicate submissions for UK sites, prolonging timelines compared with EU27. Emerging regions such as Latin America and the Middle East show incremental gains as governments upgrade research infrastructure and introduce tax incentives, but limited pediatric specialist density constrains complex trial execution for now.
Competitive Landscape
The pediatric clinical trials market is moderately fragmented. Top CROs deploy region-specific pediatricians, decentralized sampling logistics, and AI-enabled feasibility platforms to win full-service contracts. IQVIA’s global pediatric database underpins synthetic-control-arm generation, shortening trial durations for rare cancers. ICON expands its AI suite for study-startup forecasting and patient-identification algorithms, translating into quicker first-patient-in milestones.
Strategic alliances intensify: LEO Pharma’s five-year pact with ICON mobilizes 500 dermatology specialists under risk-share terms that link CRO fees to recruitment timelines. Technology disruptors such as Phesi and Pi Health license AI-driven site-selection engines to incumbents, while blockchain pilots from academic consortia test immutable consent tracking for minors. Acquisition activity continues as illustrated by Clario’s 2025 purchase of NeuroRx to bolster pediatric neuroimaging analytics[3]Axios, “Clinical Trials Company Clario Buys Imaging Specialist NeuroRx,” axios.com. Competitive advantage hinges on demonstrating integrated capabilities that compress timelines, respect child-centric ethical standards, and reduce per-patient cost.
White-space opportunities persist in decentralized home-nursing networks, culturally tailored consent tools, and adaptive-design statistical consulting. Companies that integrate these services within a scalable digital backbone are positioned to outpace peers as sponsors increasingly demand end-to-end pediatric solutions.
Pediatric Clinical Trials Industry Leaders
IQVIA
ICON plc
Thermo Fisher Scientific (PPD)
Syneos Health
Labcorp Drug Development (Covance)
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- June 2025: Biogen initiated dosing in the BRAVE Phase 3 study investigating omaveloxolone in children aged 2–15 years with Friedreich ataxia.
- September 2024: Signant Health joined IQVIA’s One Home for Sites program to unify eClinical solutions across decentralized pediatric studies.
Global Pediatric Clinical Trials Market Report Scope
As per the scope of the report, clinical trials conducted on children, for the research and development of new drugs, are called pediatric clinical trials. The Pediatric Clinical Trials market is segmented by Phase (Phase I, Phase II, Phase III, and Phase IV), Study Design (Treatment Studies and Observational Studies), Therapeutic Area (Respiratory Diseases, Infectious Diseases, Oncology, Diabetes, and Other Therapeutic Areas) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, and South America). The market report also covers the estimated market sizes and trends for 17 different countries across major regions, globally. The report offers the value (in USD million) for the above segments.
| Phase I |
| Phase II |
| Phase III |
| Phase IV |
| Interventional - Drug |
| Interventional - Device |
| Behavioral Trials |
| Observational - Cohort |
| Observational - Case-Control |
| Observational - Cross-Sectional |
| Oncology |
| Infectious Diseases |
| Respiratory Diseases |
| Endocrine & Metabolic (Diabetes) |
| Neurology |
| Rare Diseases |
| Pharma & Biopharma Companies |
| Contract Research Organizations |
| Government & Academic Institutions |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| South Korea | |
| Australia | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Phase | Phase I | |
| Phase II | ||
| Phase III | ||
| Phase IV | ||
| By Study Design | Interventional - Drug | |
| Interventional - Device | ||
| Behavioral Trials | ||
| Observational - Cohort | ||
| Observational - Case-Control | ||
| Observational - Cross-Sectional | ||
| By Therapeutic Area | Oncology | |
| Infectious Diseases | ||
| Respiratory Diseases | ||
| Endocrine & Metabolic (Diabetes) | ||
| Neurology | ||
| Rare Diseases | ||
| By Sponsor Type | Pharma & Biopharma Companies | |
| Contract Research Organizations | ||
| Government & Academic Institutions | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| South Korea | ||
| Australia | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the current size and growth outlook for the pediatric clinical trials market?
The pediatric clinical trials market is valued at USD 21.03 billion in 2026 and is projected to reach USD 26.89 billion by 2031, advancing at a 5.03% CAGR.
Which clinical trial phase holds the largest share and which is expanding fastest?
Phase II trials account for the largest 40.26% market share in 2025, while Phase I trials are growing quickest with a 6.96% CAGR through 2031.
Why is Asia-Pacific the fastest-growing region for pediatric studies?
Streamlined regulatory reviews in South Korea, Taiwan, and Australia, combined with large patient pools and cost advantages, drive a 7.18% CAGR for Asia-Pacific to 2031.
What primary factors are boosting global pediatric trial volumes?
Mandates such as the US PREA, EU Paediatric Regulation, and RACE for Children Act, plus rising chronic disease prevalence and growth in decentralized trial models, are key growth catalysts.
What recruitment challenges commonly slow pediatric trials?
Small eligible patient pools, complex dual consent-assent requirements, language barriers, and post-pandemic site staffing shortages together constrain enrollment rates to about 10% of eligible children.
Page last updated on:










