Ovarian Cancer Diagnostics & Therapeutics Market Size
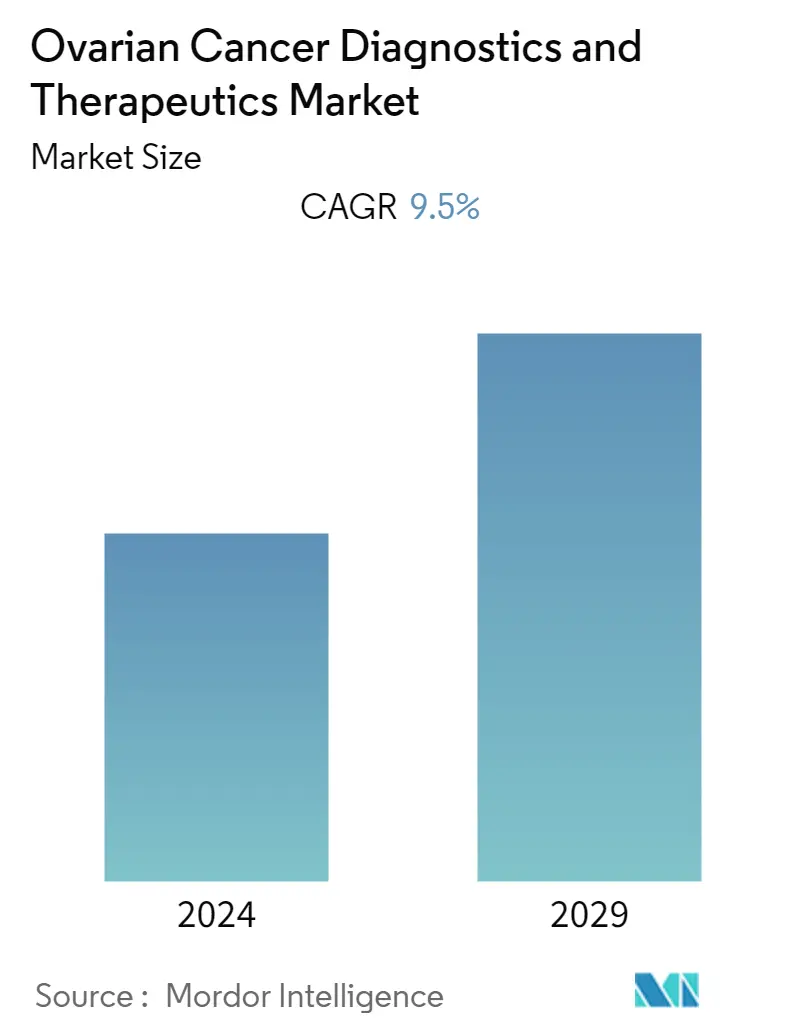
| Study Period | 2021 - 2029 |
| Base Year For Estimation | 2023 |
| CAGR | 9.50 % |
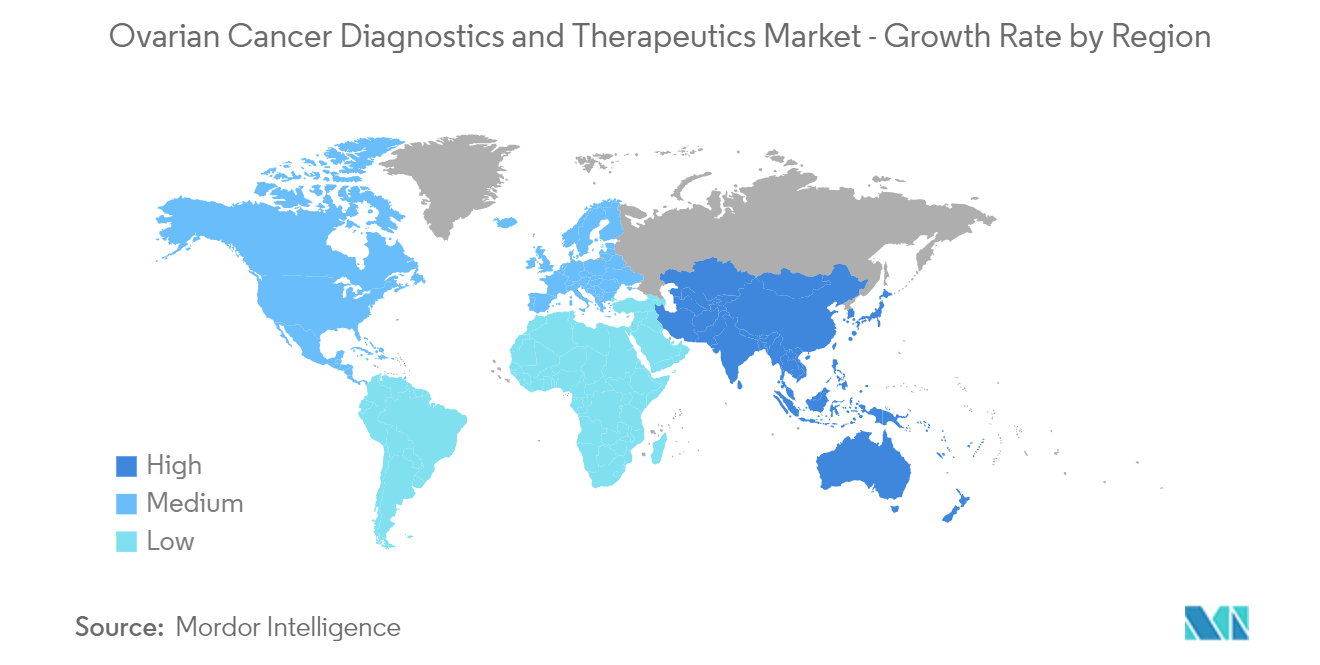
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
| Market Concentration | Medium |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Ovarian Cancer Diagnostics & Therapeutics Market Analysis
The ovarian cancer diagnostics and therapeutics market is projected to register a CAGR of 9.5% during the forecast period.
The COVID-19 pandemic affected healthcare systems globally and resulted in the interruption of usual care in many healthcare facilities, exposing vulnerable patients with cancer to significant risks. A study from ESMO Journal published in May 2021 conducted a survey in which a total of 356 centers from 54 countries across six continents participated. These centers serve 716,979 new patients with cancer every year. Most of them (88.2%) reported facing challenges in delivering care during the pandemic. Although 55.34% reduced services as part of a preemptive strategy, other common reasons included an overwhelmed system (19.94%), lack of personal protective equipment (19.10%), staff shortage (17.98%), and restricted access to medications (9.83%). Hence, the data suggest that the pandemic impacted cancer care across the world severely. However, currently, the market has reached its pre-pandemic nature in terms of demand for diagnostics and therapeutics, and it is expected to witness strong growth in the coming years.
Factors such as increasing ovarian cancer cases across the world and the use of combination therapies for the treatment of ovarian cancer are expected to drive the growth of the market.
The data published by the American Cancer Society in January 2022 mentioned that ovarian cancer was the seventh most common cancer among women globally, and 4.4% of entire cancer-related mortality among women is attributed to ovarian cancer. Thus, the increasing number of cases of ovarian cancer among the population is driving the growth of the ovarian cancer diagnostics and therapeutics market.
Similarly, in March 2022, the American Cancer Society reported that the five-year survival rate for ovarian cancer with early-stage 1 detection is around 93% and that most new cases of ovarian cancer are diagnosed at stage 3 or later, leaving little time and scope for proper treatment. However, with the increasing burden of cancer globally, people in developed countries are becoming more concerned about the disease, which is increasing the diagnosis volume and contributing to market growth. Additionally, the innovative launches by the market players are expected to drive the growth of the market. For instance, in May 2022, Inex Innovate launched the Ovarian Cancer detection test, OvaCis Rapid test, in the Southeast Asian markets.
Thus, the above-mentioned factors are expected to drive the growth of the market during the forecast period. However, the lack of awareness in emerging and low-income countries is expected to limit growth.
Ovarian Cancer Diagnostics & Therapeutics Market Trends
This section covers the major market trends shaping the Ovarian Cancer Diagnostics & Therapeutics Market according to our research experts:
CT Scan Segment is Expected to Show Better Growth Over the Forecast Period
Computed Tomography (CT) is an imaging procedure that uses special X-ray equipment to create detailed pictures, or scans, of areas inside the body. It is also called computerized tomography and computerized axial tomography (CAT). This equipment helps to see larger tumors and if the tumor is growing into nearby structures. It also helps to find enlarged lymph nodes, signs of cancer spreading to the liver or other organs, or signs that an ovarian tumor is affecting the kidneys or bladder. With such a high significance of CT scans, there is a growing demand for CT procedures to diagnose ovarian cancer, which is augmenting segment growth.
The increased sensitivity offered by this equipment is one of the major factors driving the growth of this segment. For instance, the data from PubMed published in September 2021 mentioned that the overall sensitivity of CT scan in detecting ovarian cancer is around 60.1%, whereas specificity is 93.7%. Hence, with high specificity and improved technology in CT scans, the segment is expected to witness strong growth in the coming years.
Additionally, the approval from regulatory authorities also boosts the growth of the market. For instance, in September 2021, the United States Food and Drug Administration (USFDA) granted clearance for Siemens NAEOTOM Alpha to Siemens Medical Solutions Inc. It is a technologically advanced computed tomography device designed to transform the information from X-ray photons that pass through a patient's body and are received by a detector into a detailed 3-dimensional image.
Thus, the above-mentioned factors are expected to drive the growth of the studied segment during the forecast period.
North America is Expected to Retain Large Market Share Over the Forecast Period
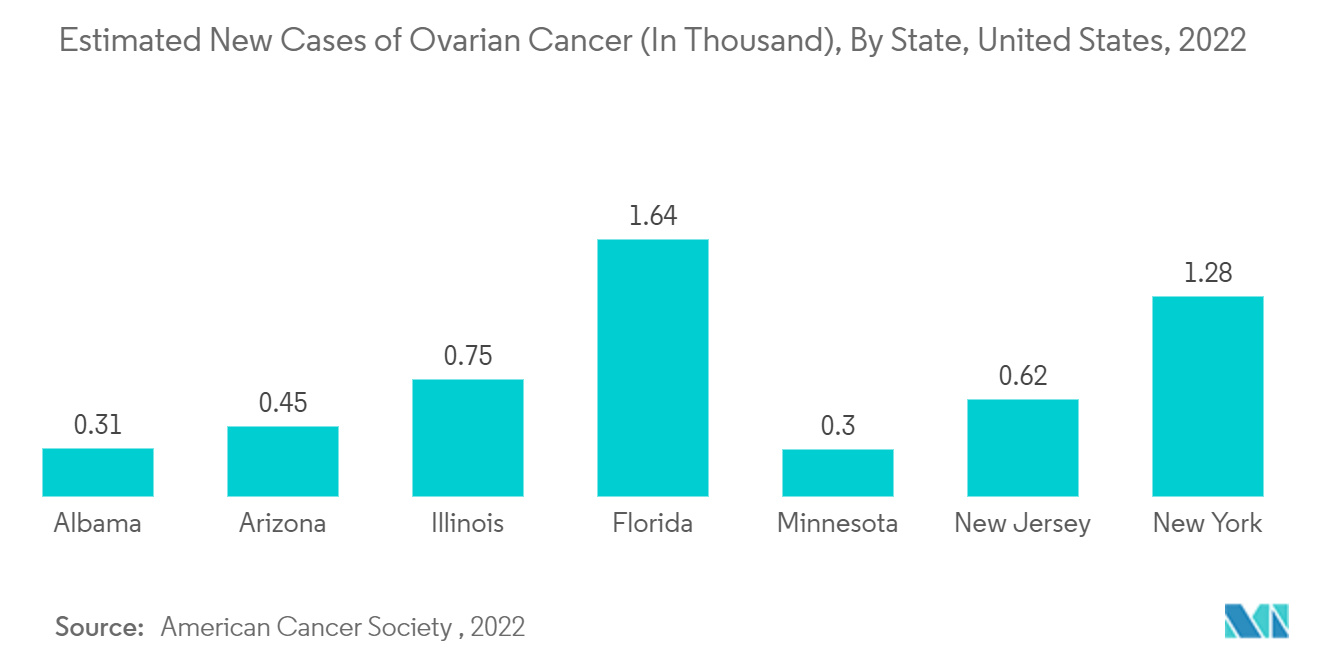
The North American region is believed to witness a strong share in the market studied. This is majorly attributed to the increasing cases of ovarian cancer in the region and the presence of key players catering to fulfill the growing demand for diagnostics and therapeutics for the same. In the North American region, the United States holds the highest share in the ovarian cancer diagnostics and therapeutics market and has contributed majorly to the same.
According to the American Cancer Society (ACS) report in January 2022, ovarian cancer was the ninth most common cancer and the fifth leading cause of cancer death among women in the United States. Furthermore, the ACS estimated that the number of individuals with ovarian cancer in the United States in 2022 will reach about 19,880, whereas a woman's risk of getting ovarian cancer during her lifetime is about 1 in 78. Additionally, most of the top global ovarian cancer diagnostics players are headquartered in the United States, and this tends to be the major advantage for the country, which is expected to drive the market.
Moreover, the increasing approvals for various advanced diagnostics and therapeutics are expected to drive the growth of the market in the forecast period. For instance, in November 2021, the USFDA approved an imaging drug known as Cytalux (pafolcianine), which is attracted to ovarian cancer tissue and illuminates it when exposed to fluorescent light, allowing surgeons to more easily and more precisely remove cancer.
Thus, the above-mentioned factors are expected to drive the growth of the studied segment during the forecast period.
Ovarian Cancer Diagnostics & Therapeutics Industry Overview
The ovarian cancer diagnostics and therapeutics market is highly competitive and consists of several major players. In terms of market share, few of the major players currently dominate the market. Key players in the market include AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb Company, Eli Lilly and Company, F. Hoffman-La Roche Ltd, GlaxoSmithKline PLC, Johnson and Johnson, Amneal Pharmaceuticals, and Siemens Healthineers AG.
Ovarian Cancer Diagnostics & Therapeutics Market Leaders
Astrazeneca plc
Glaxosmithkline Plc
Johnson and Johnson (Janssen Pharmaceuticals)
F. Hoffmann-La Roche Ltd
Siemens Healthineers AG
*Disclaimer: Major Players sorted in no particular order
Ovarian Cancer Diagnostics & Therapeutics Market News
- May 2022: HLB reported that its subsidiary Elevar Therapeutics started marketing Apealea, an ovarian cancer treatment, in Germany after listing the drug on Gelbe Liste, a German drug distribution website.
- May 2022: ImmunoGen received acceptance from the US Food and Drug Administration. The organization filed the Biologics License Application for mirvetuximab soravtansine monotherapy in patients with folate receptor alpha high platinum-resistant ovarian cancer who have been previously treated with 1-3 prior systemic treatments.
Ovarian Cancer Diagnostics & Therapeutics Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions and Market Definition
1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 Increasing Burden of Ovarian Cancer
4.2.2 Use of Combination Therapies for the Treatment of Ovarian Cancer
4.3 Market Restraints
4.3.1 Lack of Accurate Diagnosis of Ovarian Cancer
4.3.2 Lack of Awareness in Emerging and Low Income Economies
4.4 Porter's Five Forces Analysis
4.4.1 Threat of New Entrants
4.4.2 Bargaining Power of Buyers/Consumers
4.4.3 Bargaining Power of Suppliers
4.4.4 Threat of Substitute Products
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
5.1 By Cancer Type
5.1.1 Epithelial Ovarian Tumors
5.1.2 Ovarian Germ Cell Tumors
5.1.3 Other Cancer Types
5.2 By Modality
5.2.1 Diagnosis
5.2.1.1 Biopsy
5.2.1.2 Blood Tests
5.2.1.3 Ultrasound
5.2.1.4 PET
5.2.1.5 CT Scan
5.2.1.6 Other Diagnosis
5.2.2 Therapeutics
5.2.2.1 Chemotherapy
5.2.2.2 Radiation Therapy
5.2.2.3 Immunotherapy
5.2.2.4 Hormonal Therapy
5.2.2.5 Other Therapeutics
5.3 Geography
5.3.1 North America
5.3.1.1 United States
5.3.1.2 Canada
5.3.1.3 Mexico
5.3.2 Europe
5.3.2.1 Germany
5.3.2.2 United Kingdom
5.3.2.3 France
5.3.2.4 Italy
5.3.2.5 Spain
5.3.2.6 Rest of Europe
5.3.3 Asia-Pacific
5.3.3.1 China
5.3.3.2 Japan
5.3.3.3 India
5.3.3.4 Australia
5.3.3.5 South Korea
5.3.3.6 Rest of Asia-Pacific
5.3.4 Middle-East and Africa
5.3.4.1 GCC
5.3.4.2 South Africa
5.3.4.3 Rest of Middle-East and Africa
5.3.5 South America
5.3.5.1 Brazil
5.3.5.2 Argentina
5.3.5.3 Rest of South America
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 Amneal Pharmaceuticals LLC
6.1.2 Astrazeneca PLC
6.1.3 Boehringer Ingelheim International GmbH
6.1.4 Bristol Myers Squibb Company
6.1.5 Eli Lilly and Company
6.1.6 F. Hoffman-La Roche Ltd
6.1.7 Glaxosmithkline PLC
6.1.8 Johnson and Johnson (Janssen Pharmaceuticals)
6.1.9 Pfizer Inc.
6.1.10 Siemens Healthineers AG
6.1.11 Clovis Oncology
6.1.12 Abbvie (Allergan PLC)
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Ovarian Cancer Diagnostics & Therapeutics Industry Segmentation
As per the scope of the report, ovarian cancer is a type of cancer that begins in the ovaries. There are several types of ovarian cancer. Epithelial ovarian cancer is the most common type of ovarian cancer. Ovarian cancer is caused by uncontrollable cell growth in the ovarian tissue. It is a growing concern and one of the most common causes of death in women across the world.
The Ovarian Cancer Diagnostics and Therapeutics Market is Segmented by Cancer Type (Epithelial Ovarian Tumors, Ovarian Germ Cell Tumors, and Other Cancer Types), Modality (Diagnosis (Biopsy, Blood Tests, Ultrasound, PET, CT Scan, and Other Diagnosis) and Therapeutics (Chemotherapy, Radiation Therapy, Immunotherapy, Hormonal Therapy, and Other Therapeutics), and Geography (North America, Europe, Asia-Pacific, Middle-East and Africa, and South America). The market report also covers the estimated market sizes and trends of 17 countries across major regions globally. The report offers the value (in USD Million) for the above segments.
| By Cancer Type | |
| Epithelial Ovarian Tumors | |
| Ovarian Germ Cell Tumors | |
| Other Cancer Types |
| By Modality | ||||||||
| ||||||||
|
| Geography | ||||||||
| ||||||||
| ||||||||
| ||||||||
| ||||||||
|
Ovarian Cancer Diagnostics & Therapeutics Market Research FAQs
What is the current Ovarian Cancer Diagnostics and Therapeutics Market size?
The Ovarian Cancer Diagnostics and Therapeutics Market is projected to register a CAGR of 9.5% during the forecast period (2024-2029)
Who are the key players in Ovarian Cancer Diagnostics and Therapeutics Market?
Astrazeneca plc, Glaxosmithkline Plc, Johnson and Johnson (Janssen Pharmaceuticals), F. Hoffmann-La Roche Ltd and Siemens Healthineers AG are the major companies operating in the Ovarian Cancer Diagnostics and Therapeutics Market.
Which is the fastest growing region in Ovarian Cancer Diagnostics and Therapeutics Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Ovarian Cancer Diagnostics and Therapeutics Market?
In 2024, the North America accounts for the largest market share in Ovarian Cancer Diagnostics and Therapeutics Market.
What years does this Ovarian Cancer Diagnostics and Therapeutics Market cover?
The report covers the Ovarian Cancer Diagnostics and Therapeutics Market historical market size for years: 2021, 2022 and 2023. The report also forecasts the Ovarian Cancer Diagnostics and Therapeutics Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Ovarian Cancer Diagnostics and Therapeutics Industry Report
Statistics for the 2024 Ovarian Cancer Diagnostics and Therapeutics market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Ovarian Cancer Diagnostics and Therapeutics analysis includes a market forecast outlook to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.
