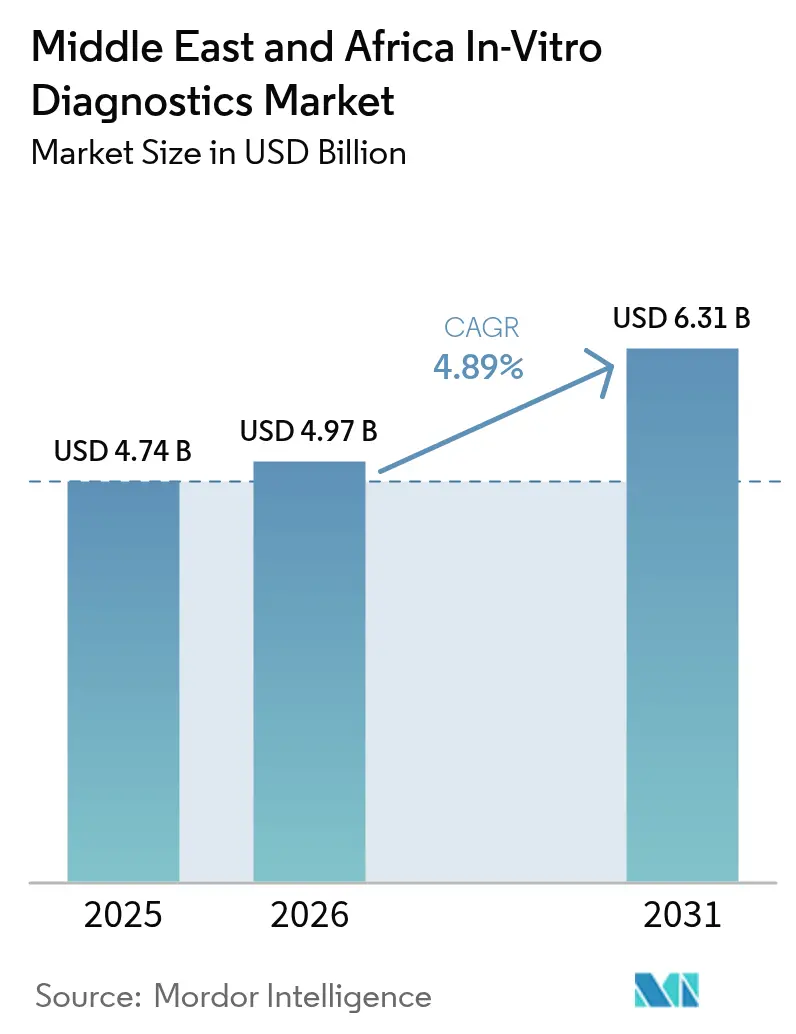
Middle East And Africa In-Vitro Diagnostics Market Analysis by Mordor Intelligence
The Middle East and Africa In-Vitro Diagnostics Market size was valued at USD 4.74 billion in 2025 and estimated to grow from USD 4.97 billion in 2026 to reach USD 6.31 billion by 2031, at a CAGR of 4.89% during the forecast period (2026-2031).
The COVID-19 pandemic had a significant impact on the in-vitro diagnostics market since there was an increasing demand for IVD kits and reagents for the rapid and accurate diagnosis of SARS-CoV-2 virus infection among the regional population. As per the 2022 report from the United Nations, with the help of several initiatives, including the establishment of the Regional Centre of Excellence for Genomic Surveillance and Bioinformatics in South Africa in collaboration with the SANBI at the University of Western Cape, the WHO increased the number of laboratories able to detect COVID-19 from two to more than 900. WHO also supported genetic sequencing efforts in Africa. In comparison to a year ago, these efforts resulted in more than 7,500 samples in the southern African region, and the amount of sequencing data on the continent increased by more than 54%. The efforts implemented by the government and related resources stimulated the market growth for in-vitro diagnostics in the studied region amid the pandemic. However, as per the analysis, the demand for IVD products may observe a slight decline post pandemic phase owing to a reduction in the number of infected patients and a surge in the vaccination rate across the region.
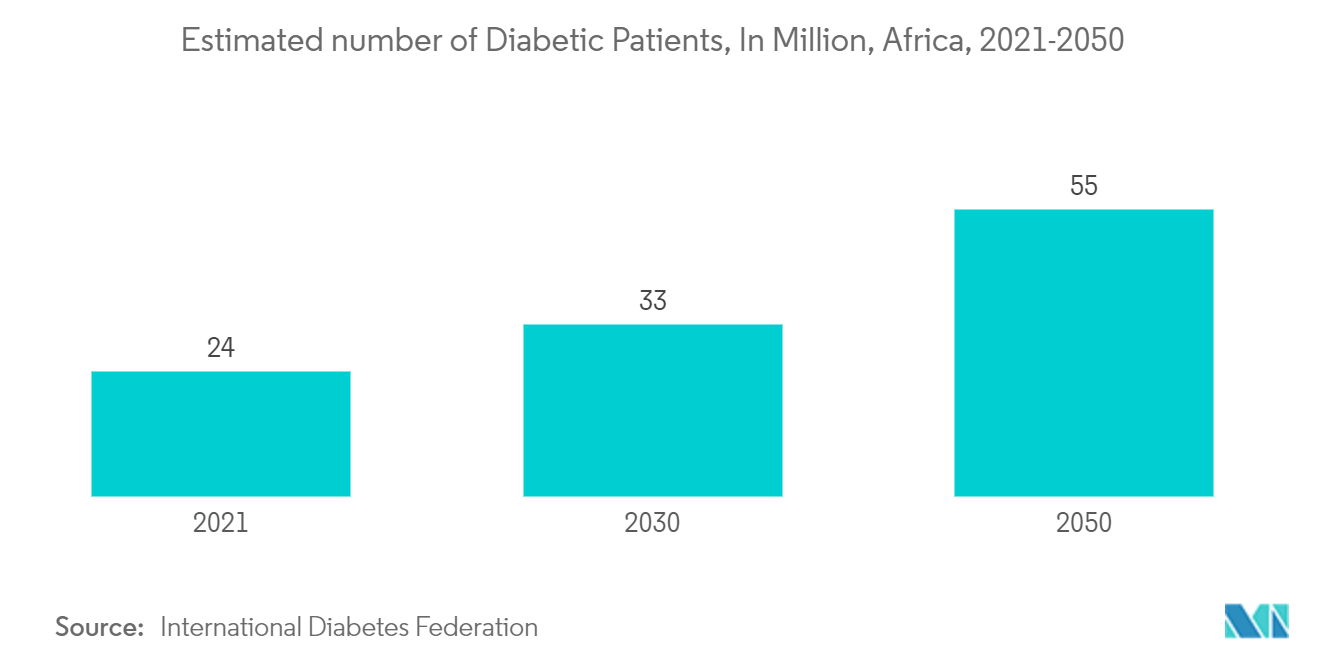
The major driving factor linked to the growth of the IVD industry in the region is the high burden of chronic diseases, especially infectious diseases, diabetes, and cancers. The Middle East and Africa region is majorly affected due to the burden of chronic diseases. As per the 2021 report from the International Diabetes Federation (IDF), 24 million adults (20-79) are living with diabetes in the IDF Africa Region. This number is estimated to increase to 33 million by 2030 and 55 million by 2045. As per the same source, 73 million adults (20-79) were living with diabetes in the IDF Middle East and North Africa region in 2021. This number is estimated to increase to 95 million by 2030 and 136 million by 2045. The prime factor responsible for this rising burden is the lack of a proper healthcare system across African countries and low awareness about novel disease diagnosis techniques. As per the 2022 review report from UNAIDS, law reform in Angola enabled the country to make use of funds from the Global Fund to combat AIDS, tuberculosis, and malaria to reach young members of critical populations by making it easier for more young people to obtain HIV testing services.
Thus, due to this rise in chronic diseases, the Middle East & Africa In-Vitro Diagnostics market is expected to witness growth over the analysis period. However, stringent regulations associated with the products may restrain the market's growth.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Middle East And Africa In-Vitro Diagnostics Market Trends and Insights
Diabetes Segment is Expected to Register High CAGR in the Application Segment
During the forecast period, the diabetes segment is expected to witness healthy growth. Factors such as the growing burden of diabetes, the increasing diagnosis of diabetes, and rising technological advancements in the field of diabetes are the main growth drivers of this segment.
For instance, as per the 2021 report from the African Union Development Agency (AUDA-NEPAD), the African High-Level Panel on Innovation and Emerging Technology (APET) was urging African nations to implement medical management technologies to enhance diabetic healthcare and lessen the burden of the condition. The strain of frequent hospital visits was lessened by the availability of more convenient and accessible healthcare solutions, which were supported by medical management technologies. Furthermore, according to the IDF Diabetes Atlas, 10th edition, published in 2021, an estimated 24 million adults aged 20-79 years were living with diabetes in the IDF Africa region, representing a regional prevalence of 4.5%. Moreover, 54% of people living with diabetes in the region are undiagnosed, the highest proportion of all IDF regions.
The IDF estimates that the increase in diabetes will be 33 million by 2030 and 55 million by 2045. This raises massive healthcare concerns, as all African countries are already struggling to cope with the diabetes burden. However, the WHO reports that diabetes in the region is a serious, chronic, and costly disease that is estimated to rise to 23.9 million cases by 2030.
Hence, the rising prevalence of the disease is expected to drive the growth of the diabetes segment over the forecast period.
GCC Region is Expected to Witness Growth Over The Forecast Period
The GCC in-vitro diagnostics market is expected to grow over the analysis period owing to the growing burden of chronic diseases and increasing healthcare expenditure, which is actively affecting the growth of the market.
The Kingdom of Saudi Arabia made a USD 150 million donations to Gavi, the Vaccine Alliance, in April 2020 to help in the organization's response to the COVID-19 pandemic. The funding was part of a USD 500 million package from the Kingdom of Saudi Arabia to support response to emergencies and preparedness efforts, the deployment of innovative diagnostics, treatments, and vaccinations, and the maintenance of adequate supplies of protective equipment for medical personnel. The government's actions during the pandemic were anticipated to boost the market growth.
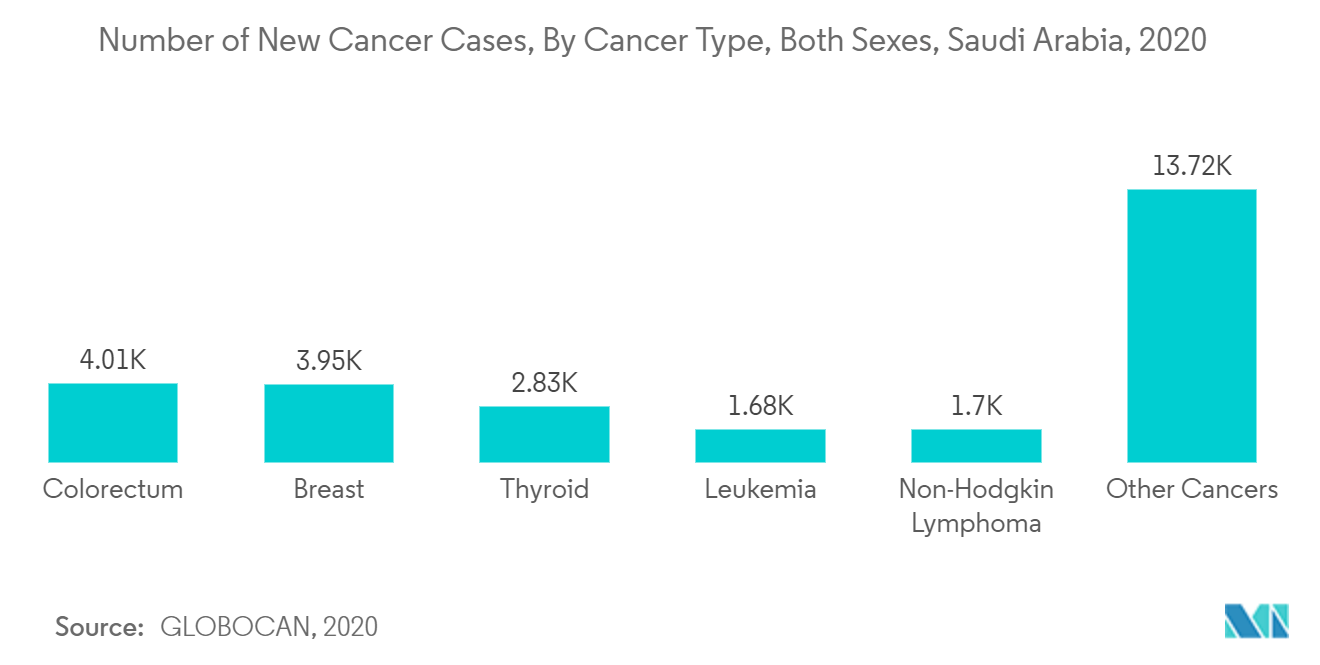
Additionally, the COVID-19 pandemic in the GCC region helped the in-vitro diagnostics market grow. For instance, according to a study published in the Cardiovascular Diabetology Journal in December 2020, diabetes mellitus (DM) was the most common comorbidity among COVID-19-positive patients, with prevalence rates of 68.3%, hypertension of 42.6%, and obesity of 42.2%. Patients with COVID-19 who were hospitalized at King Khaled University Hospital (KKUH), Riyadh, Saudi Arabia, had a high prevalence of DM. The demand for in-vitro diagnostics among the patient population was increased by the high prevalence of COVID-19 patients and diabetes mellitus patients.
Moreover, in July 2022, Saudi Arabia's Arabian International Healthcare Holding Company, TIBBIYAH, commenced a new joint venture (JV) with the Swiss company Unilabs Diagnostics AB. Likewise, in April 2022, the United Arab Emirates Sheikh Shakhbout Medical City (SSMC) became one of the first hospitals in the Middle East to launch a prominent point-of-care tool for antimicrobial management.
Therefore, owing to the aforementioned factors, it is anticipated that the studied market will witness growth over the analysis period.
Competitive Landscape
The Middle East and Africa In-Vitro Diagnostics market is competitive and consists of several major players. In terms of market share, a few of the major players currently dominate the market in the GCC. Some of the major players in the market are Abbott Laboratories, BioMerieux SA, Sysmex Corporation, F. Hoffmann-La Roche AG, and Becton, Dickinson, and Company, among others. The companies mostly monitor distribution via their branch offices or distributors operating at the domestic and international levels.
Middle East And Africa In-Vitro Diagnostics Industry Leaders
Abbott Laboratories
F. Hoffmann-La Roche AG
Becton, Dickinson and Company
Sysmex Corporation
BioMerieux SA
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- March 2022: Audere entered into a partnership with Medical Diagnostech, headquartered in South Africa, a developer, and manufacturer of lateral flow rapid diagnostic test kits. This partnership will pair Medical Diagnostech's MD SARS-CoV-2 antigen device with Audere's HealthPulse digital companion app.
- February 2022: Trinity Biotech plc received approval for its new HIV screening product, TrinScreen HIV, from the World Health Organization (WHO). TrinScreen HIV is a quick test that yields findings from a fingerstick blood sample in less than 12 minutes. The test has shown good performance in both an independent evaluation sponsored by the WHO and a multi-center clinical trial that the company conducted in Africa in 2020.
Middle East And Africa In-Vitro Diagnostics Market Report Scope
As per the scope of the report, in-vitro diagnostics is defined as a medical device used alone or in combination, intended by the manufacturer for the in vitro examination of specimens derived from the human body solely or principally to provide information for diagnostic, monitoring, or compatibility purposes. The Middle East & Africa In-Vitro Diagnostics Market is segmented by Technique (Histochemistry, Molecular Diagnostics, Hematology, Self-blood Glucose Testing, Immunochemistry, and Other Techniques), Product (Instrument, Reagent, and Other Products), Usability (Disposable IVD Device, Reusable IVD Device), Application (Infectious Disease, Diabetes, Cancer/Oncology, Cardiology, Autoimmune Disease, and Other Applications) End User (Diagnostic Laboratories, Hospitals and Clinics, and Other End Users) Diagnostic Approach (Point-of-Care Diagnostics and Centralized Laboratory-based Diagnostics) and Geography (GCC, South Africa, Rest of Middle East & Africa). The report offers the value (in USD million) for the above segments.
| Histochemistry |
| Molecular Diagnostics |
| Hematology |
| Self-blood Glucose Testing |
| Immunochemistry |
| Other Techniques |
| Instrument |
| Reagent |
| Other Products |
| Disposable IVD Device |
| Reusable IVD Device |
| Infectious Disease |
| Diabetes |
| Cancer/Oncology |
| Cardiology |
| Autoimmune Disease |
| Other Applications |
| Diagnostic Laboratories |
| Hospitals and Clinics |
| Other End Users (Academic and Medical School) |
| Point-of-Care Diagnostics |
| Centralized Laboratory-based Diagnostics |
| GCC |
| South Africa |
| Rest of Middle East & Africa |
| By Technique | Histochemistry |
| Molecular Diagnostics | |
| Hematology | |
| Self-blood Glucose Testing | |
| Immunochemistry | |
| Other Techniques | |
| By Product | Instrument |
| Reagent | |
| Other Products | |
| By Usability | Disposable IVD Device |
| Reusable IVD Device | |
| By Application | Infectious Disease |
| Diabetes | |
| Cancer/Oncology | |
| Cardiology | |
| Autoimmune Disease | |
| Other Applications | |
| By End User | Diagnostic Laboratories |
| Hospitals and Clinics | |
| Other End Users (Academic and Medical School) | |
| By Diagnostic Approach | Point-of-Care Diagnostics |
| Centralized Laboratory-based Diagnostics | |
| Geography | GCC |
| South Africa | |
| Rest of Middle East & Africa |


Key Questions Answered in the Report
How big is the Middle East and Africa In-Vitro Diagnostics Market?
The Middle East and Africa In-Vitro Diagnostics Market size is expected to reach USD 4.97 billion in 2026 and grow at a CAGR of 4.89% to reach USD 6.31 billion by 2031.
What is the current Middle East and Africa In-Vitro Diagnostics Market size?
In 2026, the Middle East and Africa In-Vitro Diagnostics Market size is expected to reach USD 4.97 billion.
Who are the key players in Middle East and Africa In-Vitro Diagnostics Market?
Abbott Laboratories, F. Hoffmann-La Roche AG, Becton, Dickinson and Company, Sysmex Corporation and BioMerieux SA are the major companies operating in the Middle East and Africa In-Vitro Diagnostics Market.
What years does this Middle East and Africa In-Vitro Diagnostics Market cover, and what was the market size in 2025?
In 2025, the Middle East and Africa In-Vitro Diagnostics Market size was estimated at USD 4.97 billion. The report covers the Middle East and Africa In-Vitro Diagnostics Market historical market size for years: 2021, 2022, 2023 and 2024. The report also forecasts the Middle East and Africa In-Vitro Diagnostics Market size for years: 2026, 2027, 2028, 2029, 2030 and 2031.
Page last updated on:








