Italy Pharmaceutical Market Analysis by Mordor Intelligence
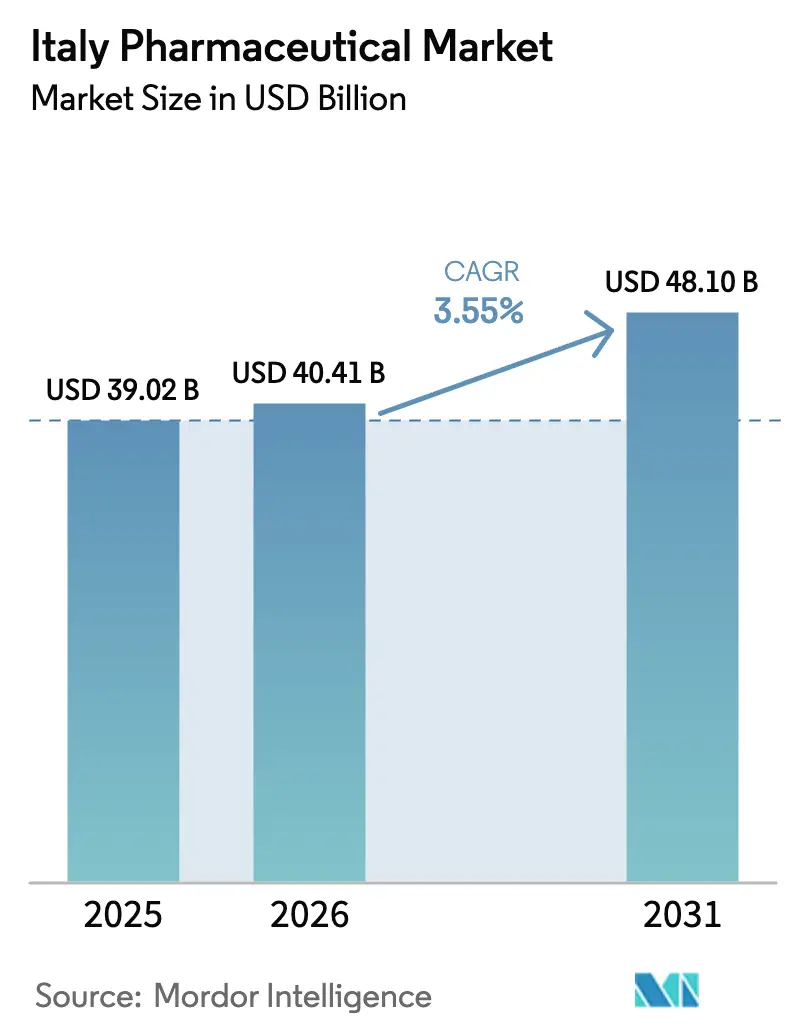
The Italy pharmaceutical market size is projected to expand from USD 40.41 billion in 2026 to USD 48.10 billion by 2031, registering a 3.55% CAGR between 2026 and 2031. A gradual shift toward high-value biologics, accelerated biosimilar substitution and regionally funded manufacturing upgrades under the National Recovery and Resilience Plan (PNRR) underpin this trajectory. While generic penetration remains stalled at a significant volume share, Italy’s position as Europe’s second-largest medicines producer supports export-oriented growth even as domestic margins tighten. Digital prescription infrastructure, broader pharmacy-led diagnostic services and rising self-care among older citizens further expand addressable demand. Capital commitments exceeding EUR 5.5 billion since 2024 from both domestic champions and multinationals underscore confidence in the long-term vitality of the Italy pharmaceutical market.
Key Report Takeaways
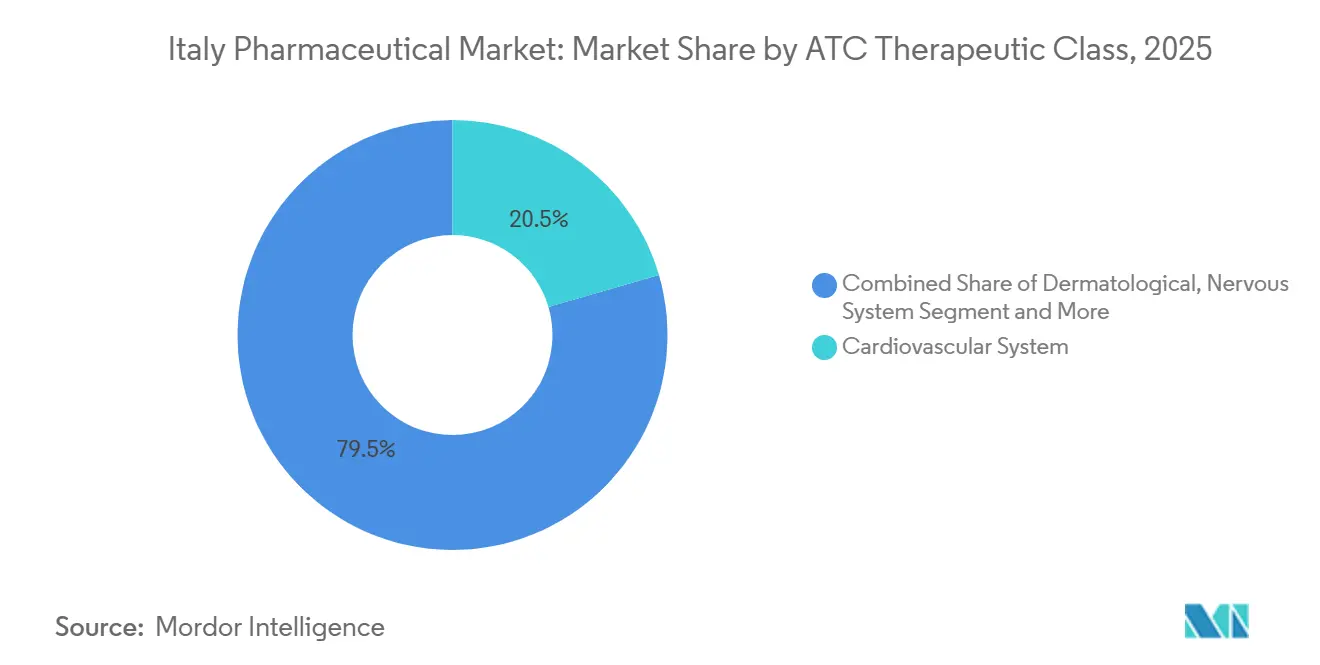
- By therapeutic class, cardiovascular therapies led with 20.54% of the Italy pharmaceutical market share in 2025, while blood and hematopoietic agents are forecast to advance at an 8.25% CAGR through 2031.
- By drug type, generics captured 55.54% of 2025 revenue, whereas biosimilars are projected to grow at a 6.65% CAGR during 2026-2031.
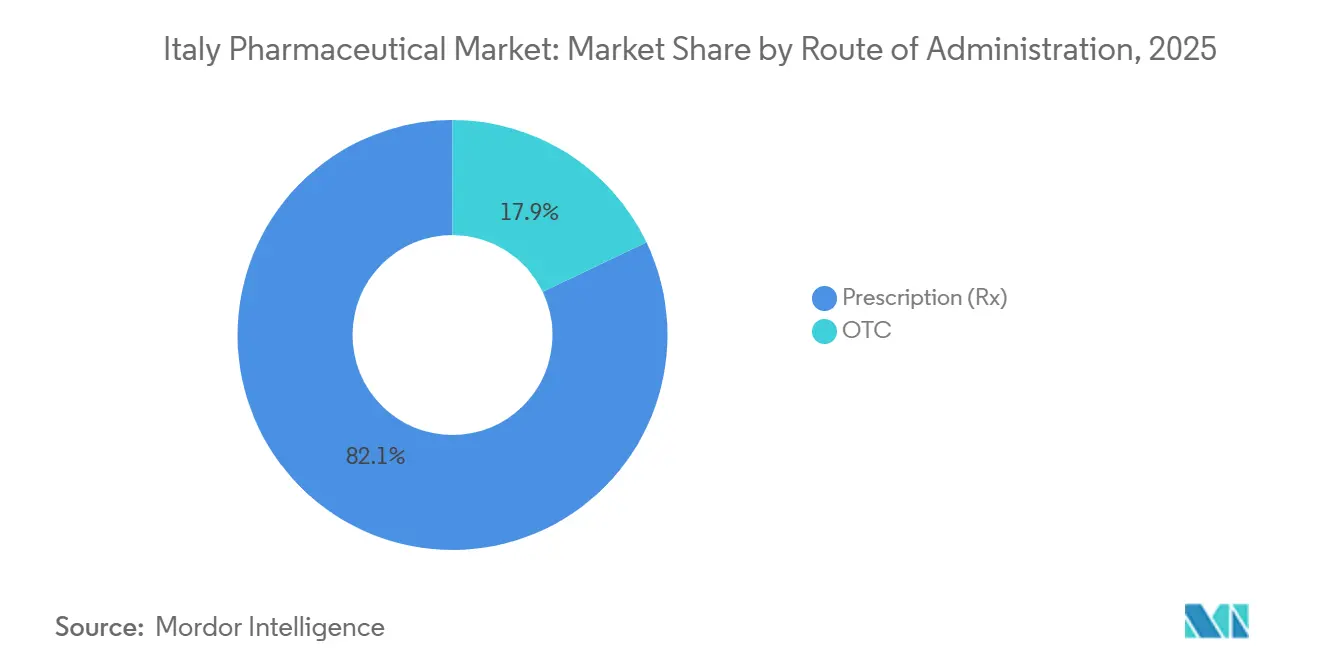
- By prescription type, prescription medicines accounted for 82.15% of 2025 sales, yet over-the-counter products are expanding at a 6.82% CAGR on the back of pharmacist-led chronic-disease screening.
- By route of administration, oral formulations generated 58.23% of 2025 revenue, but inhalation therapies will accelerate at a 6.42% CAGR owing to smart-device mandates.
- By distribution channel, hospital pharmacies dispensed 38.23% of 2025 volume, while online channels are on course for a 6.52% CAGR after Schedule C e-commerce liberalization.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Italy Pharmaceutical Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Aging population & chronic disease burden | +1.2% | National, pressure in Liguria and Friuli-Venezia Giulia | Long term (≥ 4 years) |
| Government R&D tax incentives & regional grants | +0.6% | Southern regions prioritized under PNRR | Medium term (2-4 years) |
| Biosimilar uptake post-patent cliffs | +0.9% | National, led by Lombardy and Veneto | Short term (≤ 2 years) |
| Digital health & e-prescriptions | +0.5% | National rollout, advanced in Emilia-Romagna and Tuscany | Medium term (2-4 years) |
| Surge in orphan-drug demand | +0.7% | University hospital networks nationwide | Long term (≥ 4 years) |
| PNRR funds catalyzing life-sciences reshoring | +0.8% | Manufacturing hubs in Lazio, Lombardy, Emilia-Romagna | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Aging Population & Chronic Disease Burden
Italy’s median age climbed to 48.4 years in 2025, the oldest in the EU, and citizens over 65 consume 4.2 times more medicines than working-age adults. Chronic illnesses affect 39.8% of adults, while polypharmacy among the over-75 cohort reached 28%, contributing to an 11% increase in adverse-event reports. Regions where seniors exceed 30% of residents, such as Liguria, post per-capita pharmaceutical outlays 22% above the national mean. The 2024 Chronic Care Plan now links reimbursement for cardiovascular and metabolic drugs to measurable hospital-avoidance outcomes, nudging suppliers to generate real-world evidence. Collectively, these demographics guarantee sustained demand across cardiometabolic and respiratory portfolios, bolstering the Italy pharmaceutical market even under tighter unit prices.
Government R&D Tax Incentives & Regional Grants
The PNRR earmarked EUR 1.67 billion for plant modernization and EUR 15.63 billion for broader health infrastructure, with preference for facilities located in Campania, Calabria and Sicily. A 5% tax credit on phase III trials in underserved provinces became effective in January 2025, already reversing a multi-year decline in oncology studies. Mid-cap companies capitalized on grants covering 35% of capital expenditure for sterile fill-finish lines, shortening payback periods to under five years. These fiscal carrots modestly lift forecast growth yet deliver oversized benefits to regions historically sidelined in drug development, gradually rebalancing the Italy pharmaceutical industry’s research footprint.
Biosimilar Uptake Post-Patent Cliffs
Italy achieved a 67% average biosimilar substitution rate in 2024, ahead of Germany and France, after regional health authorities mandated first-line biosimilar prescribing[1]Agenzia Italiana del Farmaco, “Rapporto Attività 2025,” aifa.gov.it. Patent expiries for adalimumab and ranibizumab should unlock EUR 800 million in annual National Health Service savings by 2027, freeing budgets for novel cell- and gene-therapies. Lombardy already records 86.8% uptake for rituximab and bevacizumab biosimilars, yet insulin molecules lag with just 12% penetration because clinicians hesitate to switch stable patients. Educational campaigns funded under the 2025 Budget Law aim to close this gap. Stronger biosimilar economics continue to support the Italy pharmaceutical market, though manufacturers face net-price compression averaging 35%.
Digital Health & E-Prescriptions Accelerating Access
Electronic Health Record coverage reached 89% of residents in 2025, enabling prescription portability across Italy and cutting median fulfillment time from 48 hours to six. Teleconsultations for diabetes and hypertension surged 67%, generating 1.8 million e-prescriptions that AIFA recognizes as equal to in-person scripts for reimbursement. Smart inhalers, compulsory for biologic asthma drugs since 2025, uploaded 4.2 million adherence events to the national database within 18 months, allowing payers to connect payment with real-world usage. Yet 21 distinct regional platforms hamper nationwide analytics, delaying the deployment of AI clinical-decision tools that could further streamline care.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High cost of innovative therapies | -0.8% | National, acute in Lazio and Campania | Medium term (2-4 years) |
| Stringent AIFA price & reimbursement | -1.1% | National, centralized through AIFA | Short term (≤ 2 years) |
| Margin pressure from generic penetration | -0.4% | National, strongest in primary care | Long term (≥ 4 years) |
| Carbon-footprint compliance costs | -0.3% | National, EU-wide alignment | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Stringent AIFA Price & Reimbursement Negotiations
AIFA’s payback clawed back EUR 1.24 billion from suppliers in 2024, 18% more than the prior year. Negotiations now average 18 months, delaying access to 14 EMA-cleared therapies, including new GLP-1 and PCSK9 agents. Managed-entry contracts cover 42% of innovative launches, yet just one-third possess robust data systems to confirm real-world outcomes, skewing financial exposure toward manufacturers. Additional 12% price cuts applied by Lombardy and Veneto hospital tenders multiply complexity. Because the 2025 Budget Law caps annual drug spending growth below projected market expansion, net realized prices could erode by 0.7 percentage points annually through 2031, dampening returns across the Italy pharmaceutical market.
High Cost of Innovative Therapies
CAR-T interventions priced between EUR 320,000 and EUR 400,000 generated waiting lists exceeding four months in 2025, pushing 23% of eligible Italians abroad for treatment. Orphan drugs consumed 11% of national drug outlays yet served only 2% of patients, reviving debate over AIFA’s EUR 50,000 cost-effectiveness ceiling. Specialty expenditure is projected to hit 58% of total pharmaceutical spending by 2030, up from 47% in 2025. Budget-strapped regions such as Lazio postponed non-urgent biologic purchases by up to 90 days, widening north–south care gaps and shaving incremental demand off the Italy pharmaceutical market size.
Segment Analysis
By ATC/Therapeutic Class: Oncology Biologics Reshape Blood Segment
Blood and hematopoietic agents are projected to generate the fastest 8.25% CAGR, positioning the subgroup as a central driver of the Italy pharmaceutical market size through 2031. CAR-T rollouts and biosimilar erythropoietins underpin this expansion, though limited infusion-center capacity currently constrains patient throughput. Cardiovascular drugs commanded 20.54% of 2025 revenue, but growth stagnates as statins and ACE inhibitors face 12% annual price erosion in regional tenders. Gastrointestinal agents anchored by GLP-1 agonists outperformed average prescription growth, reflecting rising obesity management.
Oncology biologics within the blood category increasingly dominate hospital budgets even as reimbursement hurdles persist. Biosimilar filgrastim and pegfilgrastim already captured near-total share, illustrating clinician comfort with hematology substitutes. Respiratory biologics for severe asthma gained traction after inhaler-based adherence tracking became compulsory, aiding formulary inclusion. Dermatology’s IL-17 and IL-23 inhibitors continued robust adoption among biologic-naïve psoriasis patients, reinforcing specialty-driven momentum for the Italy pharmaceutical market.

By Drug Type: Biosimilar Momentum Challenges Generic Plateau
Generics controlled 55.54% of 2025 sales but posted just 1.2% volume growth due to entrenched prescriber preferences for branded SKUs. Biosimilars, by contrast, are set to expand at a 6.65% CAGR, unlocking EUR 2.1 billion in fresh revenue as adalimumab, ranibizumab and denosumab copies push substitution rates toward 70%. The Italy pharmaceutical market share tied to branded specialty drugs remains resilient, with Novartis’s Entresto alone delivering EUR 340 million in national sales.
Teva and Viatris each launched seven additional biosimilars during 2024-2025, pricing 35-40% below originators and capturing quick hospital uptake. Regional decrees mandating biosimilar first-line use for naïve patients accelerated the shift, especially in Lombardy and Veneto. Intensifying biologics competition is pushing generic manufacturers out of low-margin commodities, consolidating supply and subtly reshaping the Italy pharmaceutical industry’s competitive contours.
By Prescription Type: OTC Self-Care Gains Regulatory Tailwinds
Prescription drugs retained an 82.15% revenue share in 2025, centered on chronic-disease therapies reimbursed by the National Health Service. Yet the OTC segment is projected to climb 6.82% annually thanks to Law 69/2024, which lets pharmacists perform point-of-care diagnostics and recommend suitable non-prescription treatments. Cost-sharing reforms shifted EUR 280 million in expenditures toward OTC analgesics, antihistamines and digestive aids last year.
Vitamin D, omega-3 and probiotics enjoyed rapid uptake among aging consumers, buoyed by nationwide preventive-care campaigns. The Italy pharmaceutical market size associated with self-medication is therefore poised to expand faster than physician-directed categories, although stringent antimicrobial stewardship has already trimmed discretionary antibiotic issuance.
By Route of Administration: Inhalation Devices Embed Digital Tracking
Oral drugs still account for 58.23% of 2025 turnover across cardiovascular, metabolic and OTC categories. However, inhalation products will post a 6.42% CAGR as January 2025 legislation obliges smart-inhaler coupling for all reimbursed asthma biologics[2]European Respiratory Society, “Smart Inhaler Adherence Data,” ersnet.org . Chiesi’s digital-biologic program advances this trend, feeding 4.2 million adherence datapoints into national records during the first half of 2026.
Parenteral formats dominate oncology and autoimmune care, with subcutaneous alternatives such as Roche’s rituximab shaving hospital chair time by 18%. Topical and transdermal platforms segment remains stable but benefits from IL-17 inhibitor popularity. Momentum in connected inhalation technology exemplifies how digital convergence reinforces specialty growth throughout the Italy pharmaceutical market.
By Distribution Channel: E-Commerce Disrupts Traditional Retail
Hospital pharmacies dispensed 38.23% of volume in 2025 as biologics and advanced injectables remain centrally procured. Decree 17/2024 opened Schedule C drugs to online sale, propelling web-based channels toward a 6.52% CAGR. Nearly 890,000 new consumers made a first digital purchase in 2025, lured by 24-hour delivery in Rome, Milan and Turin.
Independent brick-and-mortar outlets face squeezed margins from generic price wars and online cannibalization, leading to 340 closures in 2024. Pharmacy chains now embrace click-and-collect to integrate digital convenience with in-store counseling. Altogether, omnichannel evolution is redefining patient access models and bolstering competition within the Italy pharmaceutical market.
Geography Analysis
Italy’s northern triangle—Lombardy, Emilia-Romagna and Lazio—generated a conspicuous share of 2025 drug consumption and houses 71% of R&D and manufacturing infrastructure. The Italy pharmaceutical market size linked to these three regions outweighs that of the south, yet Campania, Sicily and Calabria spend 34% more per capita on branded cardiovascular products because biosimilar adoption trails national norms by up to 25 percentage points. The PNRR directs EUR 4.6 billion toward southern health infrastructure, including EUR 680 million for cold-chain upgrades expected to cut biologic spoilage to Lombardy-like levels by 2027.
Manufacturing capital continues to cluster in the center-north. Novo Nordisk’s semaglutide megasite in Lazio, Chiesi’s biotech hub in Emilia-Romagna and Novartis’s radiopharmaceutical plant in Lombardy collectively add 2,800 skilled jobs, reinforcing northern dominance. Tuscany meanwhile rose to a 14% share of national phase III oncology trials after offering 22% R&D tax credits. Conversely, Lazio’s EUR 1.1 billion deficit forced 90-day biologic purchase delays, highlighting how fiscal strain stifles access even where capacity exists.
Cross-border pharmaceutical tourism fell 18% in 2025 due to enhanced e-prescription portability across the EU, though 12,400 Italians still traveled for CAR-T therapy, indicating domestic infusion constraints. The national trade surplus in pharmaceuticals reached EUR 8.2 billion, buoyed by biologic exports and API shipments. New incentives for southern clinical trials intend to spread this upside geographically, but for now, the Italy pharmaceutical market remains regionally bifurcated between innovation-rich north and demand-heavy south.
Competitive Landscape
The top-10 suppliers controlled roughly two-thirds of 2025 sales, placing the Italy pharmaceutical market in a moderately concentrated posture[3]Farmindustria, “Industry Report 2024-2025,” farmindustria.it. Domestic champions Chiesi, Recordati and Alfasigma pumped EUR 2.1 billion into new biomanufacturing to counter multinationals’ reshoring waves. Novo Nordisk’s EUR 2.34 billion Lazio site and Johnson & Johnson’s EUR 580 million Latina upgrade exploit wage advantages and PNRR subsidies, intensifying competition along the biologics value chain.
White-space in orphan diseases remains underexploited despite 94 EMA designations secured by Italian sponsors in 2024. Recordati has already lifted peak-sales guidance for rare-disease assets by 18%, signaling strategic pivot toward neurometabolic niches. Teva and Viatris captured biosimilar volume with seven launches priced 40% below originators but exited several low-margin generic lines to protect profitability. Novartis’s spate of 14 radiopharmaceutical patent filings underscores competition shifting to precision modalities that circumvent generic substitution.
Sustainability rules now shape tender scores. Sanofi and Roche integrated end-to-end carbon metrics into 2025 bids to fulfill EU Directive 2024/825, absorbing higher overhead yet gaining evaluative credit. Smaller players lag in audit-ready emission tracking, risking disqualification and reinforcing scale advantages that could elevate future concentration in the Italy pharmaceutical industry.
Italy Pharmaceutical Industry Leaders
AbbVie Inc.
AstraZeneca plc
Bayer AG
GlaxoSmithKline plc
F. Hoffmann-La Roche AG
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- January 2026: Valo Therapeutics secured AIFA clearance for a phase I trial of PeptiCRAd-1 plus pembrolizumab in injectable solid tumors.
- July 2025: SOMAÍ launched EU-GMP cannabinoid medicines in Italy via a partnership with Materia Medica Processing.
Italy Pharmaceutical Market Report Scope
As per the scope of this report, pharmaceuticals are referred to as prescribed and non-prescription drugs. These medicines can be bought by an individual with or without the doctor's prescription and are safe for consumption for various illness with or without the doctor's consent.
The segmentation of the Italy pharmaceutical market is categorized by ATC/therapeutic class, drug type, prescription type, route of administration, and distribution channel. By ATC/therapeutic class, the market includes blood and hematopoietic organs, cardiovascular system, dermatological, gastrointestinal and metabolism, nervous system, respiratory system, and others. By drug type, it is segmented into branded, generic, and biosimilars. By prescription type, the market is divided into prescription drugs (Rx) and OTC drugs. By route of administration, the segmentation includes oral, parenteral, inhalation, topical, and others. By distribution channel, the market is segmented into hospital pharmacies, retail pharmacies, online pharmacies, and wholesalers/distributors. The report offers the value (in USD) for the above segments.
| Blood & Hematopoietic Organs |
| Cardiovascular System |
| Dermatological |
| Gastrointestinal & Metabolism |
| Nervous System |
| Respiratory System |
| Others |
| Branded |
| Generic |
| Biosimilars |
| Prescription Drugs (Rx) |
| OTC Drugs |
| Oral |
| Parenteral |
| Inhalation |
| Topical |
| Others |
| Hospital Pharmacies |
| Retail Pharmacies |
| Online Pharmacies |
| Wholesalers / Distributors |
| By ATC / Therapeutic Class | Blood & Hematopoietic Organs |
| Cardiovascular System | |
| Dermatological | |
| Gastrointestinal & Metabolism | |
| Nervous System | |
| Respiratory System | |
| Others | |
| By Drug Type | Branded |
| Generic | |
| Biosimilars | |
| By Prescription Type | Prescription Drugs (Rx) |
| OTC Drugs | |
| By Route of Administration | Oral |
| Parenteral | |
| Inhalation | |
| Topical | |
| Others | |
| By Distribution Channel | Hospital Pharmacies |
| Retail Pharmacies | |
| Online Pharmacies | |
| Wholesalers / Distributors |


Key Questions Answered in the Report
How fast is prescription-drug spending growing in Italy?
Prescription medicines are advancing more slowly than other categories, rising below the 3.55% market CAGR as cost caps and rebate mechanisms compress net prices.
Which segment is expanding the quickest?
Blood and hematopoietic agents are forecast to post an 8.25% CAGR through 2031 on the back of CAR-T and biosimilar erythropoietin uptake.
What drives Italy's strong biosimilar penetration?
Regional mandates requiring biosimilar first-line prescribing plus aggressive tender pricing boosted national substitution rates to 67% in 2024.
How will e-commerce change drug distribution?
Following Schedule C liberalization, online pharmacies are expected to grow at 6.52% annually, siphoning share from traditional retail outlets, especially in urban areas.
Why is regional disparity a concern?
Northern regions house most R&D and manufacturing capacity, while southern regions spend more per capita on branded drugs due to slower biosimilar adoption, widening access and budget gaps.
What is the impact of EU carbon rules on suppliers?
Directive 2024/825 adds roughly EUR 50 million in annual compliance costs but can improve tender scores for companies that document full supply-chain emissions.
Page last updated on:









