India Respiratory Devices Market Analysis by Mordor Intelligence
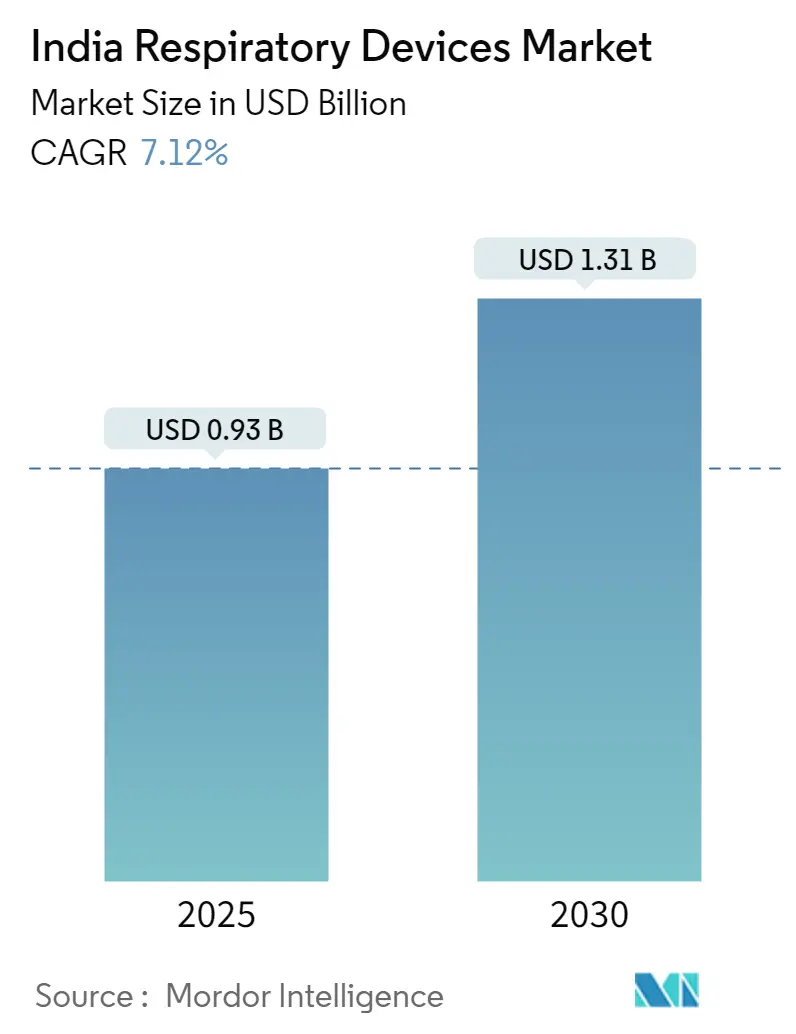
The India Respiratory Devices Market size is estimated at USD 0.93 billion in 2025, and is expected to reach USD 1.31 billion by 2030, at a CAGR of 7.12% during the forecast period (2025-2030).
The COVID-19 pandemic had a significant impact on the studied market during its initial phase. Fast-tracking innovation, revamping assembly lines, and expediting manufacturing of medical instruments and devices ranging from N95 masks and personal protective equipment (PPE) to diagnostic kits and ventilators helped Indian companies respond quickly to the COVID-19 pandemic. According to an article published by MoHFW in May 2021, ventilators were available in very limited numbers in government hospitals across the country during the initial phase of the pandemic, and very limited manufacturing of ventilators was taking place in the country and most of the suppliers abroad were not in a position to make supplies of ventilators in large quantities to India. So, local manufacturers were encouraged in India to produce 'Make in India" ventilators to address the huge projected demand of the country and orders placed on them. Hence, the COVID-19 pandemic had a significant impact on the market initially, however, currently, as the pandemic has subsided, the demand for these devices has also gone down as a result the market has lost some traction, but covid pandemic has lead to increased awarness about use of several respiratory products and thus it is expected to follow stable growth during the forecast period of the study.
The India respiratory devices market is expected to show rapid growth due to the increasing prevalence of respiratory disorders such as COPD, TB, asthma, and sleep apnea, along with technological advancements and increasing applications in home care settings.
Rapid urbanization in the country and a large amount of pollution have contributed to a higher incidence rate of respiratory diseases in India. For instance, according to the article published by PubMed in August 2021, the prevalence of COPD among the population aged 30 years and above in India was 7%. Such studies demonstrating the prevalence of respiratory diseases in the country are driving the demand for respiratory devices, ultimately boosting the growth of the market.
Furthermore, the strategic initiatives undertaken by the market players are also boosting the growth of the market. For instance, in May 2021, Vyaire Medical, a global company focused exclusively on breathing devices, entered into an agreement with GenWorks, a healthcare solutions provider with 140 locations within India and a direct presence in 27 states. Such initiatives taken by the market players are also propelling the growth of the market in the country.
Thus, factors such as rapid urbanization in the country and a large amount of pollution, and the strategic initiatives undertaken by the market players are expected to drive the growth of the respiratory devices market in India. However, the high cost of devices may hinder the growth of the market over the forecast period.
India Respiratory Devices Market Trends and Insights
Spirometers is Anticipated to Have Significant Growth Over the Forecast Period
The spirometer segment is anticipated to have significant growth in the Indian respiratory devices market over the forecast period. The spirometer is a medical device used to measure the proper functioning of the lungs. The volume of air inhaled and exhaled from the lungs is measured after maximum inhalation by the patient. A spirometer helps identify different respiratory diseases, such as bronchitis, chronic lung diseases that are obstructive, emphysema, asthma, and other breathing diseases, for screening in an occupational environment.
Furthermore, the increasing use of spirometers for the diagnosis of various respiratory diseases is expected to enhance segment growth. For instance, according to an article published by PubMed Central in August 2022, chronic obstructive pulmonary disease (COPD) diagnosis requires an airflow obstruction (AFO) using a spirometer, according to the global initiative for chronic obstructive lung disease guidelines, and spirometric testing is typically reserved for individuals with respiratory symptoms.
Additionally, the initiatives taken by the market players are expected to drive the growth of the market segment. For example, on World Chronic Obstructive Pulmonary Disease (COPD) Day in 2021, Cipla Limited launched Spirofy, India's first pneumotach-based portable, wireless spirometer.
Moreover, spirometers are also used to check the proper functioning of the lungs after treatment of pulmonary tuberculosis. For instance, according to an article published by the Indian Journal of Community Medicine in March 2022, spirometers were used to check the lung function impairment among patients suffering from tuberculosis in India. Hence, the increasing prevalence of tuberculosis in the country is also expected to enhance segment growth.
So, the uses of spirometers in India make it easier for patients to use this diagnostic tool, especially for asthma. This device is also being used more and more in hospitals and at home, so the segment is expected to grow over the next few years.


CPAP Devices Segment is Expected to Hold a Significant Share in the Market Over the Forecast Period
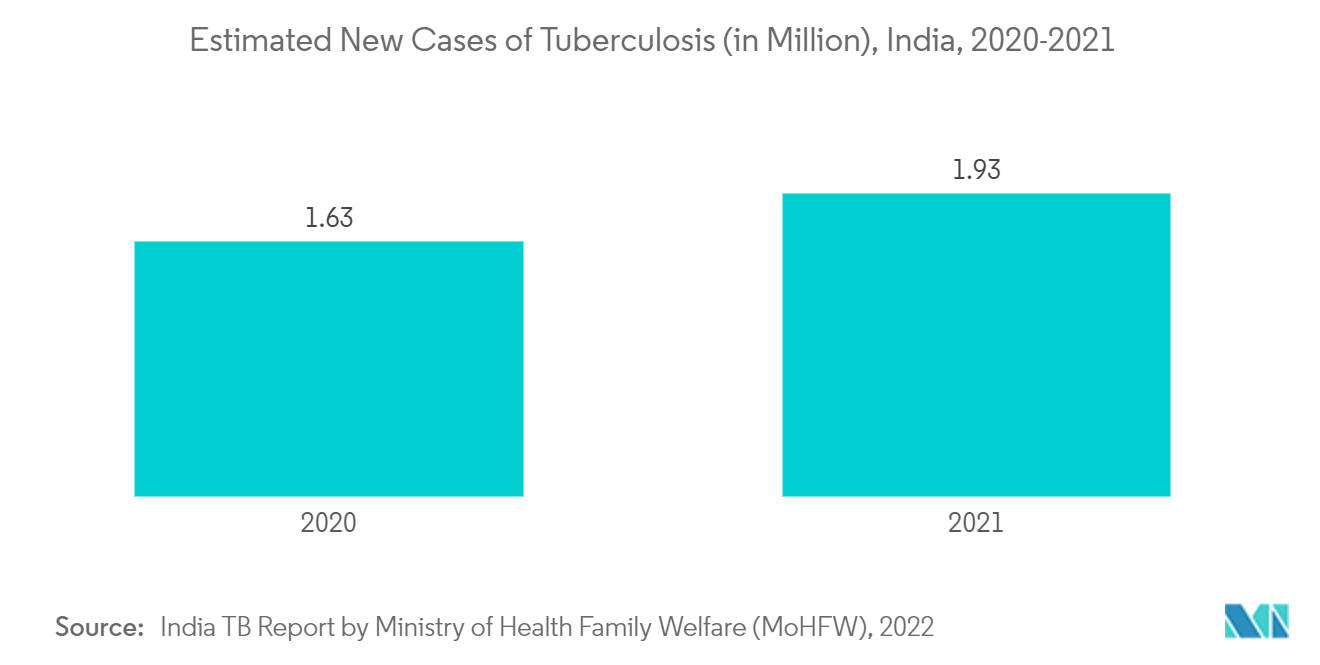
The major factors driving the growth of the segment are the rising geriatric population in the country, the increasing prevalence of sleep apnea, and increasing research and development activities with respect to CPAP devices.
CPAP or continuous positive airway pressure devices are used for the treatment of sleep apnea. The increasing prevalence of sleep apnea in the country is one of the major factors leading to the segment growth and as there is an association between old age and sleep apnea, the rising geriatric population is also expected to enhance the segment growth. For instance, according to a conference on 'dental sleep medicine' held in India in September 2021, it is estimated that around 4 million people, particularly the elderly and obese people are currently suffering from obstructive sleep apnea in India. The experts in the conference also stated CPAP devices are used for the treatment of sleep apnea and that if someone is waking up multiple times at night due to breathlessness and having a dry mouth in the morning with headache and fatigue all day, it might be due to obstructive sleep apnea.
Furthermore, key developments related to CPAP devices in the country are also leading to the growth of the segment. For instance, in June 2021, the Indian Institute of Technology, Ropar developed a power-free CPAP device called 'Jivan Vayu'. It is the Nation's one of the first such devices that functions even without electricity and is adapted to both kinds of oxygen generation units like O2 cylinders and oxygen pipelines in hospitals.
Hence, factors such as the increasing prevalence of sleep apnea, the rising geriatric population, and key developments related to CPAP devices in the country are expected to enhance the growth of the segment during the forecast period of the study.


Competitive Landscape
The India respiratory devices market is moderately competitive. Some of India's established respiratory device companies include Drägerwerk AG & Co. KGaA, GE Healthcare, Fisher & Paykel Healthcare Ltd, Koninklijke Philips N.V., and Devilbiss-AMS Healthcare among others. These companies present in the market focus on product innovations, expansions, finding new markets, or innovating their core competency to expand their market share.
India Respiratory Devices Industry Leaders
Drägerwerk AG & Co. KGaA
GE Healthcare
Fisher & Paykel Healthcare Ltd
Koninklijke Philips N.V.
Devilbiss-AMS Healthcare
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- November 2022 : Xplore Health Technologies launched a first-of-its-kind respiratory muscle training (RMT) device called Airofit PRO in collaboration with Airofit, Denmark.
- October 2022: AirPhysio partnered with Apollo Hospitals Group and Medsmart to launch respiratory devices in India.


India Respiratory Devices Market Report Scope
As per the scope of the report, respiratory devices supply respiratory gas mechanically to patients with impaired respiratory function. The respiratory gas is usually enriched with oxygen and conveyed into the lungs with a positive pressure generated by the device. The respiratory devices include respiratory diagnostic devices, therapeutic devices, and breathing devices for administering long-term artificial respiration. It may also include a breathing apparatus used for resuscitation by forcing oxygen into the lungs of a person who has undergone asphyxia. The India Respiratory Devices Market is Segmented by Type (Diagnostic and Monitoring Devices (Spirometers, Sleep Test Devices, Peek Flow Meters, Pulse Oximeters, Capnographs, and Other Diagnostics and Monitoring Devices), Therapeutic Devices (CPAP Devices, BiPAP Devices, Humidifiers, Nebulizers, Oxygen Concentrators, Ventilators, Inhalers, and Other Therapeutic Devices) and Disposables (Masks, Breathing Circuits and Other Disposables)). The report offers the value (in USD million) for the above segments.
| Diagnostic and Monitoring Devices | Spirometers |
| Sleep Test Devices | |
| Peak Flow Meters | |
| Pulse Oximeters | |
| Capnographs | |
| Other Diagnostic and Monitoring Devices | |
| Therapeutic Devices | CPAP Devices |
| BiPAP Devices | |
| Humidifiers | |
| Nebulizers | |
| Oxygen Concentrators | |
| Ventilators | |
| Inhalers | |
| Other Therapeutic Devices | |
| Disposables | Masks |
| Breathing Circuits | |
| Other Disposables |
| By Type | Diagnostic and Monitoring Devices | Spirometers |
| Sleep Test Devices | ||
| Peak Flow Meters | ||
| Pulse Oximeters | ||
| Capnographs | ||
| Other Diagnostic and Monitoring Devices | ||
| Therapeutic Devices | CPAP Devices | |
| BiPAP Devices | ||
| Humidifiers | ||
| Nebulizers | ||
| Oxygen Concentrators | ||
| Ventilators | ||
| Inhalers | ||
| Other Therapeutic Devices | ||
| Disposables | Masks | |
| Breathing Circuits | ||
| Other Disposables | ||


Key Questions Answered in the Report
How big is the India Respiratory Devices Market?
The India Respiratory Devices Market size is expected to reach USD 0.93 billion in 2025 and grow at a CAGR of 7.12% to reach USD 1.31 billion by 2030.
What is the current India Respiratory Devices Market size?
In 2025, the India Respiratory Devices Market size is expected to reach USD 0.93 billion.
Who are the key players in India Respiratory Devices Market?
Drägerwerk AG & Co. KGaA, GE Healthcare, Fisher & Paykel Healthcare Ltd, Koninklijke Philips N.V. and Devilbiss-AMS Healthcare are the major companies operating in the India Respiratory Devices Market.
What years does this India Respiratory Devices Market cover, and what was the market size in 2024?
In 2024, the India Respiratory Devices Market size was estimated at USD 0.86 billion. The report covers the India Respiratory Devices Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the India Respiratory Devices Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
India Respiratory Devices Market Report
Statistics for the 2025 India Respiratory Devices market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. India Respiratory Devices analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.





