Hyperhidrosis Treatment Market Size and Share
Hyperhidrosis Treatment Market Analysis by Mordor Intelligence
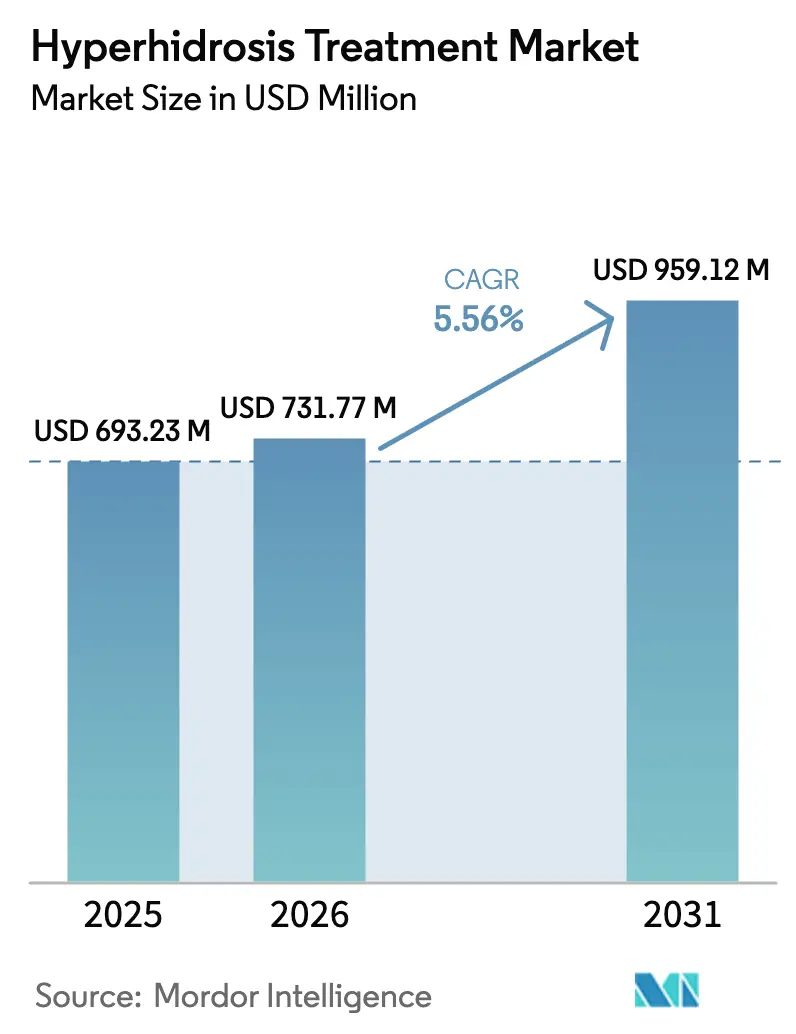
The Hyperhidrosis Treatment Market size is expected to grow from USD 693.23 million in 2025 to USD 731.77 million in 2026 and is forecast to reach USD 959.12 million by 2031 at 5.56% CAGR over 2026-2031.
The industry is transitioning from traditional surgical approaches to precision biologics, energy-based devices, and digitally enabled topical therapies. The U.S. Food and Drug Administration’s approval of sofpironium bromide (Sofdra) in June 2024 underscores the growing acceptance of non-invasive treatment pathways. This development, coupled with Candesant Biomedical’s successful financing for its Brella patch, reflects strong investor confidence in outpatient solutions. While botulinum toxin A injections remain a key revenue driver, the emergence of EU MDR-certified microwave thermolysis platforms and direct-to-consumer tele-dermatology services is expanding patient access and reshaping competitive strategies. Reimbursement policies remain a critical factor: U.S. payers require antiperspirant step therapy before approving prescription topicals, whereas Europe and Japan are showing increased flexibility toward injectables and device-based procedures. Furthermore, the growth of e-commerce subscriptions and home-use iontophoresis kits is driving incremental demand beyond traditional clinical settings, adding a sustainable growth dimension to the hyperhidrosis treatment market.
Key Report Takeaways
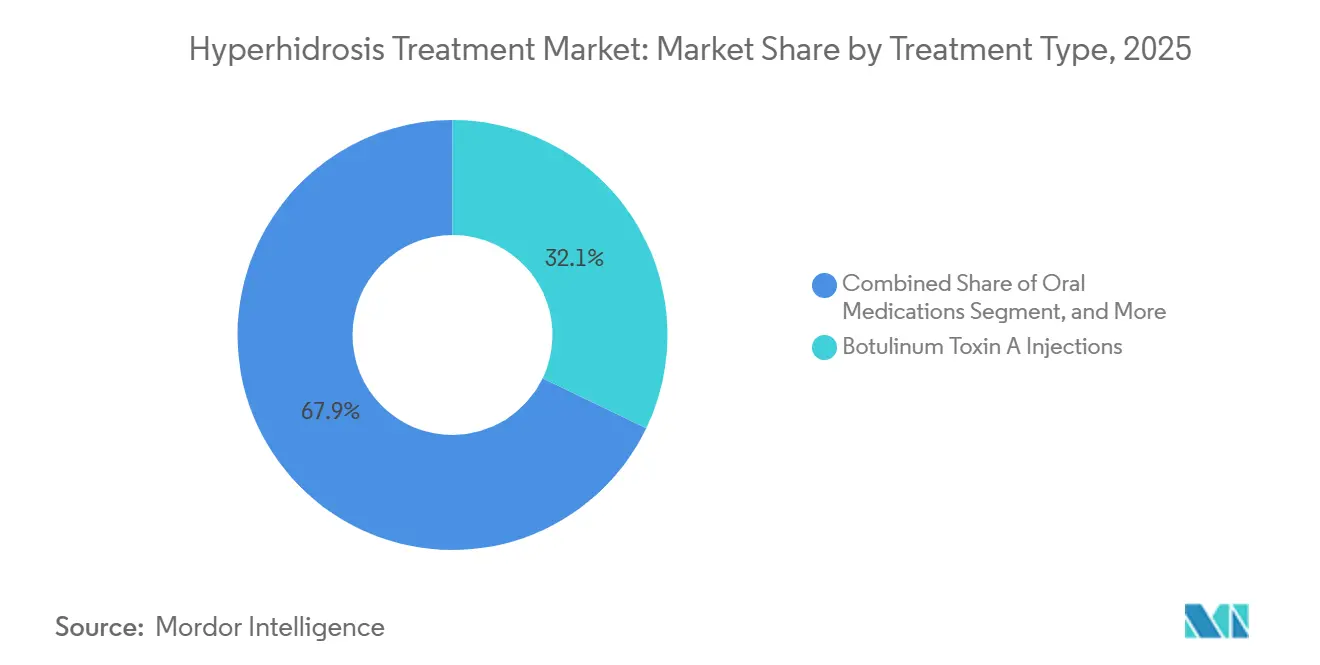
- By treatment type, botulinum toxin A injections held 32.13% of hyperhidrosis treatment market share in 2025, whereas microwave thermolysis is projected to post the fastest 7.54% CAGR through 2031.
- By disease type, primary focal hyperhidrosis accounted for 75.35% of 2025 demand, while secondary generalized cases are set to expand at a 7.43% CAGR to 2031.
- By site, axillary hyperhidrosis accounted for 50.45% of the 2025 volume, but palmar cases will drive growth at a 7.88% CAGR over the forecast horizon.
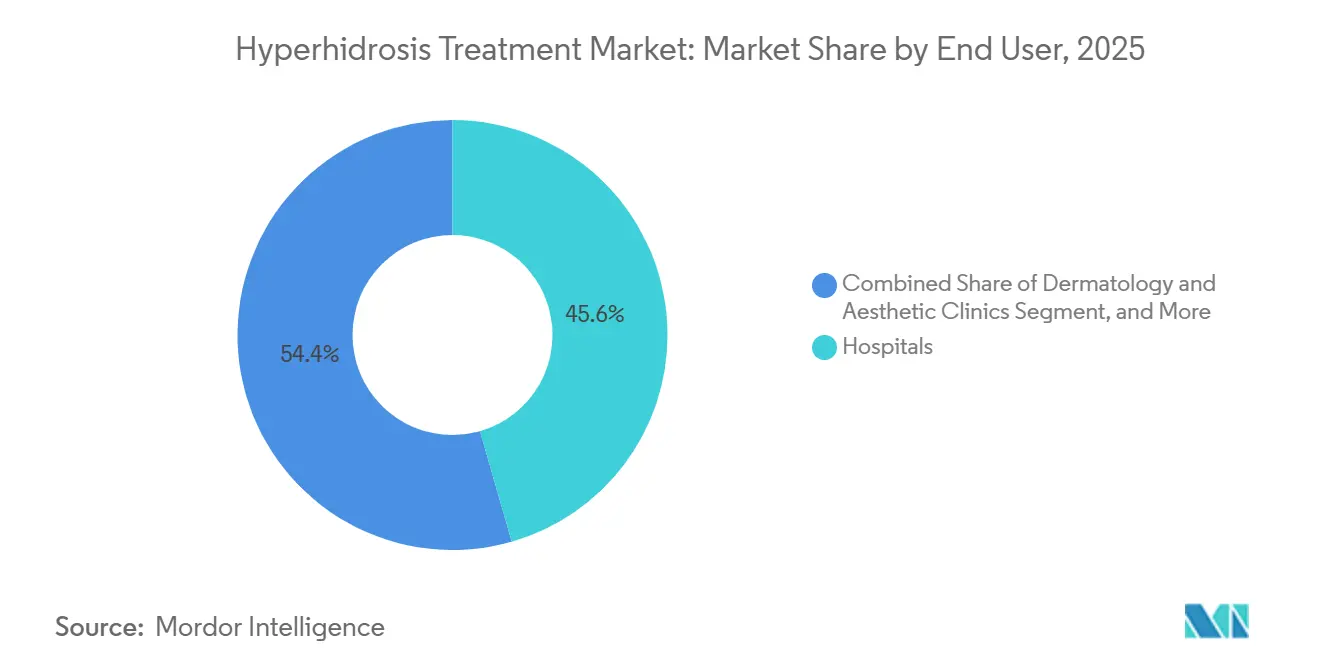
- By end user, dermatology and aesthetic clinics accounted for 45.56% of 2025 revenue, while home-care and OTC channels are forecast to grow at an 8.76% CAGR.
- By distribution channel, retail pharmacies accounted for 52.17% of 2025 sales, yet e-commerce is expected to grow at an 8.54% CAGR through 2031.
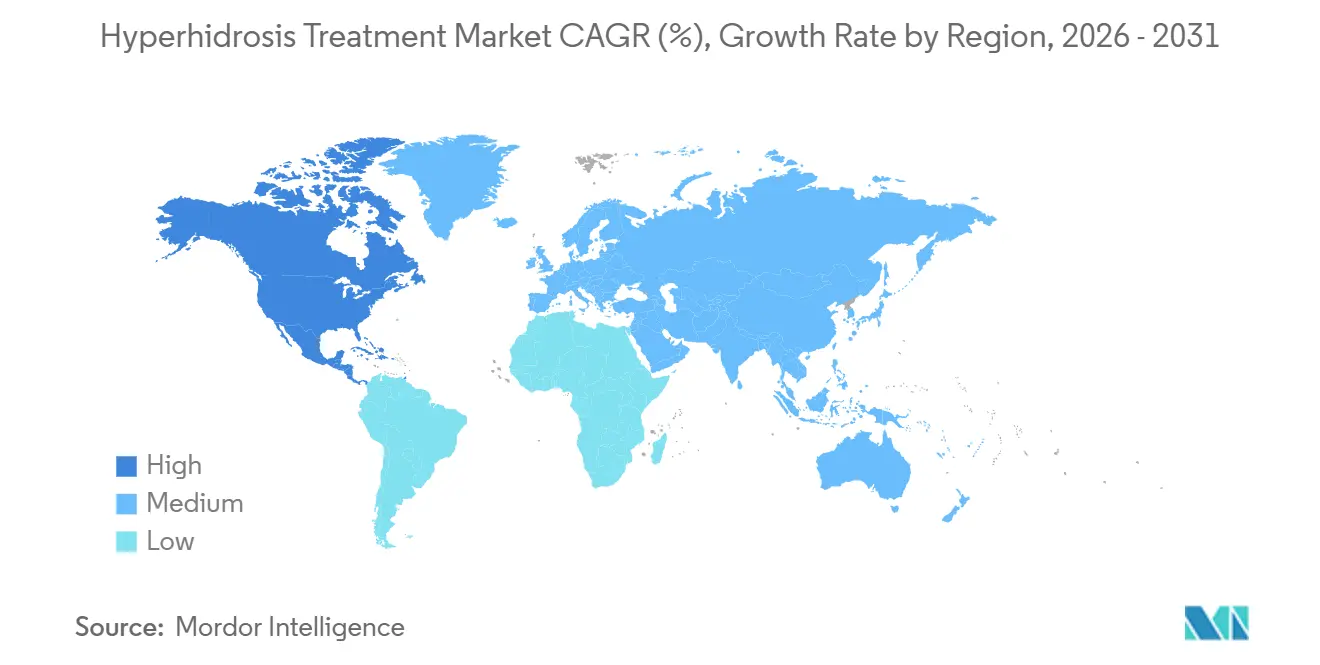
- By geography, North America contributed 42.56% of 2025 turnover, but Asia-Pacific is anticipated to log the fastest 6.43% CAGR on the back of Japanese product launches.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of January 2026.
Global Hyperhidrosis Treatment Market Trends and Insights
Drivers Impact Analysis
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Increasing Prevalence and Diagnosis of Hyperhidrosis | +0.9% | Global, strongest in North America and Western Europe | Medium term (2-4 years) |
| Rising Demand for Minimally and Non-Invasive Treatment Modalities | +1.2% | North America, Europe, APAC urban centers | Short term (≤ 2 years) |
| Regulatory Approvals of Innovative Topical and Device-Based Therapies | +1.0% | North America, Europe, APAC spillover | Medium term (2-4 years) |
| Technological Advancements in Energy-Based and Digital Platforms | +0.8% | North America, Europe, Japan | Long term (≥ 4 years) |
| Growing Consumer Awareness and Quality-of-Life Focus | +0.7% | Global | Medium term (2-4 years) |
| Expansion of Tele-Dermatology and Direct-to-Consumer Channels | +0.6% | North America, Europe, APAC urban markets | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Increasing Prevalence and Diagnosis of Hyperhidrosis
Epidemiological data indicate that sweating disorders affect 4.8% of the U.S. population, or 15.3 million individuals, and 5.5% of Sweden's population, underscoring their significant prevalence in high-income markets. The adoption of tools such as the Hyperhidrosis Disease Severity Scale has streamlined payer documentation processes and accelerated clinical presentation timelines. The FDA approval of Sofdra, following the successful CARDIGAN trial, has established objective responder endpoints as critical benchmarks for reimbursement. Simultaneously, advocacy initiatives are driving patient engagement, effectively converting unmet needs into treated prevalence within the hyperhidrosis treatment market.
Rising Demand for Minimally and Non-Invasive Treatment Modalities
High compensatory sweating rates of 40%-89% following endoscopic thoracic sympathectomy have driven patients toward reversible treatment options[1]Health Products Regulatory Authority, “Risk Assessment of ETS,” hpra.ie. Candesant’s FDA-cleared Brella patch delivers a quick, three-minute, office-based solution with a 63.6% responder rate, eliminating the need for systemic drug exposure. Similarly, MiraDry’s dual-frequency microwave thermolysis has received EU MDR clearance, positioning it to enter more stringent European markets by 2025. These advancements are driving growth in the hyperhidrosis treatment market by addressing patient demand for effective, non-surgical, and low-maintenance solutions.
Regulatory Approvals of Innovative Topical and Device-Based Therapies
With Sofdra now approved in the United States and under PMDA review in Japan, the topical anticholinergic segment is regaining momentum after a six-year stagnation. Recent updates from the European Medicines Agency (EMA) to botulinum toxin labeling, along with EU Medical Device Regulation (MDR) certification for devices, are standardizing clinical dosing and safety data. This regulatory alignment is facilitating faster payer acceptance. As a result, the hyperhidrosis treatment market, supported by a strong pipeline, is well-positioned for accelerated cross-regional launches, enabling quicker recovery of R&D investments.
Technological Advancements in Energy-Based and Digital Platforms
MiraDry’s 84% sweat-reduction data at 12 months using dual-frequency delivery show that device engineering continues to narrow the durability gap versus surgery. Real-time thermal feedback in fractional lasers is reducing adverse events below 5%. Digital advances matter as well: Botanix’s tele-prescribing portal handles asynchronous photo consults, achieving 40% of Sofdra’s first-half sales online. Technology is broadening geographies and demographics served by the hyperhidrosis treatment market.
Restraints Impact Analysis
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High Treatment Costs and Limited Reimbursement Coverage | -0.8% | Global, acute in emerging markets & U.S. uninsured | Short term (≤ 2 years) |
| Limited Long-Term Efficacy of Existing Therapies | -0.5% | Global | Medium term (2-4 years) |
| Adverse Events and Safety Concerns with Surgical and Energy Devices | -0.4% | Global, higher scrutiny in Europe post-MDR | Medium term (2-4 years) |
| Supply Chain and Pricing Volatility of Key Therapeutic Inputs | -0.3% | Global, tightest in North America & Europe | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
High Treatment Costs and Limited Reimbursement Coverage
Monthly outlays of USD 580-700 for Qbrexza demand prior authorization, capping mainstream adoption[2]Cigna, “Coverage Policy for Hyperhidrosis Treatments,” cigna.com. Botulinum toxin reimbursements are quantity-limited, and miraDry’s USD 2,000-3,000 fee is seldom covered, painting a price-sensitive reality for the hyperhidrosis treatment market. In lower-income regions, generic aluminum chloride remains the only viable choice, muting near-term premium-therapy penetration.
Limited Long-Term Efficacy of Existing Therapies
Antibody development reduces botulinum toxin response in up to 3% of repeat recipients, while topical anticholinergic adherence wanes after six months due to side effects. Iontophoresis maintenance every 9-10 days deters busy patients. Surgical sympathectomy’s compensatory-sweating risk fuels regret litigation, signaling an unsolved need for lasting, low-burden solutions within the hyperhidrosis treatment market.
Segment Analysis
By Treatment Type: Injectables Lead, Energy Devices Surge
Botulinum toxin A injections captured 32.13% of 2025 demand, translating to robust cash flow for AbbVie’s flagship product line, while Cigna and Blue Cross cover axillary administrations once aluminum chloride fails[3]. Yet microwave thermolysis outpaces the hyperhidrosis treatment market size with a 7.54% CAGR, leveraging miraDry’s EU MDR clearance and 84% 12-month sweat-reduction data to attract clinics seeking capital-light, single-session revenue.
Topical anticholinergics such as Sofdra lessen systemic exposure, filling safety gaps left by Qbrexza. Meanwhile, home iontophoresis systems tested at 9-10-day efficacy cycles supply palmar and plantar sufferers with an OTC workaround, though regimen fatigue remains a headwind. ETS volumes slide further as reversible device options proliferate, leaving surgery a shrinking niche inside the hyperhidrosis treatment market.

By Disease Type: Primary Focal Dominates, Secondary Cases Accelerate
Primary focal hyperhidrosis generated 75.35% of 2025 turnover, buoyed by established diagnostic grading and multiple FDA-approved modalities. Insurer alignment around HDSS scoring eases claim workflows, reinforcing its dominant share within the hyperhidrosis treatment market.
Secondary generalized hyperhidrosis is rising at 7.43% CAGR as oncology regimens and metabolic comorbidities swell patient counts; 30%-50% of hormone-therapy recipients experience problematic sweating, opening demand for systemic anticholinergics and adjunct topicals. Selective M3 antagonists in early pipelines could offer lower side-effect loads, signaling future competition for entrenched oral options.
By Site of Hyperhidrosis: Axillary Sites Lead, Palmar Cases Gain
Axillary presentations owned 50.45% of 2025 procedure volume, underpinned by Sofdra, Qbrexza, and injectables that fit a well-reimbursed care pathway. Device players such as Brella and miraDry specifically target underarm glands, reinforcing incumbent dominance.
Palmar hyperhidrosis, however, is forecast to eclipse overall hyperhidrosis treatment market growth at a 7.88% CAGR, propelled by home-use iontophoresis kits and emergent fast-dry gels aimed at hand-sweating professionals. Plantar and craniofacial sites remain tougher to treat, but clinical-strength wipes and refined laser protocols are making incremental inroads.
By End User: Clinics Dominate, Homecare Channels Surge
Dermatology and aesthetic clinics delivered 45.56% of 2025 revenue thanks to procedure-heavy injectables and microwave thermolysis sessions, each of which fit high-throughput practice economics. Hospitals manage complex secondary cases yet remain volume-constrained.
Home care and OTC demand is set to grow at an 8.76% CAGR as direct-to-consumer brands leverage e-commerce scale. SweatBlock’s towelette subscriptions and Dermadry’s USD 499 kits let consumers bypass appointment queues, extending the hyperhidrosis treatment market size beyond brick-and-mortar care.

By Distribution Channel: Retail Pharmacies Lead, E-Commerce Accelerates
Retail pharmacies filled 52.17% of 2025 prescriptions, driven by PBM-routed Qbrexza and Sofdra volumes and a constant stream of OTC aluminum chloride refills. Pharmacist counseling improves persistence, anchoring current dominance.
E-commerce should post an 8.54% CAGR through 2031 as Amazon and regional e-pharmacies scale auto-ship routines and co-pay assistance plug-ins. Botanix’s integrated script-to-door model already funnels 40% of Sofdra’s sales through digital touchpoints, foreshadowing wider channel shift across the hyperhidrosis treatment market.
Geography Analysis
In 2025, North America captured 42.56% of sales, buoyed by favorable coverage policies, high per-capita spending, and a swift embrace of biologics. Cigna's 2024 update, which removed the requirement for iontophoresis trials prior to injectable authorizations, streamlined treatment initiation. Meanwhile, Sofdra's direct-to-consumer debut gained momentum online, and U.S. consumers are benefiting from reduced out-of-pocket expenses due to cross-border procedure tourism in Mexico. This dynamic creates a landscape where revenue in the hyperhidrosis treatment market is both accelerated and redistributed.
Europe made strides, bolstered by botulinum toxin reimbursement parity and the July 2025 EU MDR nod for microwave thermolysis in Germany, France, and Italy. The EMA's standardized labeling boosted clinician dosing confidence. However, France's stringent cost-effectiveness evaluations tempered the entry of premium topicals. Despite these challenges, statutory coverage for severe cases solidifies Europe's pivotal role in the hyperhidrosis treatment arena.
Asia-Pacific is set to expand at a 6.43% CAGR, spearheaded by Japan. Here, Maruho's glycopyrronium tosylate formulation, with a 3.75% concentration, boasted a 51.6% responder rate in late-stage trials. China's growing aesthetic demand and India's 35% boom in e-pharmacy broaden the market's reach, even as modest household incomes dampen prescription rates. Meanwhile, Australia and South Korea, driven by a surge in private clinic demand, contribute to the region's status as the fastest-growing in the hyperhidrosis treatment market.
Competitive Landscape
The hyperhidrosis treatment market is moderately fragmented. The injectables segment is led by AbbVie, Merz Pharma, and Revance. In Q3 2024, AbbVie achieved a therapeutic turnover of USD 848 million, driven by its Botox Savings incentive program. Organon's USD 1.2 billion acquisition of Dermavant highlights big pharma's strategic focus on late-stage topical franchises, potentially challenging Qbrexza’s market share.
Digital-first entrants are gaining traction in the market. Botanix captured 40% of Sofdra’s initial demand through telehealth channels, while Candesant’s Brella patch, with its 12-month durability claim, threatens to disrupt repeat neurotoxin treatments. Iontophoresis device providers Dermadry and Hidrex are targeting cost-conscious consumers with products priced between USD 499-950, gradually eroding the market share of traditional clinic operators.
Technology-driven collaborations are increasing. In 2025, Dermata partnered with Revance to develop combined topical and injectable regimens to extend the efficacy of neurotoxins. With advancements such as microneedle-delivered neurotoxins and selective muscarinic antagonists in the pipeline, market leaders must adopt an acquisitive approach to remain competitive in the rapidly evolving hyperhidrosis treatment landscape.
Hyperhidrosis Treatment Industry Leaders
AbbVie Inc.
Eli Lilly (Dermira)
Sientra Inc.
Merz Pharma
Brickell Biotech Inc.
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- March 2025: Dr. August Wolff GmbH & Co. KG Arzneimittel, a German pharmaceutical company, announced the submission of an extension of the indication for its 1% glycopyrronium bromide (GPB) cream. The extension aims to include adolescents (aged 12 years and older) with severe primary axillary hyperhidrosis.
- January 2025: Dermata and Revance began a Phase 2a trial pairing topical prabotulinumtoxinA with injectable daxibotulinumtoxinA to lengthen symptom-free intervals.
- September 2024: Organon acquired Dermavant for up to USD 1.2 billion, adding late-stage topical anticholinergics to its dermatology roster.
Global Hyperhidrosis Treatment Market Report Scope
As per scope of the report, hyperhidrosis treatment involves medical options to reduce excessive sweating, including topical agents, medications, Botox injections, and surgical procedures. The goal is to control symptoms and improve quality of life.
The Hyperhidrosis Treatment Market is Segmented by Treatment Type (Topical Antiperspirants & Anticholinergics, Botulinum Toxin A Injections, Iontophoresis Devices, Microwave Thermolysis, ETS, Laser & Energy-Based Therapies, Oral Medications, and Other Treatment Types), Disease Type (Primary Focal and Secondary Generalised), Site (Axillary, Palmar, Plantar, Craniofacial, and Other), End User (Hospitals, Dermatology & Aesthetic Clinics, Ambulatory Surgical Centers, and Homecare & OTC Channels), Distribution Channel (Hospital Pharmacies, Retail Pharmacies & Drug Stores, and E-Commerce), and Geography (North America, Europe, Asia-Pacific, Middle East & Africa, and South America). The report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers value (in USD million) for the above segments.
| Topical Antiperspirants & Anticholinergics |
| Botulinum Toxin A Injections |
| Iontophoresis Devices |
| Microwave Thermolysis |
| Endoscopic Thoracic Sympathectomy (ETS) |
| Laser & Energy-Based Therapies |
| Oral Medications |
| Other Treatment Types |
| Primary Focal Hyperhidrosis |
| Secondary Generalised Hyperhidrosis |
| Axillary (Underarms) |
| Palmar (Hands) |
| Plantar (Feet) |
| Craniofacial |
| Other Site of Hyperhidrosis |
| Hospitals |
| Dermatology & Aesthetic Clinics |
| Ambulatory Surgical Centers |
| Homecare & OTC Channels |
| Hospital Pharmacies |
| Retail Pharmacies & Drug Stores |
| E-Commerce |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East & Africa | GCC |
| South Africa | |
| Rest of Middle East & Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Treatment Type | Topical Antiperspirants & Anticholinergics | |
| Botulinum Toxin A Injections | ||
| Iontophoresis Devices | ||
| Microwave Thermolysis | ||
| Endoscopic Thoracic Sympathectomy (ETS) | ||
| Laser & Energy-Based Therapies | ||
| Oral Medications | ||
| Other Treatment Types | ||
| By Disease Type | Primary Focal Hyperhidrosis | |
| Secondary Generalised Hyperhidrosis | ||
| By Site of Hyperhidrosis | Axillary (Underarms) | |
| Palmar (Hands) | ||
| Plantar (Feet) | ||
| Craniofacial | ||
| Other Site of Hyperhidrosis | ||
| By End User | Hospitals | |
| Dermatology & Aesthetic Clinics | ||
| Ambulatory Surgical Centers | ||
| Homecare & OTC Channels | ||
| By Distribution Channel | Hospital Pharmacies | |
| Retail Pharmacies & Drug Stores | ||
| E-Commerce | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| Japan | ||
| India | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East & Africa | GCC | |
| South Africa | ||
| Rest of Middle East & Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What is the projected size of the hyperhidrosis treatment market by 2031?
It is forecast to reach USD 959.12 million by 2031, expanding at a 5.56% CAGR from 2026.
Which therapy currently holds the largest share in treating excessive sweating?
Botulinum toxin A injections led with 32.13% hyperhidrosis treatment market share in 2025.
Which region will post the fastest growth in demand for sweat-management solutions?
Asia-Pacific is expected to grow at 6.43% CAGR, helped by Japanese topical launches and wider device adoption.
How long does microwave thermolysis like miraDry typically last?
Dual-frequency miraDry data showed 84% sweat reduction sustained 12 months after a single session.
Why is Sofdra gaining attention among dermatologists?
Sofdra offers topical anticholinergic efficacy with reduced systemic exposure and is available through direct-to-consumer tele-prescribing.
Are home-use devices effective for palmar hyperhidrosis?
Iontophoresis kits such as Dermadry's system achieve dryness for 9-10 days per session, supporting regular but clinic-free management.
Page last updated on:









