Pharmaceutical Spray Drying Market Size and Share
Pharmaceutical Spray Drying Market Analysis by Mordor Intelligence
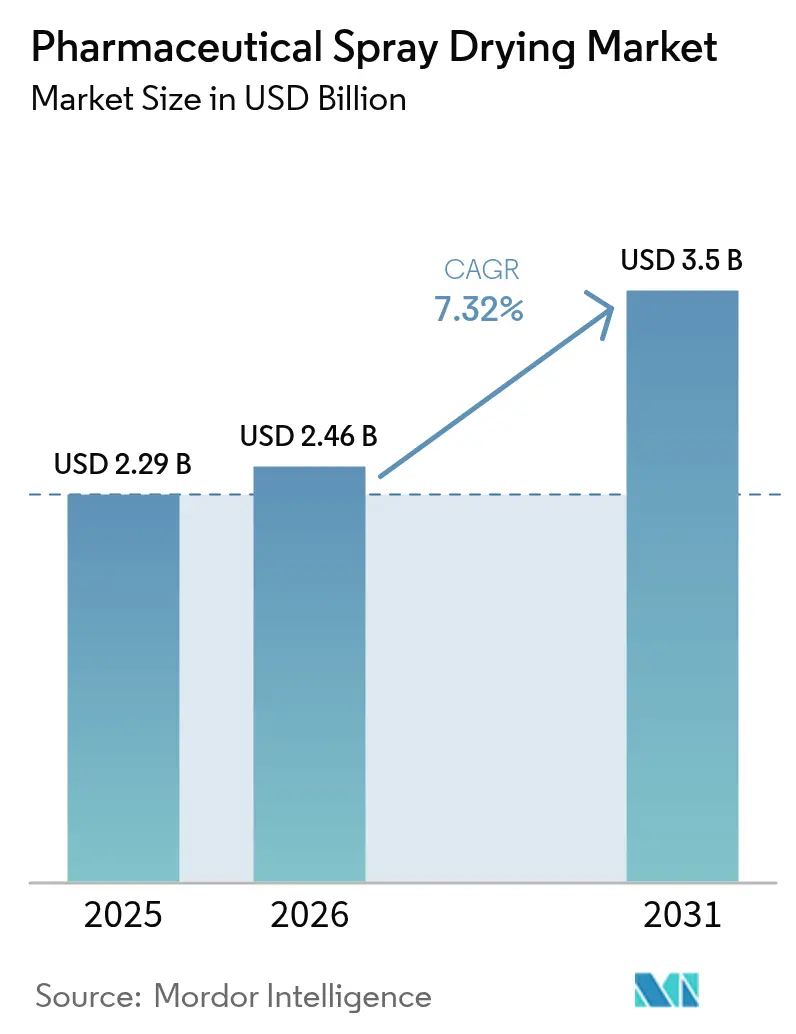
The Pharmaceutical Spray Drying Market size was valued at USD 2.29 billion in 2025 and is estimated to grow from USD 2.46 billion in 2026 to reach USD 3.5 billion by 2031, at a CAGR of 7.32% during the forecast period (2026-2031).
Strong demand for amorphous solid dispersions that improve drug solubility, a rapid shift toward inhalable biologics, and the expansion of contract development and manufacturing organization (CDMO) capacity keep the pharmaceutical spray drying market on a steady growth path. Equipment vendors are shipping more closed-loop units that capture 95% of solvent vapors, a feature that satisfies stricter volatile organic compound rules in the United States and the European Union. Sponsors are also directing R&D budgets toward early-stage screening, which is pushing laboratory and pilot throughput higher and shortening formulation cycles. The emergence of digital-twin controls that predict outlet temperature, particle size, and residual moisture in real time improves batch consistency and reduces the need for re-work, which strengthens the cost case for spray drying. At the same time, a limited pool of excipient suppliers and capital costs that exceed USD 10 million for a single GMP suite remain structural hurdles that slow adoption in smaller companies.
Key Report Takeaways
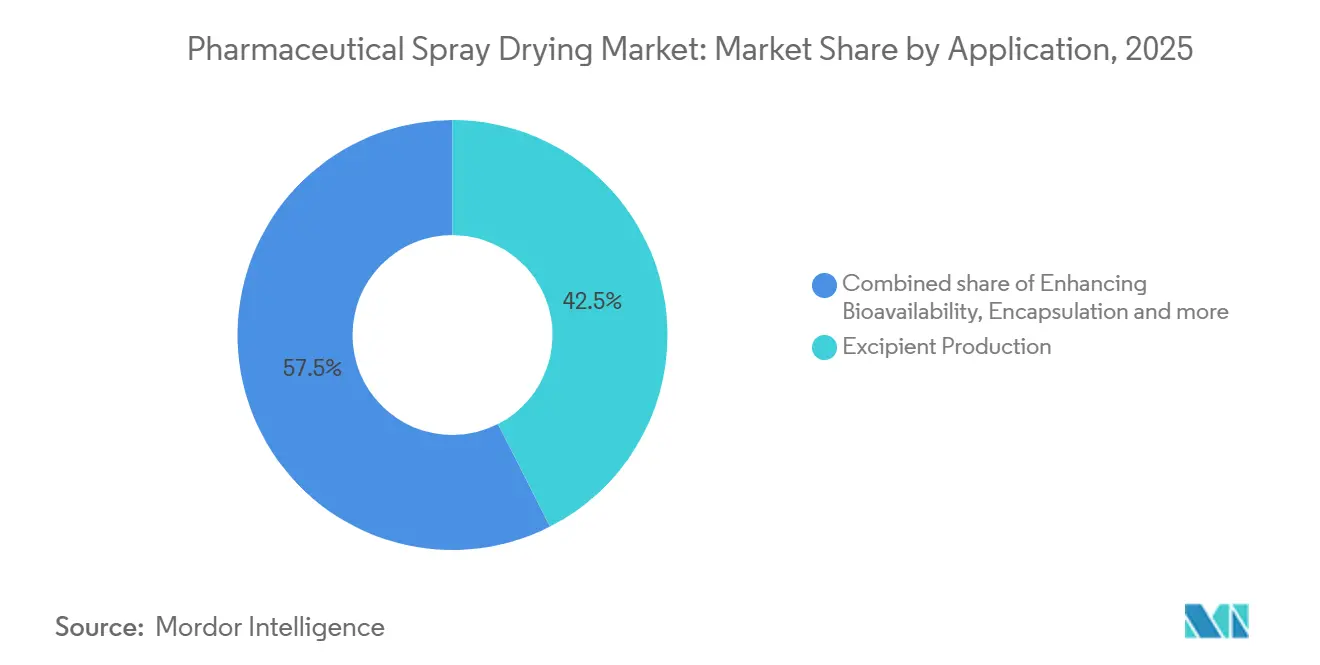
- By application, excipient production commanded 42.55% of the pharmaceutical spray drying market share in 2025. Bioavailability enhancement applications are forecast to post the fastest growth at a 9.85% CAGR through 2031.
- By formulation type, small-molecule APIs led with 46.53% of the pharmaceutical spray drying market size in 2025. Vaccines are projected to expand at a 9.75% CAGR between 2026 and 2031, outpacing all other formulation types.
- By spray-dryer type, rotary atomizers accounted for 41.15% of installations in 2025, while fluidized and closed-loop systems are advancing at a 9.82% CAGR through 2031.
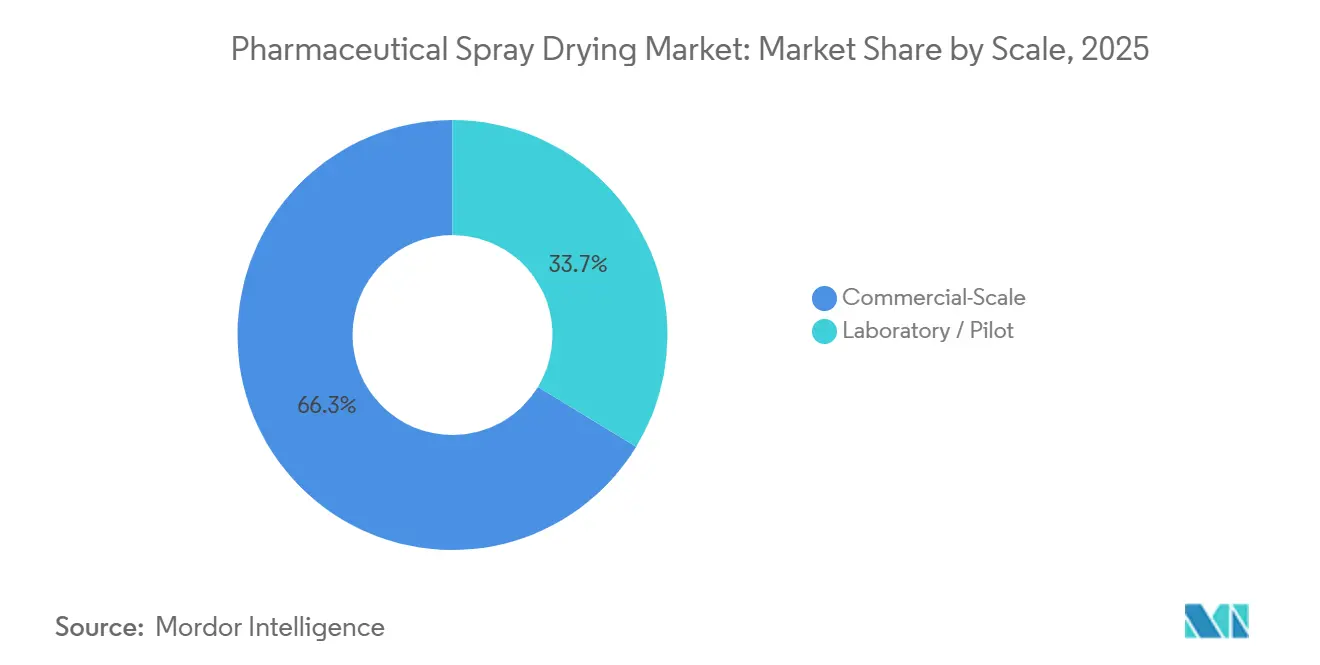
- By scale, commercial-scale operations commanded 66.32% of the pharmaceutical spray drying market size in 2025; laboratory/pilot facilities are projected to grow at 9.29% CAGR.
- By service type, in-house manufacturing commanded 58.21% of the market size in 2025; contract (CDMO/CMO) services are projected to grow at 10.56% CAGR.
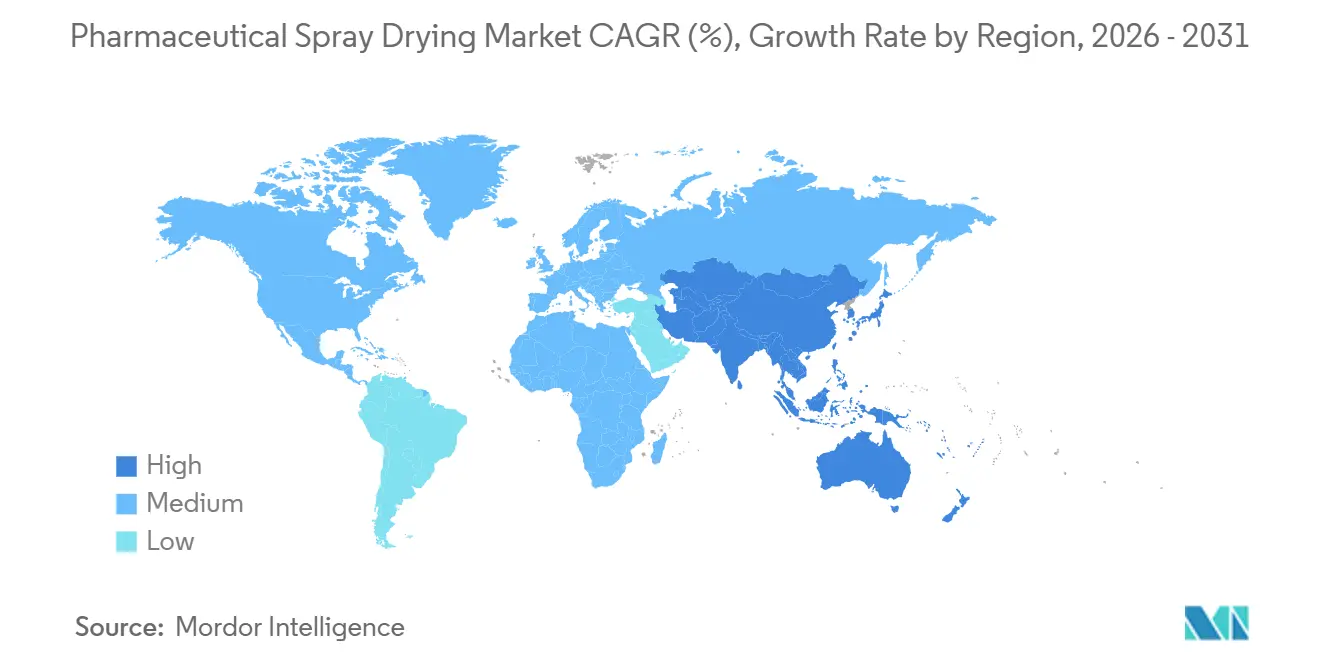
- By geography, North America captured 39.52% share of the pharmaceutical spray drying market size in 2025; Asia-Pacific will accelerate at an 8.32% CAGR during 2026-2031.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Pharmaceutical Spray Drying Market Trends and Insights
Driver Impact Analysis*
| Driver | % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Growing demand for enhanced drug solubility | +1.8% | Global, stronger in North America and Europe | Medium term (2-4 years) |
| Rising outsourcing to CDMOs | +2.1% | North America, Europe, China, India | Short term (≤ 2 years) |
| Expansion of inhalable & oral thin-film therapies | +1.3% | North America, Europe, Japan | Medium term (2-4 years) |
| Continuous closed-loop manufacturing | +1.2% | Europe, North America, spillover to Asia-Pacific | Long term (≥ 4 years) |
| AI-driven digital-twin optimization | +0.9% | North America, Europe | Long term (≥ 4 years) |
| Temperature-shift high-solids feeds | +0.7% | Global, early uptake in Europe | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
Growing Demand for Enhanced Drug Solubility
Roughly 70% of new chemical entities in late-stage development display poor aqueous solubility, driving formulation scientists to adopt spray drying to create amorphous solid dispersions that dissolve two- to ten-times faster than crystalline forms. FDA approvals of spray-dried drugs such as telaprevir and vemurafenib validate the platform’s ability to meet regulatory expectations on stability and performance. Targeted oncology assets with hydrophobic backbones increasingly rely on hydroxypropyl methylcellulose acetate succinate or Soluplus carriers to remain in solution after oral dosing. The trend lifts the pharmaceutical spray drying market as sponsors funnel pipeline drugs into specialized CDMOs that maintain large compatibility libraries. Broader acceptance of amorphous science by clinicians also improves market prospects because therapeutic exposure can be achieved with lower drug loads.
Rising Outsourcing to CDMOs for Spray-Dried Dispersions
Supply disruptions exposed the vulnerability of single-site manufacturing, prompting companies to pivot toward asset-light operating models. Hovione’s USD 100 million capacity expansion in Ireland and the United States highlights confidence in sustained demand for contracted services. CDMOs compress development timelines by leveraging validated GMP suites, digital data sets, and regulatory expertise that small biotechnology clients cannot replicate internally. The outsourcing wave adds volume to the pharmaceutical spray drying market and promotes standardization of polymer libraries, atomization protocols, and real-time release testing.
Expansion of Inhalable & Oral Thin-Film Therapies
Respirable powders sized between 1 µm and 5 µm deposit in the alveolar region and bypass hepatic metabolism, a benefit that elevates the commercial appeal of pulmonary delivery for peptides and vaccines[1]“Thermostable Vaccine Formulations Using Spray Drying Technology,” Nature Communications, nature.com. Trehalose and leucine matrices protect thermolabile actives against shear and temperature stress during spray drying and aerosolization. Parallel interest in oral thin films that dissolve within 30 seconds aligns with the needs of pediatric and geriatric patients. FDA guidance released in 2024 on inhalation products underscores the importance of tight particle-size distribution and low residual moisture, two parameters that spray drying controls well, adding momentum to the pharmaceutical spray drying market.
Continuous Closed-Loop Manufacturing Adoption
The European Union limits VOC emissions to 20 mg/m³, splitting economics in favor of closed-loop nitrogen systems that capture 95% of organic solvents. Solvent recovery cuts annual raw-material bills by as much as USD 500,000 on 500 kg-per-day lines and removes explosive atmospheres, which increases plant safety. GEA’s ConsiGma platform links spray drying, granulation, and tableting inside one continuous line, shrinking batch times from three days to half a day. Continuous designs make process validation simpler because smaller residence times reduce variability, which sustains CAGR uplift in the pharmaceutical spray drying market.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High capital & operating costs for GMP units | –1.4% | Global, more acute in emerging markets | Short term (≤ 2 years) |
| Stringent validation & solvent-emission rules | –1.1% | North America, Europe, Japan | Medium term (2-4 years) |
| Limited supply of spray-drying-grade excipients | –0.8% | Global, supply centers in Europe and North America | Medium term (2-4 years) |
| Energy intensity & sustainability concerns | –0.6% | Global, regulatory pressure in Europe | Long term (≥ 4 years) |
| Source: Mordor Intelligence | |||
High Capital & Operating Costs for GMP Units
Building a single GMP spray drying suite costs USD 10 million–USD 15 million once cleanroom, solvent recovery, and process analytical technology are included. Operating expenses can reach USD 1 million per year for a 500 kg-per-day plant that consumes nitrogen, electricity, and validation labor. Smaller firms in emerging markets struggle to justify the expense because financing is scarce and payback horizons exceed seven years. Although CDMO fees lower the upfront burden, prices of USD 5,000–USD 15,000 per kilogram erode margins on high-volume generics, which tempers growth in parts of the pharmaceutical spray drying market.
Stringent Validation & Solvent-Emission Rules
FDA 21 CFR Part 11 compliance mandates electronic audit trails and process verification that can add six months to a commercial launch and raise analytical costs by USD 1 million[2]Q8(R2) Pharmaceutical Development,” U.S. Food and Drug Administration, fda.gov. U.S. NESHAP standards and the European Industrial Emissions Directive cap solvent exhaust to levels that open-cycle dryers cannot meet without scrubbers, which cost USD 2 million–USD 5 million. The double burden of quality and environmental rules slows investment timelines and compresses the pharmaceutical spray drying market CAGR in cost-sensitive regions.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Application: Excipient Primacy Masks Bioavailability Surge
Excipient production held 42.55% of the pharmaceutical spray drying market share in 2025, anchored by large-volume outputs of lactose, microcrystalline cellulose, and hydroxypropyl methylcellulose powders. Manufacturers favor rotary atomizers because they handle viscous feeds and deliver the flow characteristics that tablet presses require. Yet bioavailability enhancement is expanding at a 9.85% CAGR through 2031 as drug pipelines contain more BCS Class II and IV molecules that need amorphous solid dispersions to reach target exposures.
The pharmaceutical spray drying market is therefore tilting toward value-added dispersion services that rescue otherwise unviable candidates. Encapsulation, which embeds active ingredients in enteric or taste-masking polymers, is also rising in pediatric and geriatric niches. Niche uses such as probiotic and enzyme stabilization add incremental demand, but the growth inflection is clearly linked to solubility challenges that mainstream chemistries cannot solve.

By Formulation Type: Small Molecules Dominate, Vaccines Accelerate
Small-molecule APIs accounted for 46.53% of pharmaceutical spray drying market revenue in 2025 as formulators continued to convert crystalline actives into fast-dissolving dispersions. Rotary and nozzle systems manage the thermal profile well enough to protect synthetic compounds, securing the installed base. Vaccines, however, are projected to grow at a 9.75% CAGR between 2026 and 2031, supported by global investment in thermostable pulmonary and oral platforms that reduce cold-chain reliance.
Thermal stress remains a hurdle for mRNA and lipid nanoparticle constructs, yet cryoprotectants such as trehalose preserve bioactivity below 50 °C outlet temperatures. Biologics and peptides, once viewed as unsuitable for spray drying, now benefit from low-shear nozzle designs and closed-loop nitrogen setups that keep oxygen levels below flammability limits. Collectively, these innovations raise the long-term addressable share of the pharmaceutical spray drying market size.
By Spray Dryer Type: Closed-Loop Systems Capture Sustainability Premium
Rotary atomizers remained the workhorse with 41.15% of installed units in 2025, serving high-throughput excipient lines. Nonetheless, fluidized and closed-loop setups are expanding at 9.82% CAGR to 2031 because they cut solvent usage and meet emission caps without add-on scrubbers.
Closed-loop nitrogen dryers recover up to 98% of ethanol, acetone, or dichloromethane, improving plant safety and operating economics. Nozzle atomizers, prized for finer droplets, are gaining share in peptide and vaccine work where thermal exposure must remain low. Others, such as ultrasonic systems, serve ultra-low moisture niches, but their contribution to the pharmaceutical spray drying market size is still minor.
By Scale: Pilot Units Outpace Commercial as Formulation Complexity Rises
Commercial plants processed 66.32% of total tonnage in 2025 because excipient makers and generics suppliers require 300 kg-plus daily throughput to keep unit costs competitive. Yet laboratory and pilot units are rising at a 9.29% CAGR through 2031, mirroring a pipeline dominated by poorly soluble molecules that demand rapid formulation screening.
Pilot lines, with 1 kg–50 kg capacity, enable proof-of-concept work without tying up commercial assets. They let sponsors fine-tune polymer ratios, inlet temperatures, and atomization pressures, cutting discovery risk and late-stage failures. Vendors now sell modular skids that can be re-configured for new APIs, which further encourages uptake and enlarges the pharmaceutical spray drying market.
By Service Type: CDMO Outsourcing Reshapes Value Chain
In-house manufacturing controlled 58.21% of revenue in 2025, but CDMO services are set to grow 10.56% CAGR through 2031 as sponsors prioritize speed and capital efficiency. CDMOs lower barriers with validated suites, large excipient inventories, and regulatory know-how.
They pool equipment across multiple clients, pushing overall spray dryer utilization up and unit economics down. The shift concentrates demand in fewer high-capacity hubs, especially Ireland, Switzerland, and North Carolina, which boosts the geographic spread of the pharmaceutical spray drying market and raises competition among service providers.
Geography Analysis
North America contributed 39.52% of the pharmaceutical spray drying market share in 2025. FDA programs that fast-track continuous manufacturing, combined with a deep bench of CDMOs, make the United States the largest single market. Capacity expansions in New Jersey and California focus on high-potency APIs and inhalation powders. Canada and Mexico are fast followers as both nations attract near-shoring contracts from U.S. sponsors that want supply resilience.
Asia-Pacific is forecast to post the highest CAGR at 8.32% through 2031. China, with clusters in Shanghai and Suzhou, offers GMP spray drying at 30%–50% lower cost than Western peers, luring early-phase projects. India’s Production-Linked Incentive scheme offsets capital outlays by up to 20%, boosting installations in Hyderabad and Ahmedabad. Japan and South Korea focus on high-value biologic dispersions, while Australia supplies clinical trial material for oncology and rare disease studies.
Europe ranks as the second-largest regional segment. Strict solvent-emission rules favor closed-loop installations, and equipment makers like GEA and Syntegon reinforce the leadership position. Ireland, Germany, and Switzerland cluster CDMO capacity that serves both regional and transatlantic clients. Meanwhile, South America and the Middle East remain nascent pockets, installing pilot units to localize excipient production and reduce import dependence, yet their combined contribution to the pharmaceutical spray drying market size stays modest.
Competitive Landscape
The pharmaceutical spray drying market shows moderate fragmentation. Equipment vendors such as GEA Group, BÜCHI Labortechnik, and SPX FLOW sell systems that range from USD 100,000 lab skids to USD 10 million commercial plants. Service providers including Hovione, Catalent, Lonza, and Seqens differentiate on containment, regulatory record, and digital analytics. Large pharmaceutical companies such as Pfizer and Novartis still run captive suites, mainly to guard proprietary know-how and ensure on-time supply.
Adoption of closed-loop continuous lines gives first movers a cost and compliance edge. GEA’s ConsiGma platform illustrates this shift by unifying spray drying, granulation, and tableting within one environment[3]“ConsiGma Continuous Manufacturing Platform,” GEA Group, gea.com. Smaller vendors such as ProCepT innovate in modular pilots that shorten tech-transfer time to six months, which suits biotech budgets. Over the forecast horizon, consolidation is likely as emission and validation rules grow stricter, raising capital thresholds. Niche firms that embed AI-driven digital twins may capture share because they cut failure rates and improve batch predictability, but they will need strategic partnerships with CDMOs to scale.
Pharmaceutical Spray Drying Industry Leaders
European SprayDry Technologies
SPX FLOW Inc.
GEA Group AG
BÜCHI Labortechnik AG
Shandong Tianli Drying Tech
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- October 2025: Hovione completed a USD 100 million expansion in New Jersey that doubled its U.S. spray drying capacity.
- October 2025: Codis launched as a global CDMO specializing in commercial-scale spray drying, amorphous solid dispersions, and particle-engineering technologies.
Global Pharmaceutical Spray Drying Market Report Scope
As per the scope of the report, spray drying is a drying technology used in the pharmaceutical business to turn liquid or slurry into a dry or coarse powder with the help of hot gas. It is commonly used to make active pharmaceutical ingredients (APIs) in medications.
The pharmaceutical spray drying market is segmented by application, formulation type, spray dryer type, scale, service type, and geography. The application segment of the pharmaceutical spray drying market is further segmented into excipient production, encapsulation, enhancing bioavailability, and other applications. By formulation type, the market is segmented into small-molecule APIs, biologics & peptides, vaccines (including mRNA/LNP), inhalable formulations, and others. By spray dryer type, the market is segmented into rotary atomizer, nozzle atomizer, fluidized/closed-loop, and others. By scale, the market is segmented into laboratory/pilot and commercial-scale. By service type, the market is segmented into in-house manufacturing and contract (CDMO/CMO) services. The geography segment is further segregated into North America, Europe, Asia-Pacific, the Middle East, Africa, and South America. The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD) for the above segments.
| Excipient Production |
| Encapsulation |
| Enhancing Bioavailability |
| Other Applications |
| Small-Molecule APIs |
| Biologics & Peptides |
| Vaccines (Incl. mRNA/LNP) |
| Inhalable Formulations |
| Others |
| Rotary Atomizer |
| Nozzle Atomizer |
| Fluidized / Closed-Loop |
| Others |
| Laboratory / Pilot |
| Commercial-Scale |
| In-house Manufacturing |
| Contract (CDMO/CMO) Services |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Application | Excipient Production | |
| Encapsulation | ||
| Enhancing Bioavailability | ||
| Other Applications | ||
| By Formulation Type | Small-Molecule APIs | |
| Biologics & Peptides | ||
| Vaccines (Incl. mRNA/LNP) | ||
| Inhalable Formulations | ||
| Others | ||
| By Spray Dryer Type | Rotary Atomizer | |
| Nozzle Atomizer | ||
| Fluidized / Closed-Loop | ||
| Others | ||
| By Scale | Laboratory / Pilot | |
| Commercial-Scale | ||
| By Service Type | In-house Manufacturing | |
| Contract (CDMO/CMO) Services | ||
| Geography | North America | United States |
| Canada | ||
| Mexico | ||
| Europe | Germany | |
| United Kingdom | ||
| France | ||
| Italy | ||
| Spain | ||
| Rest of Europe | ||
| Asia-Pacific | China | |
| India | ||
| Japan | ||
| Australia | ||
| South Korea | ||
| Rest of Asia-Pacific | ||
| Middle East and Africa | GCC | |
| South Africa | ||
| Rest of Middle East and Africa | ||
| South America | Brazil | |
| Argentina | ||
| Rest of South America | ||


Key Questions Answered in the Report
What CAGR is forecast for pharmaceutical spray drying between 2026 and 2031?
The market is projected to grow at a 7.32% CAGR over 2026-2031.
Which region is expanding the fastest?
Asia-Pacific is advancing at an 8.32% CAGR, driven by capacity gains in China and India.
How large was North America’s market share in 2025?
North America held 39.52% of global share in 2025.
Why are closed-loop systems gaining popularity?
They recover up to 98% of solvents, meet strict VOC rules, and reduce raw-material costs by up to USD 500,000 a year.
Which application segment is growing the quickest?
Bioavailability enhancement, expanding at a 9.85% CAGR through 2031.
What capital is needed for a GMP spray drying suite?
A commercial GMP unit typically requires USD 10 million-USD 15 million, including cleanroom and solvent-recovery infrastructure.
Page last updated on:










