France In-Vitro Diagnostics Market Analysis by Mordor Intelligence
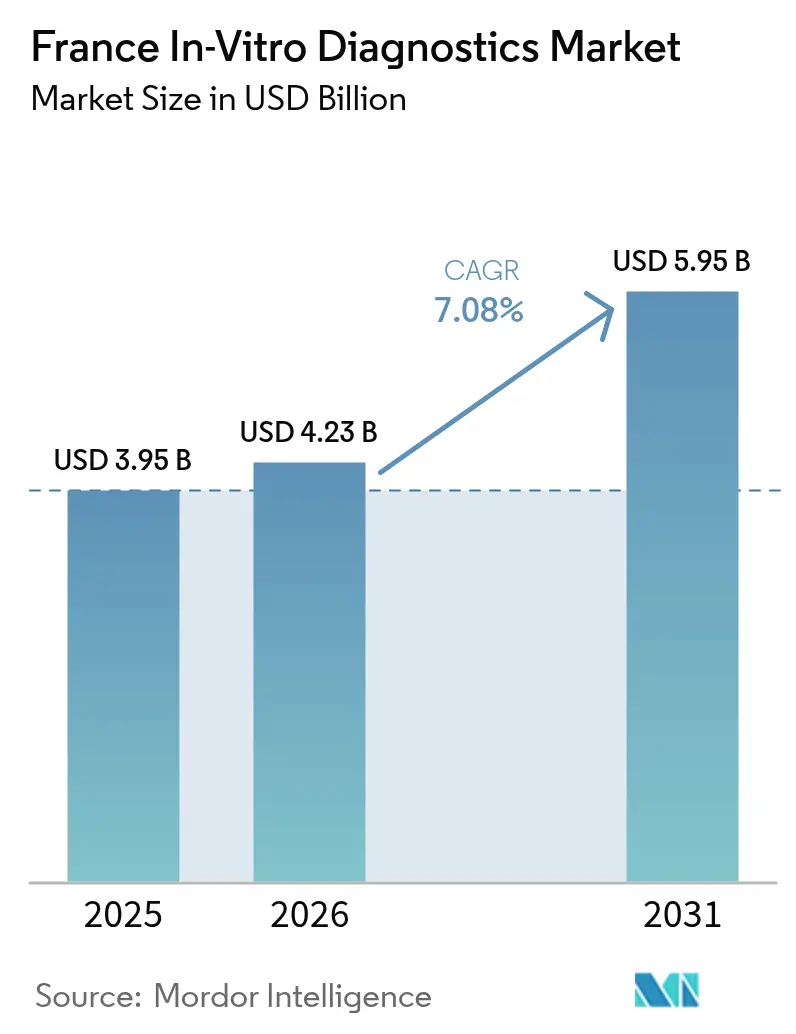
France in vitro diagnostics market size in 2026 is estimated at USD 4.23 billion, growing from 2025 value of USD 3.95 billion with 2031 projections showing USD 5.95 billion, growing at 7.08% CAGR over 2026-2031. Diagnostic testing underpins roughly 70% of clinical decisions and continues to gain relevance as chronic disease cases rise and preventive‐care models expand. Regulatory tightening under the European Union’s In Vitro Diagnostic Regulation (IVDR) is lengthening approval cycles yet driving demonstrable quality gains. Laboratory consolidation, especially among investor-backed chains, is steering volumes toward high-throughput hubs while home-based testing platforms widen patient access. Technology convergence—automation, artificial intelligence, and digital connectivity—remains the pivotal competitive lever as suppliers look to improve turnaround time, accuracy, and data integration[1]Organisation for Economic Co-operation and Development, “Health at a Glance: Europe 2024,” oecd.org.
Key Report Takeaways
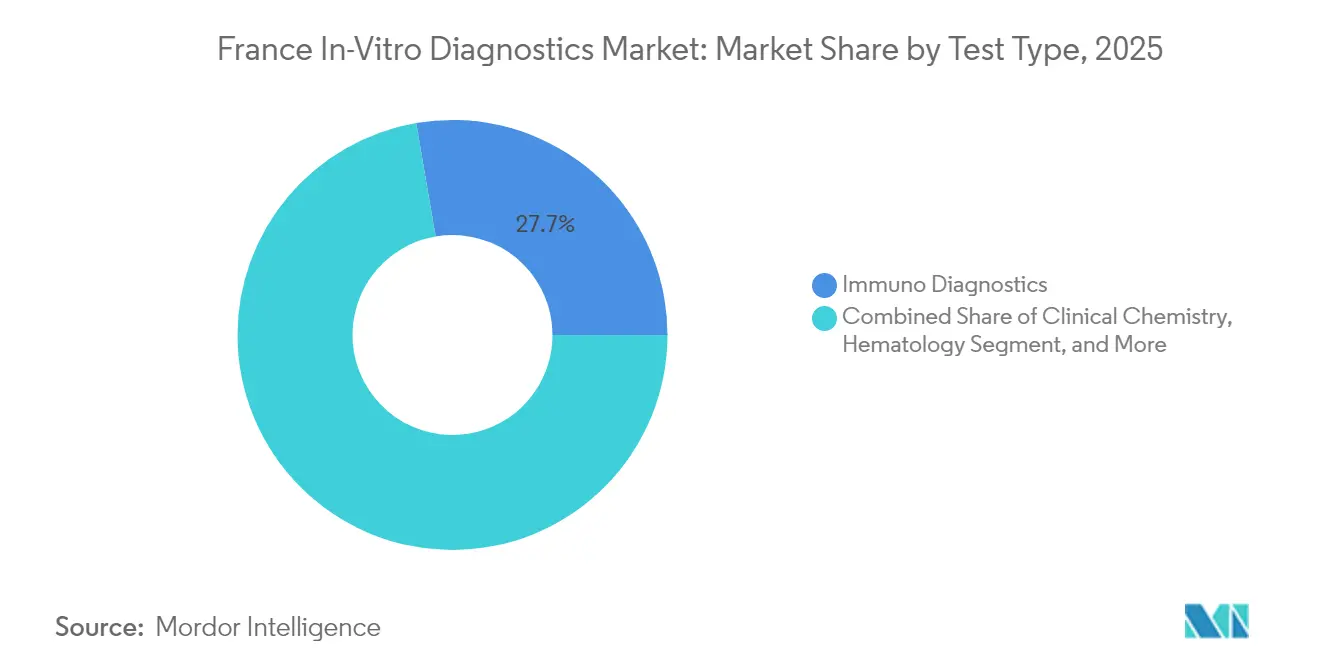
- By test type, Immuno Diagnostics led with 27.70% revenue share in 2025, whereas Molecular Diagnostics is forecast to advance at a 9.18% CAGR through 2031.
- By product & service, Reagents & Kits accounted for 64.80% of the France in vitro diagnostics market share in 2025; Software & Services are poised to grow at a 11.74% CAGR to 2031.
- By specimen, blood testing commanded 44.60% share of the France in vitro diagnostics market size in 2025, while salivary testing will expand at a 10.08% CAGR during 2026-2031.
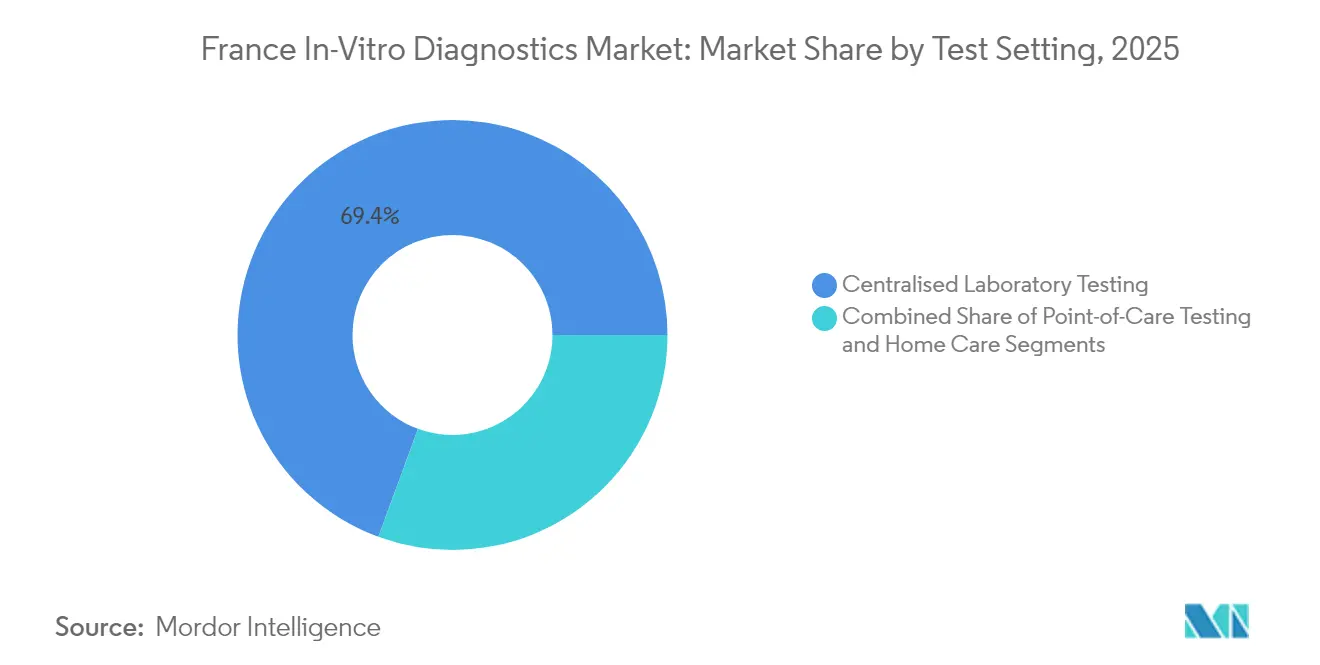
- By test setting, centralized laboratories captured 69.40% of the France in vitro diagnostics market in 2025; self-testing solutions are rising at an 10.92% CAGR to 2031.
- By application, infectious disease diagnostics held 29.90% share of the France in vitro diagnostics market size in 2025 and oncology diagnostics is progressing at a 9.62% CAGR through 2031.
- By end user, independent diagnostic laboratories represented 44.95% of the France in vitro diagnostics market share in 2025, while home-care users are on track for a 11.56% CAGR over the outlook period.
Note: Market size and forecast figures in this report are generated using Mordor Intelligence’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
France In-Vitro Diagnostics Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Chronic & infectious disease surge | +2.1% | National—highest in large metro areas | Long term (≥ 4 years) |
| Reimbursement expansion for high-value tests | +1.5% | National—roll-out starts in major cities | Medium term (2-4 years) |
| Laboratory automation & digital workflows | +1.3% | National—concentrated in chain laboratories | Medium term (2-4 years) |
| Private-lab consolidation | +0.7% | Urban & suburban clusters | Short term (≤ 2 years) |
| Consumer shift to near-patient testing | +0.6% | Early uptake in Paris, Lyon, Marseille | Medium term (2-4 years) |
| Source: Mordor Intelligence | |||
Escalating chronic and infectious disease burden expanding test volumes
France’s aging profile and rising multimorbidity are enlarging test menus across chemistry, immunoassay, and molecular panels. The share of citizens aged ≥65 is projected to reach 29% by 2050, sustaining high diagnostic demand. Infectious disease panels still represent 30.2% of application revenues, reflecting vigilance after the COVID-19 crisis. Antimicrobial-resistance surveillance is accelerating the uptake of rapid molecular assays that identify pathogens and resistance markers in hours rather than days. Preventive screening programs embed testing into routine care pathways, further lifting volumes across national laboratories and community settings.
National health insurance reimbursement expansion for high-value diagnostics
Policy makers are moving toward ‘coverage-with-evidence’ schemes that reward assays delivering clear clinical utility. Companion diagnostics benefit first, aligning with precision oncology regimens that require biomarker confirmation before targeted therapy initiation. Government reimbursement also extends to select digital diagnostics, incentivizing interoperability between test platforms and electronic health records. This environment encourages innovation while nudging suppliers to prove real-world outcome gains.
Rapid laboratory automation and digital workflow adoption enhancing throughput
Multi-site chains deploy total laboratory automation lines, robotics, and image-analysis algorithms to process >1,000 samples daily with minimal manual intervention. Integrating laboratory information systems with hospital records reduces transcription errors and supports fast clinical decision making. Flexible data architectures enable dynamic load balancing, reallocating instruments in real time to match fluctuating sample inflows.
Rising consumer preference for near-patient and home-based testing solutions
Self-sampling kits for diabetes, infectious disease screening, and fertility tracking gain traction as users seek privacy and convenience. Uptake accelerated during the pandemic, familiarizing consumers with nasal swabs and digital result portals. Mobile apps now interpret results, trend data, and transmit findings securely to physicians, bolstering chronic-disease monitoring programs.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Stringent IVDR Compliance Increasing Time-to-Market | -0.8% | National, with greater impact on SMEs | Medium term (2-4 years) |
| Shortage of Qualified Medical Biologists & Technicians Limiting Capacity Expansion | -1.2% | National, with acute impact in rural areas | Long term (≥ 4 years) |
| Low-Cost Self-Testing Alternatives Cannibalising Central-Lab Revenues | -0.5% | National, with higher penetration in urban areas | Short term (≤ 2 years) |
| Source: Mordor Intelligence | |||
Stringent IVDR compliance increasing time-to-market
The IVDR imposes a risk-based device classification and robust clinical-evidence dossier, stretching approval cycles for innovative assays[2]EUR-Lex, “Regulation (EU) 2024/1860,” eur-lex.europa.eu. July 2024 amendments added mandatory supply-shortage notifications and phased Eudamed registration, further intensifying administrative load. More than 70% of manufacturers have redirected resources to regulatory functions, delaying product launches and potentially limiting test availability during the transition period.
Shortage of qualified medical biologists and technicians limiting capacity expansion
France lists laboratory professions among its top workforce shortages, with retirements outpacing new entrants[3]World Health Organization, “Health Workforce Shortage in Europe,” who.int. Rural regions feel the pinch most acutely, experiencing longer turnaround times and service gaps. Automation mitigates repetitive workload but cannot replace specialist interpretation of complex results. Stakeholders pursue scholarship programs and cross-training initiatives, yet near-term relief remains constrained.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Test Type: Molecular diagnostics redefining clinical practice
Immuno Diagnostics secured 27.70% of the France in vitro diagnostics market share in 2025, supported by its role in routine hormone, autoimmune, and infectious-disease panels. Large installed analyzer bases and reagent tie-ins ensure stable demand. Molecular Diagnostics, projected to expand at a 9.18% CAGR between 2026 and 2031, increasingly permeates oncology, infectious disease, and hereditary-disease management. Platform trends favor multiplex PCR and next-generation sequencing, shrinking turnaround time from days to hours. Integrated devices such as BIOFIRE SPOTFIRE consolidate multiple respiratory targets into a single cartridge, underscoring the shift toward syndromic panels. Clinical Chemistry, Hematology, and Coagulation continue to provide core hospital metrics, though revenue growth trails molecular assays because of commoditized pricing. Point-of-care cartridges address decentralized needs, broadening access in emergency and outpatient contexts.
Growing emphasis on precision medicine propels companion diagnostics that identify actionable genomic alterations. Laboratories adopt automated extraction and library-prep stations to handle rising sample numbers without proportionate staff increases. This adoption cements molecular testing’s trajectory toward mainstream use, even for conditions historically monitored by immunoassay or microscopy. As a result, the France in vitro diagnostics market expects a rebalanced revenue mix, with molecular diagnostics capturing a progressively larger slice of overall spending.

By Product & Service: Software integration driving value creation
Reagents & Kits captured 64.80% of the France in vitro diagnostics market in 2025, reflecting the consumables-based economics of clinical testing. Proprietary chemistries with demonstrated sensitivity improvements preserve premium pricing, especially in viral-load and oncology panels. Instruments deliver lower share yet underpin long-term customer lock-in, as analyzer selection dictates future reagent pipelines. Software & Services, growing at 11.74% CAGR to 2031, provide laboratories with analytics, quality-control dashboards, and AI-driven decision support. Healthcare networks allocate capital toward interoperable middleware that bridges analyzer outputs and hospital information systems, reinforcing vendor relationships beyond physical hardware supply.
Service contracts now bundle remote monitoring, predictive maintenance, and workflow optimization consulting. This shift positions solution providers as partners in cost containment and regulatory compliance rather than mere equipment vendors. Consequently, software revenues buffer cyclical capital spending, smoothing supplier cash flows and elevating overall customer lifetime value within the France in vitro diagnostics market.
By Specimen: Saliva testing gaining clinical acceptance
Blood specimens remained dominant at 44.60% of the France in vitro diagnostics market size in 2025 because of their multiparametric biomarker richness and entrenched phlebotomy routines. Automated hematology and chemistry lines sustain high throughput and consistent quality metrics. Urine testing occupies the next tier, leveraging its non-invasive collection for metabolic and renal surveillance. Salivary diagnostics, forecast to rise at a 10.08% CAGR through 2031, benefit from painless collection suited for pediatric, geriatric, and remote contexts. Advanced spectrometry and sequencing now detect oncogenic mutations, cortisol rhythms, and viral RNA in micro-volume saliva, broadening clinical readiness.
Investments in stabilizing buffers extend sample viability during transport, enabling mail-in programs that connect rural patients to urban reference labs. Tissue biopsies and stool samples continue to anchor oncology and gastroenterology workflows, albeit with modest growth as liquid biopsy and non-invasive screening options gain traction.
By Test Setting: Self-testing revolution reshaping access
Centralized laboratories processed 69.40% of national test volumes in 2025, leveraging automated tracks and robotics for speed, standardization, and cost control. High fixed costs are offset by heavy throughput, with private-equity-backed chains optimizing logistics across hub-and-spoke networks. Point-of-care testing fills critical gaps in emergency wards and physician offices, providing decision-grade results within minutes. Self-testing platforms, posting an 10.92% CAGR to 2031, empower consumers through intuitive sample collection and smartphone-enabled result interpretation. Regulatory bodies issued new guidance on labeling, digital instructions, and post-market surveillance to safeguard test accuracy outside clinical environments.
Cloud-linked devices feed longitudinal datasets into telehealth consults, supporting medication titration and lifestyle counseling. This patient-centric evolution lifts overall France in vitro diagnostics market penetration, especially among populations that previously faced mobility, time, or stigma barriers to routine testing.

By Application: Oncology diagnostics driving precision medicine
Infectious disease panels retained 29.90% share of the France in vitro diagnostics market in 2025. Respiratory multiplex assays, sexually transmitted infection screens, and antimicrobial-resistance profiling form the backbone of public-health surveillance. Diabetes monitoring remains sizable thanks to steady prevalence and mandatory HbA1c tracking. Oncology diagnostics are projected to increase at a 9.62% CAGR during 2026-2031 as liquid biopsies and comprehensive genomic profiling transition from specialty centers to broader clinical use. Circulating tumor DNA tests complement tissue biopsy, guiding therapy selection and relapse monitoring without invasive procedures. Cardiovascular biomarker panels evolve toward multi-analyte risk scores, and prenatal testing shifts to non-invasive cell-free DNA approaches.
Growing payer acceptance of outcome-linked reimbursement accelerates advanced cancer diagnostics. Laboratories partner with oncology clinics to integrate genomic reports directly into tumor boards, streamlining precision-therapy decisions and elevating the strategic importance of oncology within the overall France in vitro diagnostics market.
By End User: Home-care users driving market expansion
Independent diagnostic laboratories held 44.95% France in vitro diagnostics market share in 2025, benefiting from economies of scale, specialized expertise, and robust purchasing power secured through consolidation. Hospital laboratories remain indispensable for acute care, offering round-the-clock testing and advanced esoteric panels critical to inpatient management. Physician-office laboratories cater to point-of-care needs but face reimbursement pressure and instrument-utilization challenges.
Home-care and self-testing users will grow at a 11.56% CAGR to 2031 as device miniaturization and digital literacy improve. Portable readers interpret lateral-flow cartridges for infections, metabolic markers, and reproductive health, feeding encrypted data to clinicians. This trend expands total diagnostic spend by engaging previously underserved users and shifting some burden away from overstretched hospital labs. Academic and research institutes continue to pilot emerging modalities such as spatial transcriptomics, ensuring France remains at the forefront of diagnostic innovation.
Geography Analysis
Regional dynamics shape access and innovation within the France in vitro diagnostics market. Paris-Île-de-France hosts the largest concentration of reference laboratories and med-tech headquarters, bolstered by proximity to major teaching hospitals and venture capital. Grand Est’s Alsace Biovalley cluster specializes in molecular diagnostics and imaging, nurturing start-ups via incubator programs and public-private grants. The Auvergne-Rhône-Alpes region, anchored by Lyon, benefits from hospital networks such as Hospices Civils de Lyon, which deploy high-throughput automation suites that process ≥1,200 microbiology samples per day.
Southern hubs around Marseille integrate port logistics with supply-chain efficiency, facilitating reagent imports and analyzer distribution. Government commitment, exemplified by a EUR 25 billion health-science investment fund, sustains R&D pipelines and helps SMEs navigate IVDR compliance hurdles. While urban centers enjoy dense laboratory coverage, rural départements experience technician shortages and longer sample transit times, prompting mobile collection programs and telepathology pilots. Initiatives such as tele-expertise platforms enable rural clinicians to obtain specialized second opinions, narrowing geographic disparities. Cross-border collaboration with Germany, Switzerland, and Italy strengthens market resilience and knowledge transfer. French laboratories participate in EU antimicrobial-resistance surveillance networks, sharing data and alerting authorities to emerging threats. The net result is a geographically balanced yet interconnected ecosystem that sustains demand and fosters innovation across the France in vitro diagnostics market.
Competitive Landscape
Market concentration remains moderate. Roche, Abbott, bioMérieux, Siemens Healthineers, and Becton Dickinson collectively control an estimated 60-65% of national revenues through comprehensive reagent and analyzer portfolios. bioMérieux leverages domestic roots and a 10.3% organic sales jump in the first nine months of 2024 to fortify leadership in syndromic panels. Roche deepens digital connectivity, embedding algorithmic decision support within its cobas lines. Abbott expands point-of-care offerings while linking home glucose meters to cloud dashboards.
Specialized entrants target high-growth niches—liquid biopsy, non-invasive prenatal testing, and AI-assisted digital pathology—challenging incumbents on agility and depth. Becton Dickinson’s announced spin-off of its Biosciences and Diagnostic Solutions unit underscores portfolio optimization trends. Partnerships proliferate, pairing analyzer manufacturers with software firms to produce integrated end-to-end solutions. Procurement contracts increasingly stipulate performance-based metrics, compelling suppliers to prove throughput, uptime, and clinical impact.
Technology differentiation focuses on multiplexing capacity, automation compatibility, and cybersecurity safeguards. Suppliers offering seamless reagent-instrument-software stacks gain an edge, especially within consolidated laboratory chains negotiating at national scale. Meanwhile, government emphasis on domestic manufacturing resilience during supply disruptions encourages dual-sourcing strategies, granting smaller French innovators entry points into hospital formularies.
France In-Vitro Diagnostics Industry Leaders
Thermo Fischer Scientific Inc
QIAGEN N.V.
Siemens Healthcare GmbH
F. Hoffmann-La Roche AG
Abbott Laboratories
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2025: The French National Authority for Health published its 2025-2030 strategic roadmap prioritizing integrated care, demographic change adaptation, and digital-technology adoption, signaling ongoing support for high-value diagnostics.
- July 2024: Regulation (EU) 2024/1860 introduced phased Eudamed roll-out and six-month supply-disruption notification mandates for IVDs, refining IVDR implementation timelines.
France In-Vitro Diagnostics Market Report Scope
As per the scope of this report, in vitro diagnostics involves medical devices and consumables that are utilized to perform in vitro tests on various biological samples. They are used for the diagnosis of various medical conditions, such as diabetes, cancer, and others. The France In-Vitro Diagnostics Market is Segmented by Test Type (Clinical Chemistry, Molecular Diagnostics, Immuno Diagnostics, and Other Techniques), Product (Instrument, Reagent, and Other Products), Usability (Disposable IVD Devices, Reusable IVD Devices), Application (Infectious Disease, Diabetes, Cancer/Oncology, Cardiology, and Other Applications) and End-users (Diagnostic Laboratories, Hospitals, and Clinics and Other End-users). The report offers the value (in USD million) for the above segments.
| Clinical Chemistry |
| Molecular Diagnostics |
| Immuno Diagnostics |
| Hematology |
| Coagulation |
| Microbiology |
| Point-of-Care Testing |
| Other Test Types |
| Instruments |
| Reagents & Kits |
| Software & Services |
| Blood |
| Urine |
| Saliva |
| Tissue & Biopsy |
| Stool |
| Other Specimens |
| Centralised Laboratory Testing |
| Point-of-Care Testing |
| Self-Testing / Home Care |
| Infectious Disease |
| Diabetes |
| Cancer / Oncology |
| Cardiology |
| Autoimmune Disorders |
| Prenatal & Newborn Screening |
| Other Applications |
| Independent Diagnostic Laboratories |
| Hospital-based Laboratories |
| Physician Office Laboratories |
| Academic & Research Institutes |
| Home Care & Self-Testing Users |
| Other End Users |
| By Test Type | Clinical Chemistry |
| Molecular Diagnostics | |
| Immuno Diagnostics | |
| Hematology | |
| Coagulation | |
| Microbiology | |
| Point-of-Care Testing | |
| Other Test Types | |
| By Product & Service | Instruments |
| Reagents & Kits | |
| Software & Services | |
| By Specimen | Blood |
| Urine | |
| Saliva | |
| Tissue & Biopsy | |
| Stool | |
| Other Specimens | |
| By Test Setting | Centralised Laboratory Testing |
| Point-of-Care Testing | |
| Self-Testing / Home Care | |
| By Application | Infectious Disease |
| Diabetes | |
| Cancer / Oncology | |
| Cardiology | |
| Autoimmune Disorders | |
| Prenatal & Newborn Screening | |
| Other Applications | |
| By End User | Independent Diagnostic Laboratories |
| Hospital-based Laboratories | |
| Physician Office Laboratories | |
| Academic & Research Institutes | |
| Home Care & Self-Testing Users | |
| Other End Users |


Key Questions Answered in the Report
How large will diagnostic testing revenue be in France by 2031?
The France in vitro diagnostics market size is projected at USD 5.95 billion by 2031 under a 7.08% CAGR.
Which segment shows the fastest growth momentum?
Molecular diagnostics is forecast at 9.18% CAGR as precision oncology, infectious-disease surveillance, and genetic screening expand.
What drives the move toward at-home testing?
Higher consumer digital literacy, pandemic-era familiarity with self-sampling, and smartphone-linked result interpretation fuel an 10.92% CAGR in self-testing volumes.
How does IVDR affect French suppliers?
Stricter evidence requirements, phased Eudamed registration, and supply-notification rules extend approval timelines and raise compliance costs, particularly for SMEs.
What workforce challenges loom for laboratories?
France faces a 1.2% CAGR drag from shortages of medical biologists and technicians, especially outside major urban centers, prompting investment in automation and training.
Page last updated on:








