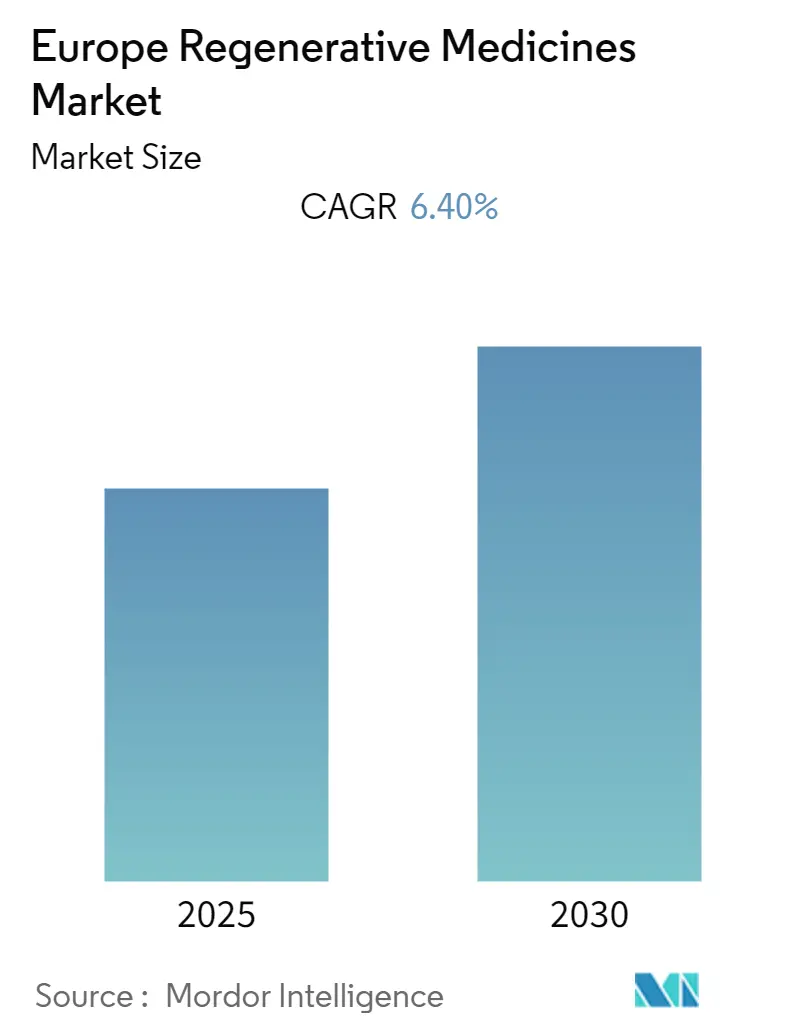
Europe Regenerative Medicines Market Analysis by Mordor Intelligence
The Europe Regenerative Medicines Market is expected to register a CAGR of 6.4% during the forecast period.
The outbreak of the COVID-19 pandemic is anticipated to have a significant impact on the regenerative medicines market in Europe. According to the research article published by European Respiratory Journal in June 2020, titled ''Current status of cell-based therapies for respiratory virus infections: applicability to COVID-19, novel therapeutic approaches such as cell-based therapies, especially using mesenchymal stem cells were found to have demonstrated the safety and efficacy in providing effective treatment in patients with acute respiratory distress syndrome (ARDS). Thus, there has been an increasing focus on developing stem cell-based therapies in treating SARS-CoV-2 viral infection. Thus, owing to the aforementioned factors, the studied market is expected to witness a significant impact during the pandemic.
The key factors propelling the growth of the regenerative medicines market in Europe are the increasing adoption of stem-cell technologies, gene therapies, and tissue engineering, and the technological advancements in regenerative medicines.
According to the clinical trial registry as of August 26th, 2022, there were 1,868 clinical trials of stem cell therapy, of which 82 studies were specifically targeted toward regenerative medicines across the world. This is likely to increase the demand for stem cell-based products and regenerative medicines in the market over the next few years. According to the clinicaltrials.gov database, the majority of active recruiting trials are present in Europe. Acute lymphoblastic leukemia (ALL), acute megakaryoblastic leukemia (AML), and acute graft versus host disease (AGHD) are some of the current indications that are primarily being focused upon by the majority of the universities, research centers, and companies. Thus, it is evident that there are growing initiatives associated with stem cells and regenerative medicines, and it is estimated that over the forecast period, the success rate and further approval will rise gradually, which is primarily expected to drive the growth of the market.
Furthermore, according to the United Nations Population Fund's World Population Dashboard's Statistics 2021, Italy has approximately 23.8% of the population aged 65 and above. Thus, the increasing aging population is prone to chronic diseases as a result, demand for regenerative medicines is predicted to rise over the forecast period.
Moreover, as per the article published in August 2022, titled ''European first as County Durham stroke patient, 28, takes part in stem cell treatment trial, a young patient from County Durham is the first in Europe to take part in a new trial to discover whether stem cell treatment can improve recovery from strokes. Such instances are expected to increase the demand for cell therapy, thereby, boosting the growth of the market over the forecast period.
However, regulatory and ethical issues and the high cost of treatments are expected to hinder the growth of the market.
Europe Regenerative Medicines Market Trends and Insights
Cardiovascular Application is Expected to Cover a Large Share of the Market Over the Forecast Period
Cardiac stem-cell therapy has great potential to prompt myocardial regeneration in patients with ischemic heart disease. The selection of the most suitable cell type is pivotal for its successful application. Various cell types, including crude bone marrow mononuclear cells, skeletal myoblast, and hematopoietic and endothelial progenitors, have already advanced into the clinical arena, based on promising results from different experimental and preclinical studies. Different next-generation cell types are also under investigation, for the treatment of diseased myocardium.
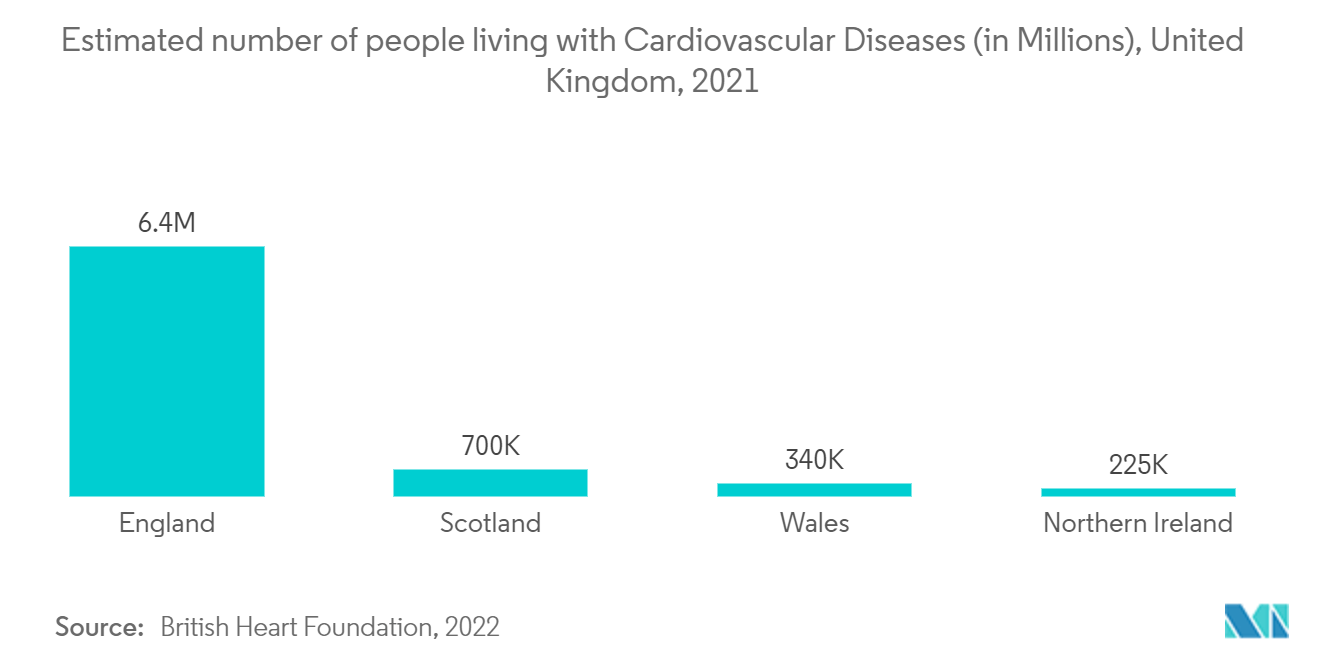
As per the fact sheet published in August 2022, by the British Heart Foundation, around 7.6 million people are living with heart and circulatory diseases in the United Kingdom. Around 4 million males and 3.6 million females are living with heart and circulatory diseases in the United Kingdom. As per the same source, there are 2.3 million people in the United Kingdom living with coronary heart disease (CHD) - around 1.5 million men and 830,000 women. As the incidence of CVD increases in Europe, the demand for cardiac regenerative techniques in applications like cardiac stem (CSCs) and progenitor cells (CPCs) is also expected to increase. This is expected to help the market to grow in the future.
Thus, the segment is anticipated to witness significant growth over the forecast period due to the abovementioned factors.


France is Expected to Dominate the Europe Regenerative Medicines Market
France is expected to dominate the market owing to factors such as the increasing adoption of innovative therapies in the treatment of chronic diseases that will drive industry expansion. The rising incidence of several chronic diseases in the country will further boost product demand. Moreover, high investment in the development of healthcare infrastructure and high spending capacity on healthcare will propel market growth. Moreover, government funding and reimbursement policies in facilities along with new product launches in the region will impel the market size.
In September 2021, Christian Jorgensen, Rosanna Feirrera Lopez, Loïc Le Coz, and Marion Bertrand-Marchand M.D., formed a series of Circles aimed at standardizing orthobiologic treatments through real-world evidence. Unlike a traditional study, all clinicians will be free to use their professional judgment in the use of devices, clinical protocol, and patient selection, but by aggregating and benchmarking their long-term patient outcomes, will begin answering key clinical questions surrounding various regenerative medicine products. This collaborative, titled 'Regenerative Medicine Services of France' planned to enroll its first patient in mid-September.
Moreover, in September 2022, KCAS expanded its transatlantic presence with the acquisition of France-based active biomarkers. Moreover in June 2021, Yposkesi built its second commercial facility for cell and gene therapy manufacturing at its campus in Corbeil-Essonnes, France. Project SKY is intended to be a new global resource for drug developers of biologics. These Developments in the region are expceted to boost the growth of the market in the region over the forecast period.
Therefore, owing to the aforesaid factors the growth of the studied market is anticipated in the France Region.


Competitive Landscape
The Europe regenerative medicines market is moderately competitive, due to the presence of most of the key global players in the region. Many of these global players have their headquarters in European countries, which increases the accessibility of regenerative medicines throughout the continent. The major players in the studied market include Abbvie Inc. (Allergan plc), Medtronic plc, Smith and Nephew (Osiris Therapeutics), Cook Biotech Incorporated, Thermo Fisher Scientific, Merck KGaA, Integra Lifesciences Corporation among others.
Europe Regenerative Medicines Industry Leaders
Merck KGaA
Integra LifeSciences Corporation
Cook Biotech Incorporated
Thermo Fisher Scientific
Smith & Nephew (Osiris Therapeutics)
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- September 2022: Kite announced that the European Commission (EC) approved its CAR T-cell therapy Tecartus (brexucabtagene autoleucel) for the treatment of adult patients 26 years of age and above with relapsed or refractory (r/r) B-cell precursor acute lymphoblastic leukemia (ALL).
- August 2022: Charles River received approval to commercially produce allogeneic cell therapy products for distribution in Europe from European Medicines Agency (EMA).


Europe Regenerative Medicines Market Report Scope
As per the scope of this report, regenerative medicine are used to repair, replace, and regenerate the tissues and organs affected by injury, disease, or the natural aging process. These medicines restore the functionality of cells and tissues and are used in several degenerative disorders, such as dermatology, neurodegenerative diseases, cardiovascular, and orthopedic applications. The Europe Regenerative Medicines market is segmented by Type of Technology, Application, and Geography. By Type of Technology, the market is segmented into Stem Cell Therapy, Biomaterial, Tissue Engineering, and Other Types of Technologies. By Application, the market is segmented into Bone Graft Substitutes, Osteoarticular Diseases, Dermatology, Cardiovascular, Central Nervous System, and Other Applications. By Geography, the market is segmented into Germany, United Kingdom, France, Italy, Spain, and Rest of Europe. The report offers the value (in USD million) for the above segments.
| Stem Cell Therapy |
| Biomaterial |
| Tissue Engineering |
| Other Types of Technologies |
| Bone Graft Substitutes |
| Osteoarticular Diseases |
| Dermatology |
| Cardiovascular |
| Central Nervous System |
| Other Applications |
| Germany |
| United Kingdom |
| France |
| Italy |
| Spain |
| Rest of Europe |
| By Type of Technology | Stem Cell Therapy |
| Biomaterial | |
| Tissue Engineering | |
| Other Types of Technologies | |
| By Application | Bone Graft Substitutes |
| Osteoarticular Diseases | |
| Dermatology | |
| Cardiovascular | |
| Central Nervous System | |
| Other Applications | |
| By Geography | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe |


Key Questions Answered in the Report
What is the current Europe Regenerative Medicines Market size?
The Europe Regenerative Medicines Market is projected to register a CAGR of 6.40% during the forecast period (2025-2030)
Who are the key players in Europe Regenerative Medicines Market?
Merck KGaA, Integra LifeSciences Corporation, Cook Biotech Incorporated, Thermo Fisher Scientific and Smith & Nephew (Osiris Therapeutics) are the major companies operating in the Europe Regenerative Medicines Market.
What years does this Europe Regenerative Medicines Market cover?
The report covers the Europe Regenerative Medicines Market historical market size for years: 2019, 2020, 2021, 2022, 2023 and 2024. The report also forecasts the Europe Regenerative Medicines Market size for years: 2025, 2026, 2027, 2028, 2029 and 2030.
Page last updated on:
Europe Regenerative Medicines Market Report
Statistics for the 2025 Europe Regenerative Medicines market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Europe Regenerative Medicines analysis includes a market forecast outlook for 2025 to 2030 and historical overview. Get a sample of this industry analysis as a free report PDF download.



